Meclofenamate
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Kiran Singh, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
Warning
See full prescribing information for complete Boxed Warning.
Cardiovascular Risk
Gastrointestinal Risk
|
Overview
Meclofenamate is an analgesic that is FDA approved for the treatment of juvenile arthritis,rheumatoid arthritis,osteoarthritis,dysmenorrhea,ankylosing spondylitis and gouty arthritis. There is a Black Box Warning for this drug as shown here. Common adverse reactions include diarrhea.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- Meclofenamate sodium capsules are indicated:
- For reduction of fever in adults
- For relief of mild to moderate pain in adults
- For relief of signs and symptoms of juvenile arthritis.
- For relief of the signs and symptoms of rheumatoid arthritis
- For relief of the signs and symptoms of osteoarthritis.
- For treatment of primary dysmenorrhea.
- For acute or long-term use in the relief of signs and symptoms of the following:
- Acute painful shoulder (Acute subacromial bursitis/supraspinatus tendinitis)
- Acute gouty arthritis
- Meclofenamate sodium capsules are also indicated for the treatment of idiopathic heavy menstrual blood loss.
- As with all non-steroidal anti-inflammatory drugs, selection of meclofenamate sodium capsules require a careful assessment of the benefit/risk ratio.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Meclofenamate in adult patients.
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Meclofenamate in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
- There is limited information regarding FDA-Labeled Use of Meclofenamate in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Meclofenamate in pediatric patients.
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Meclofenamate in pediatric patients.
Contraindications
- Meclofenamate sodium capsules are contraindicated in patients with known hypersensitivity to meclofenamate sodium.
- Meclofenamate sodium capsules should not be given to patients who have experienced asthma, urticaria, or allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic-like reactions to NSAIDs have been reported in such patients.
- Meclofenamate sodium capsules are contraindicated for the treatment of peri-operative pain in the setting of coronary artery bypass graft (CABG) surgery.
Warnings
|
Warning
See full prescribing information for complete Boxed Warning.
Cardiovascular Risk
Gastrointestinal Risk
|
Cardiovascular Effects
- Cardiovascular Thrombotic Events: Clinical trials of several COX-2 selective and nonselective NSAIDs of up to 3 years duration have shown an increased risk of serious cardiovascular (CV) thrombotic events, myocardial infarction, and stroke, which can be fatal. All NSAIDs, both COX-2 selective and nonselective, may have a similar risk. Patients with known CV disease or risk factors for CV disease may be at greater risk. To minimize the potential risk for an adverse CV event in patients treated with an NSAID, the lowest effective dose should be used for the shortest duration possible. Physicians and patients should remain alert for the development of such events, even in the absence of previous CV symptoms. Patients should be informed about the signs and/or symptoms of serious CV events and the steps to take if they occur.
- There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID does increase the risk of serious GI events.
- Two large, controlled, clinical trials of a COX-2 selective NSAID for the treatment of pain in the first 10 to 14 days following CABG surgery found an increased incidence of myocardial infarction and stroke.
- Hypertension: NSAIDs, including meclofenamate sodium, can lead to onset of new hypertension or worsening of preexisting hypertension, either of which may contribute to the increased incidence of CV events. Patients taking thiazides or loop diuretics may have impaired response to these therapies when taking NSAIDs. NSAIDs, including meclofenamate sodium, should be used with caution in patients with hypertension. Blood pressure (BP) should be monitored closely during the initiation of NSAID treatment and throughout the course of therapy.
- Congestive Heart Failure and Edema: Fluid retention and edema have been observed in some patients taking NSAIDs. Meclofenamate sodium should be used with caution in patients with fluid retention or heart failure.
