Molecular tweezers
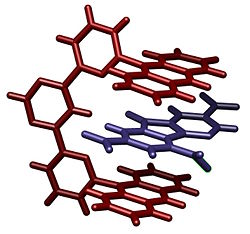
Molecular tweezers, sometimes termed molecular clips, are noncyclic macrocyclic molecular complexes with open cavities capable of binding guests.[3] The open cavity of the molecular tweezer may bind guests using non-covalent bonding which includes hydrogen bonding, metal coordination, hydrophobic forces, van der Waals forces, π-π interactions, and/or electrostatic effects. These complexes are a subset of macrocyclic molecular receptors and their structure is that the two "arms" that bind the guest molecule between them are only connected at one end.
Examples
One example of molecular tweezers has been reported by Lehn and coworkers. This molecule is capable of binding aromatic guests.[1] The molecular tweezers are composed of two anthracene arms held at a distance that allows aromatic guests to gain π-π interactions from both.
Another class of molecular tweezers is composed of two substituted porphyrin macrocycles tethered by a amide linker with a variable length. This example of a molecular tweezer shows the potential mobility of this class of molecules, as the orientation of the porphyrin planes which comprise the tweezer can be altered by the guest which is bound [4]
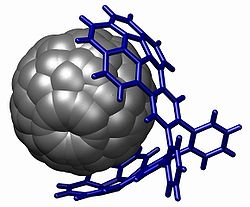
Yet another structure for molecular tweezers which specifically bind fullerenes is called a buckycatcher and has been reported.[2] This molecular tweezer is composed of two concaved corannulene pincers that complement the surface of the convex fullerene guest. An association constant (Ka) of 8600 M-1 between the host buckycatcher and a C60 fullerene was calculated using 1H NMR spectroscopy.
The above examples show the potential reactivity and specificity of these molecules. The binding site between the planes of the tweezer can be designed to bind to an appropriate guest with resulting high association constants and consequent stability, depending on the design of the tweezer. That makes this overall class of macromolecule truly a synthetic molecular receptor.
External links
- Journal of Chemical Education Featured Molecules December 2004: Nanoscale Molecular Tweezers and article
- Crystalmaker molecular tweezers
References
- ↑ 1.0 1.1 A. Petitjean, R. G. Khoury, N. Kyritsakas and J. M. Lehn (2004). "Dynamic Devices. Shape Switching and Substrate Binding in Ion-Controlled Nanomechanical Molecular Tweezers". J. Am. Chem. Soc. 126 (21): 6637–6647. doi:10.1021/ja031915r.
- ↑ 2.0 2.1 A. Sygula, F. R. Fronczek, R. Sygula, P. W. Rabideau and M. M. Olmstead (2007). "A Double Concave Hydrocarbon Buckycatcher". J. Am. Chem. Soc. 129 (13): 3842–3843. doi:10.1021/ja070616p.
- ↑ Frank-Gerrit Klärner and Björn Kahlert (2003). "Molecular Tweezers and Clips as Synthetic Receptors. Molecular Recognition and Dynamics in Receptor-Substrate Complexes". Acc. Chem. Res. 36 (12): 919–932. doi:10.1021/ar0200448.
- ↑ X. Huang, N. Fujioka, G. Pescitelli, F. Koehn, R. T. Williamson, K. Nakanishi and N. Berova (2002). "Absolute Configurational Assignments of Secondary Amines by CD-sensitive Dimeric Zinc Porphyrin Host". J. Am. Chem. Soc. 124 (17): 10320–10335. doi:10.1021/ja020520p.