Gastrointestinal Effects
- Risk of Ulceration, Bleeding, and Perforation: NSAIDs, including meclofenamate sodium, can cause serious gastrointestinal (GI) adverse events including inflammation, bleeding, ulceration, and perforation of the stomach, small intestine, or large intestine, which can be fatal. These serious adverse events can occur at any time, with or without warning symptoms, in patients treated with NSAIDs. Only one in five patients, who develop a serious upper GI adverse event on NSAID therapy, is symptomatic. Upper GI ulcers, gross bleeding, or perforation caused by NSAIDs occur in approximately 1% of patients treated for 3 to 6 months, and in about 2% to 4% of patients treated for one year. These trends continue with longer duration of use, increasing the likelihood of developing a serious GI event at some time during the course of therapy. However, even short-term therapy is not without risk.
- NSAIDs should be prescribed with extreme caution in those with a prior history of ulcer disease or gastrointestinal bleeding. Patients with a prior history of peptic ulcer disease and/or gastrointestinal bleeding who use NSAIDs have a greater than 10-fold increased risk for developing a GI bleed compared to patients with neither of these risk factors. Other factors that increase the risk for GI bleeding in patients treated with NSAIDs include concomitant use of oral corticosteroids or anticoagulants, longer duration of NSAID therapy, smoking, use of alcohol, older age, and poor general health status. Most spontaneous reports of fatal GI events are in elderly or debilitated patients and therefore, special care should be taken in treating this population.
- To minimize the potential risk for an adverse GI event in patients treated with an NSAID, the lowest effective dose should be used for the shortest possible duration. Patients and physicians should remain alert for signs and symptoms of GI ulceration and bleeding during NSAID therapy and promptly initiate additional evaluation and treatment if a serious GI adverse event is suspected. This should include discontinuation of the NSAID until a serious GI adverse event is ruled out. For high risk patients, alternate therapies that do not involve NSAIDs should be considered.
Renal Effects
- Long-term administration of NSAIDs has resulted in renal papillary necrosis and other renal injury. Renal toxicity has also been seen in patients in whom renal prostaglandins have a compensatory role in the maintenance of renal perfusion. In these patients, administration of a non-steroidal anti-inflammatory drug may cause a dose dependent reduction in prostaglandin formation and, secondarily, in renal blood flow, which may precipitate overt renal decompensation. Patients at greatest risk of this reaction are those with impaired renal function, heart failure, liver dysfunction, those taking diuretics and ACE inhibitors, and the elderly. Discontinuation of NSAID therapy is usually followed by recovery to the pretreatment state.
- Advanced Renal Disease: No information is available from controlled clinical studies regarding the use of meclofenamate sodium in patients with advanced renal disease. Therefore, treatment with meclofenamate sodium is not recommended in these patients with advanced renal disease. If meclofenamate sodium therapy must be initiated, close monitoring of the patient's renal function is advisable.
Anaphylactoid Reactions
- As with other NSAIDs, anaphylactoid reactions may occur in patients without known prior exposure to meclofenamate sodium. Meclofenamate sodium should not be given to patients with the aspirin triad. This symptom complex typically occurs in asthmatic patients who experience rhinitis with or without nasal polyps, or who exhibit severe, potentially fatal bronchospasm after taking aspirin or other NSAIDs. Emergency help should be sought in cases where an anaphylactoid reaction occurs.
Skin Reactions
- NSAIDs, including meclofenamate sodium, can cause serious skin adverse events such as exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. These serious events may occur without warning. Patients should be informed about the signs and symptoms of serious skin manifestations and use of the drug should be discontinued at the first appearance of skin rash or any other sign of hypersensitivity.
Pregnancy
- In late pregnancy, as with other NSAIDs, meclofenamate sodium should be avoided because it may cause premature closure of the ductus arteriosus.
Precautions
General
- Meclofenamate sodium cannot be expected to substitute for corticosteroids or to treat corticosteroid insufficiency. Abrupt discontinuation of corticosteroids may lead to disease exacerbation. Patients on prolonged corticosteroid therapy should have their therapy tapered slowly if a decision is made to discontinue corticosteroids.
- The pharmacological activity of meclofenamate sodium in reducing fever and inflammation may diminish the utility of these diagnostic signs in detecting complications of presumed noninfectious, painful conditions.
Hepatic Effects
- Borderline elevations of one or more liver tests may occur in up to 15% of patients taking NSAIDs including meclofenamate sodium. These laboratory abnormalities may progress, may remain unchanged, or may be transient with continuing therapy. Notable elevations of ALT or AST (approximately 3 or more times the upper limit of normal) have been reported in approximately 1% of patients in clinical trials with NSAIDs. In addition, rare cases of severe hepatic reactions, including jaundice and fatal fulminant hepatitis, liver necrosis and hepatic failure, some of them with fatal outcomes have been reported.
- A patient with symptoms and/or signs suggesting liver dysfunction, or in whom an abnormal liver test has occurred, should be evaluated for evidence of the development of a more severe hepatic reaction while on therapy with meclofenamate sodium. If clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash, etc.), meclofenamate sodium should be discontinued.
Hematological Effects
- Anemia is sometimes seen in patients receiving NSAIDs, including meclofenamate sodium. This may be due to fluid retention, occult or gross GI blood loss, or an incompletely described effect upon erythropoiesis. Patients on long-term treatment with NSAIDs, including meclofenamate sodium, should have their hemoglobin or hematocrit checked if they exhibit any signs or symptoms of anemia.
- NSAIDs inhibit platelet aggregation and have been shown to prolong bleeding time in some patients. Unlike aspirin, their effect on platelet function is quantitatively less, of shorter duration, and reversible. Patients receiving meclofenamate sodium who may be adversely affected by alterations in platelet function, such as those with coagulation disorders or patients receiving anticoagulants, should be carefully monitored.
Preexisting Asthma
- Patients with asthma may have aspirin-sensitive asthma. The use of aspirin in patients with aspirin-sensitive asthma has been associated with severe bronchospasm which can be fatal. Since cross reactivity, including bronchospasm, between aspirin and other non-steroidal anti-inflammatory drugs has been reported in such aspirin-sensitive patients, meclofenamate sodium should not be administered to patients with this form of aspirin sensitivity and should be used with caution in patients with preexisting asthma.
Adverse Reactions
Clinical Trials Experience
Incidence Greater Than 1%
- The following adverse reactions were observed in clinical trials and included observations from more than 2,700 patients, 594 of whom were treated for one year and 248 for at least 2 years.
Gastrointestinal: The most frequently reported adverse reactions associated with meclofenamate sodium involve the gastrointestinal system. In controlled studies of up to 6 months duration, these disturbances occurred in the following decreasing order of frequency with the approximate incidences in parentheses: diarrhea (10% to 33%), nausea with or without vomiting (11%), other gastrointestinal disorders (10%), and abdominal pain1. In long-term uncontrolled studies of up to 4 years duration, one third of the patients had at least one episode of diarrhea some time during meclofenamate sodium therapy.
- In approximately 4% of the patients in controlled studies, diarrhea was severe enough to require discontinuation of meclofenamate sodium. The occurrence of diarrhea is dose related, generally subsides with dose reduction, and clears with termination of therapy. The incidence of diarrhea in patients with osteoarthritis is generally lower than that reported in patients with rheumatoid arthritis.
- Other reactions less frequently reported were pyrosis, flatulence, anorexia, constipation, stomatitis, and peptic ulcer. The majority of the patients with peptic ulcer had either a history of ulcer disease or were receiving concomitant anti-inflammatory drugs, including corticosteroids which are known to produce peptic ulceration.
Cardiovascular: edema
Dermatologic: rash, urticaria, pruritus
Central Nervous System: headache, dizziness
Special Senses: tinnitus
- Incidence between 3% and 9%. Those reactions occurring in 1% to 3% of patients are not marked with an asterisk.
Incidence Less Than 1%—Probably Causally Related
- The following adverse reactions were reported less frequently than 1% during controlled clinical trials and through voluntary reports since marketing. The probability of a causal relationship exists between the drug and these adverse reactions.
Gastrointestinal: bleeding and/or perforation with or without obvious ulcer formation, colitis, cholestatic jaundice
Renal: renal failure
Hematologic: neutropenia, thrombocytopenic purpura, leukopenia, agranulocytosis, hemolytic anemia, eosinophilia, decrease in hemoglobin and/or hematocrit
Dermatologic: erythema multiforme, Stevens-Johnson Syndrome, exfoliative dermatitis
Hepatic: alteration of liver function tests
Allergic: lupus and serum sickness-like symptoms
Incidence Less Than 1%—Causal Relationship Unknown
- Other reactions have been reported but under conditions where a causal relationship could not be established. However, in these rarely reported events, that possibility cannot be excluded. Therefore, these observations are listed to alert physicians.
Cardiovascular: palpitations
Central Nervous System: malaise, fatigue, paresthesia, insomnia, depression
Special Senses: blurred vision, taste disturbances, decreased visual acuity, temporary loss of vision, reversible loss of color vision, retinal changes including macular fibrosis, macular and perimacular edema, conjunctivitis, iritis
Renal: nocturia
Gastrointestinal: paralytic ileus
Dermatologic: erythema nodosum, hair loss
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Meclofenamate in the drug label.
Drug Interactions
- ACE-inhibitors: Reports suggest that NSAIDs may diminish the antihypertensive effect of ACE-inhibitors. This interaction should be given consideration in patients taking NSAIDs concomitantly with ACE-inhibitors.
- Aspirin: When meclofenamate sodium in administered with aspirin, its protein binding is reduced, although the clearance of free meclofenamate sodium is not altered. The clinical significance of this interaction is not known; however, as with other NSAIDs, concomitant administration of meclofenamate sodium capsules and aspirin is not generally recommended because of the potential of increased adverse effects.
- Furosemide: Clinical studies, as well as post-marketing observations, have shown that meclofenamate sodium can reduce the natriuretic effect-of furosemide and thiazides in some patients. This response has been attributed to inhibition of renal prostaglandin synthesis. During concomitant therapy with NSAIDs, the patient should be observed closely for signs of renal failure (see PRECAUTIONS: Renal Effects), as well as to assure diuretic efficacy.
- Lithium: NSAIDs have produced an elevation of plasma lithium levels and a reduction in renal lithium clearance. The mean minimum lithium concentration increased 15% and the renal clearance was decreased by approximately 20%. These effects have been attributed to inhibition of renal prostaglandin synthesis by the NSAID. Thus, when NSAIDs and lithium are administered concurrently, subjects should be observed carefully for signs of lithium toxicity.
- Methotrexate: NSAIDs have been reported to competitively inhibit methotrexate accumulation in rabbit kidney slices. This may indicate that they could enhance the toxicity of methotrexate. Caution should be used when NSAIDs are administered concomitantly with methotrexate.
- Warfarin: The effects of warfarin and NSAIDs on GI bleeding are synergistic, such that users of both drugs together have a risk of serious GI bleeding higher than users of either drug alone.
Use in Specific Populations
Pregnancy
- Teratogenic Effects. Pregnancy Category C: Reproductive studies conducted in rats and rabbits have not demonstrated evidence of developmental abnormalities. However, animal reproduction studies are not always predictive of human response. There are no adequate and well controlled studies in pregnant women.
- Nonteratogenic Effects: Because of the known effects of non-steroidal anti-inflammatory drugs on the fetal cardiovascular system (closure of ductus arteriosus), use during pregnancy (particularly late pregnancy) should be avoided.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Meclofenamate in women who are pregnant.
Labor and Delivery
- In rat studies with NSAIDs, as with other drugs known to inhibit prostaglandin synthesis, an increased incidence of dystocia, delayed parturition, and decreased pup survival occurred. The effects of meclofenamate sodium on labor and delivery in pregnant women are unknown.
Nursing Mothers
- It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human-milk and because of the potential for serious adverse reactions in nursing infants from meclofenamate sodium, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
- Safety and effectiveness in pediatric patients below the age of 14 have not been established.
Geriatic Use
- As with any NSAIDs, caution should be exercised in treating the elderly (65 years and older).
Gender
There is no FDA guidance on the use of Meclofenamate with respect to specific gender populations.
Race
There is no FDA guidance on the use of Meclofenamate with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Meclofenamate in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Meclofenamate in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Meclofenamate in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Meclofenamate in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
- Intravenous
Monitoring
There is limited information regarding Monitoring of Meclofenamate in the drug label.
- Description
IV Compatibility
There is limited information regarding IV Compatibility of Meclofenamate in the drug label.
Overdosage
- The following is based on the little information available concerning overdosage with meclofenamate sodium and related compounds. After a massive overdose, CNS stimulation may be manifested by irrational behavior, marked agitation and generalized seizures. Following this phase, renal toxicity (falling urine output, rising creatinine, abnormal urinary cellular elements) may be noted with possible oliguria or anuria and azotemia. A 24 year-old male was anuric for approximately one week after ingesting an overdose of 6 to 7 grams of meclofenamate sodium. Spontaneous diuresis and recovery subsequently occurred.
- Management consists of emptying the stomach by emesis or lavage and instilling an ample dose of activated charcoal into the stomach. There is some evidence that charcoal will actively absorb meclofenamate sodium, but dialysis or hemoperfusion may be less effective because of plasma protein binding. The seizures should be controlled by an appropriate anticonvulsant regimen. Attention should be directed throughout, by careful monitoring, to the preservation of vital functions and fluid-electrolyte balance. Dialysis may be required to correct serious azotemia or electrolyte imbalance.
Pharmacology
| |
Meclofenamate
| |
| Systematic (IUPAC) name | |
| 2-[(2,6-dichloro-3-methylphenyl)amino]benzoic acid | |
| Identifiers | |
| CAS number | |
| ATC code | M01 M02AA18 (WHO) |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 296.14864 g/mol |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | Oral |
Mechanism of Action
There is limited information regarding Meclofenamate Mechanism of Action in the drug label.
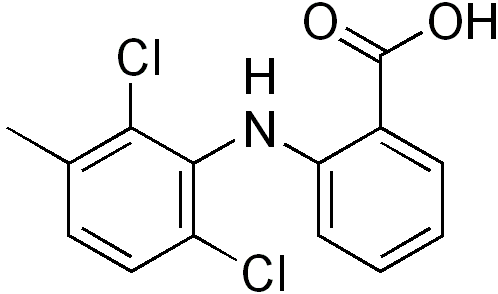
Structure
- Meclofenamate sodium, USP is N-(2,6-dichloro-m-tolyl) anthranilic acid, sodium salt, monohydrate. It is an anti-inflammatory drug for oral administration. Meclofenamate sodium capsules, USP contain 50 mg or 100 mg meclofenamic acid as the sodium salt and the following inactive ingredients: colloidal silicon dioxide, FD&C Blue No. 1, gelatin, magnesium stearate, microcrystalline cellulose, pregelatinized starch, FD&C Red No. 3, sodium lauryl sulfate, titanium dioxide and D&C Yellow No. 10.
- The structural formula of meclofenamate sodium is:

- It is a white to creamy white, odorless to almost odorless, crystalline powder with melting point 287° to 291°C, molecular weight 336.15, and it is freely soluble in water.
Pharmacodynamics
- Meclofenamate sodium is a non-steroidal agent which has demonstrated anti-inflammatory, analgesic, and antipyretic activity in laboratory animals. The mode of action, like that of other non-steroidal anti-inflammatory agents, is not known. Therapeutic action does not result from pituitary-adrenal stimulation. In animal studies, meclofenamate sodium was found to inhibit prostaglandin synthesis and to compete for binding at the prostaglandin receptor site. In vitro, meclofenamate sodium was found to be an inhibitor of human leukocyte 5-lipoxygenase activity. These properties may be responsible for the anti-inflammatory action of meclofenamate sodium. There is no evidence that meclofenamate sodium alters the course of the underlying disease.
- In several human isotope studies, meclofenamate sodium, at a dosage of 300 mg/day, produced a fecal blood loss of 1 to 2 mL per day, and 2 to 3 mL per day at 400 mg/day. Aspirin, at a dosage of 3.6 g/day, caused a fecal blood loss of 6 mL per day.
- In a multiple-dose, 1-week study in normal human volunteers, meclofenamate sodium had little or no effect on collagen-induced platelet aggregation, platelet count, or bleeding time. In comparison, aspirin suppressed collagen-induced platelet aggregation and increased bleeding time. The concomitant administration of antacids (aluminum and magnesium hydroxides) does not interfere with absorption of meclofenamate sodium.
Pharmacokinetics
- Meclofenamate sodium is rapidly absorbed in man following single and multiple oral doses with peak plasma concentrations occurring in 0.5 to 2 hours. Based on a comparison to a suspension of meclofenamic acid, meclofenamate sodium is completely bioavailable.
- The plasma concentrations of meclofenamic acid decline monoexponentially following oral administration. In a study in ten healthy subjects following a single oral dose the apparent elimination half-life ranged from 0.8 to 5.3 hours. After the administration of meclofenamate sodium for 14 days every 8 hours, the apparent elimination half-life ranged from 0.8 to 2.1 hours with no evidence of accumulation of meclofenamic acid in plasma.
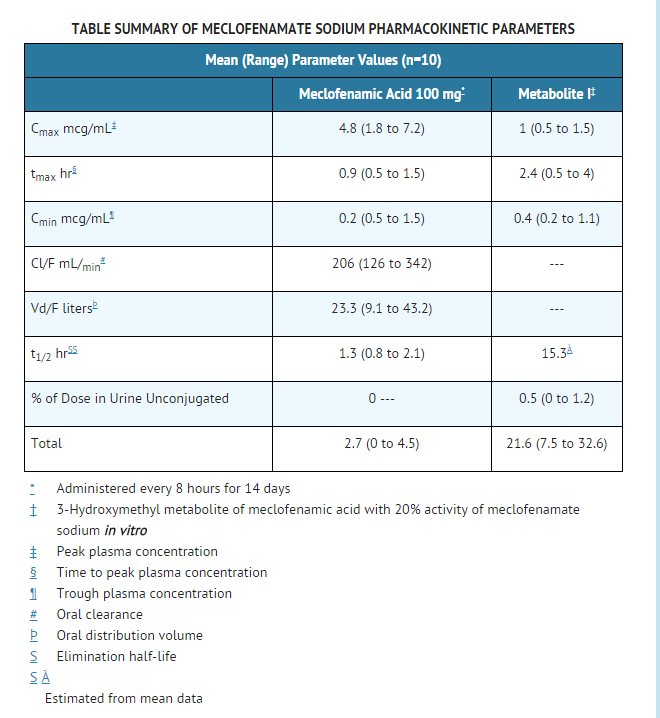
- Meclofenamic acid is extensively metabolized to an active metabolite (Metabolite I; 3-hydroxymethyl metabolite of meclofenamic acid) and at least six other less well characterized minor metabolites. Only this Metabolite I has been shown in vitro to inhibit cyclooxygenase activity with approximately one fifth the activity of meclofenamate sodium. Metabolite I (3-hydroxymethyl metabolite of meclofenamic acid) with a mean half-life of approximately 15 hours did accumulate following multiple dosing. After the administration of 100 mg meclofenamate sodium for 14 days every 8 hours, Metabolite I reached a peak plasma concentration of only 1 mcg/mL. By contrast, the peak concentration was 4.8 mcg/mL for the parent compound on both days 1 and 14. Therefore, the accumulation of Metabolite I is probably not clinically significant.
- Approximately 70% of the administered dose is excreted by the kidneys with 8% to 35% excreted as predominantly conjugated species of meclofenamic acid and Metabolite I (see TABLE). Other metabolites, whose excretion rates are unknown, account for the remaining 35% to 62% of the dose excreted in the urine. The remainder of the administered dose (approximately 30%) is eliminated in the feces (apparently through biliary excretion). There is insufficient experience to know if meclofenamate sodium or its metabolites accumulate in patients with compromised renal or hepatic function. Therefore, meclofenamate sodium should be used with caution in these patients. Trace amounts of meclofenamate sodium are excreted in human breast milk.
- Meclofenamic acid is greater than 99% bound to plasma proteins over a wide drug concentration range.
- Unlike most NSAIDs, which when administered with food have a decrease in rate but not in extent of absorption, meclofenamic acid is decreased in both. It has been reported that following the administration of meclofenamate sodium capsules one-half hour after a meal, the average extent of bioavailability decreased by 26%, the average peak concentration (Cmax) decreased 4-fold and the time to Cmax was delayed by 3 hours.
Nonclinical Toxicology
- An 18-month study in rats revealed no evidence of carcinogenicity.
Clinical Studies
- Controlled clinical trials comparing meclofenamate sodium with aspirin demonstrated comparable efficacy in rheumatoid arthritis.
- The meclofenamate sodium treated patients had fewer reactions involving the special senses, specifically tinnitus, but more gastrointestinal reactions, specifically diarrhea.
- The incidence of patients who discontinued therapy due to adverse reactions was similar for both the meclofenamate sodium and aspirin-treated groups.
- The improvement with meclofenamate sodium reported by patients and the reduction of the disease activity as evaluated by both physicians and patients with rheumatoid arthritis are associated with a significant reduction in number of tender joints, severity of tenderness, and duration of morning stiffness.
- The improvement reported by patients and as evaluated by physicians in patients treated with meclofenamate sodium for osteoarthritis is associated with a significant reduction in night pain, pain on walking, degree of starting pain, and pain on passive motion. The function of knee joints also improved significantly.
- Meclofenamate sodium has been used in combination with gold salts or corticosteroids in patients with rheumatoid arthritis. Studies have demonstrated that meclofenamate sodium contributes to the improvement of patients' conditions while maintained on gold salts or corticosteroids. Data are inadequate to demonstrate that meclofenamate sodium in combination with salicylates produces greater improvement than that achieved with meclofenamate sodium alone.
- In controlled clinical trials of patients with mild to moderate pain, meclofenamate sodium 50 mg provided significant pain relief. In these studies of episiotomy and dental pain, meclofenamate sodium 100 mg demonstrated additional benefit in some patients. The onset of analgesic effect was generally within one hour and the duration of action was 4 to 6 hours.
- In controlled clinical trials of patients with dysmenorrhea, meclofenamate sodium 100 mg t.i.d. provided significant reduction in the symptoms associated with dysmenorrhea.
- In randomized double-blind crossover trials of meclofenamate sodium 100 mg t.i.d. versus placebo in women with heavy menstrual blood loss (MBL), meclofenamate sodium treatment was usually associated with a reduction in menstrual flow.
- The graph below is a scatter plot of menstrual flow from the average of two menstrual periods on meclofenamate sodium treatments (vertical axis) versus two menstrual periods on placebo (horizontal axis) for 55 women. Of note, although the amount of reduction in MBL was variable, some degree of reduction occurred in 90% of women in this study.

- The points on the graph represent the mean MBL for each subject when treated for two periods with placebo and two periods with meclofenamate sodium. To ease in interpretation, the following examples may be helpful. Point A represents a woman who had MBL of 459 mL while on placebo, and 405 mL on meclofenamate sodium. Point B represents a woman who had MBL of 472 mL while on placebo, and 64 mL when treated with meclofenamate sodium.
- In association with this reduction in menstrual blood loss, the duration of menses was decreased by one day; tampon/pad usage was decreased by an average of two per day on the 2 days of heaviest flow; and symptoms of dysmenorrhea were significantly reduced.
How Supplied
- The 50 mg capsule is a hard-shell gelatin capsule with a coral opaque cap and a coral opaque body axially printed with MYLAN over 2150 in black ink on both the cap and body. The capsule is filled with an off-white powder blend. They are available as follows:
- NDC 0378-2150-01
- bottles of 100 capsules
- The 100 mg capsule is a hard-shell gelatin capsule with a coral opaque cap and a white opaque body axially printed with MYLAN over 3000 in black ink on both the cap and body. The capsule is filled with an off-white powder blend. They are available as follows:
- NDC 0378-3000-01
- bottles of 100 capsules
- NDC 0378-3000-05
- bottles of 500 capsules
Storage
- Store at 20° to 25°C (68° to 77°F).
Images
Drug Images
{{#ask: Page Name::Meclofenamate |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel




{{#ask: Label Page::Meclofenamate |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Information for Patients
- Patients should be informed of the following information before initiating therapy with an NSAID and periodically during the course of ongoing therapy. Patients should also be encouraged to read the NSAID Medication Guide that accompanies each prescription dispensed.
- Meclofenamate sodium, like other NSAIDs, may cause serious CV side effects, such as MI or stroke, which may result in hospitalization and even death. Although serious CV events can occur without warning symptoms, patients should be alert for the signs and symptoms of chest pain, shortness of breath, weakness, slurring of speech, and should ask for medical advice when observing any indicative sign or symptoms. Patients should be apprised of the importance of this follow-up.
- Meclofenamate sodium, like other NSAIDs, can cause GI discomfort and, rarely, serious GI side effects, such as ulcers and bleeding, which may result in hospitalization and even death. Although serious GI tract ulcerations and bleeding can occur without warning symptoms, patients should be alert for the signs and symptoms of ulcerations and bleeding, and should ask for medical advice when observing any indicative sign or symptoms including epigastric pain, dyspepsia, melena, and hematemesis. Patients should be apprised of the importance of this follow-up: Risk of Ulceration, Bleeding, and Perforation).
- Meclofenamate sodium, like other NSAIDs, can cause serious skin side effects such as exfoliative dermatitis, SJS, and TEN, which may result in hospitalizations and even death. Although serious skin reactions may occur without warning, patients should be alert for the signs and symptoms of skin rash and blisters, fever, or other signs of hypersensitivity such as itching, and should ask for medical advice when observing any indicative signs or symptoms. Patients should be advised to stop the drug immediately if they develop any type of rash and contact their physicians as soon as possible.
- Patients should promptly report signs or symptoms of unexplained weight gain or edema to their physicians.
- Patients should be informed of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). If these occur, patients should be instructed to stop therapy and seek immediate medical therapy.
- Patients should be informed of the signs of an anaphylactoid reaction (e.g., difficulty breathing, swelling of the face or throat). If these occur, patients should be instructed to seek immediate emergency help.
- In late pregnancy, as with other NSAIDs, meclofenamate sodium should be avoided because it will cause premature closure of the ductus arteriosus.
Precautions with Alcohol
- Alcohol-Meclofenamate interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- MECLOFENAMATE SODIUM®[1]
Look-Alike Drug Names
- A® — B®[2]
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "meclofenamate sodium capsule".
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Page Name=Meclofenamate
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Meclofenamate |Label Name=Meclofenamate11.png
}}
{{#subobject:
|Label Page=Meclofenamate |Label Name=Meclofenamate11.png
}}