Measles epidemiology and demographics
|
Measles Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Measles epidemiology and demographics On the Web |
|
American Roentgen Ray Society Images of Measles epidemiology and demographics |
|
Risk calculators and risk factors for Measles epidemiology and demographics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Joseph Nasr, M.D.[2] ;Guillermo Rodriguez Nava, M.D. [3];
Global Overview
- Leading vaccine-preventable killer of children. Measles remains a leading cause of vaccine-preventable childhood mortality worldwide.
- Steep global resurgence since 2019. After a historic low of 132,490 reported cases in 2016, global measles reports surged to 869,770 in 2019, driven by large outbreaks in the DRC, Madagascar, Samoa, Ukraine, and Brazil, with vaccine hesitancy cited as a major driver; in 2019 the WHO listed vaccine hesitancy among the top 10 global health challenges[1][2][3].
- Pandemic shock to coverage. Routine immunization disruptions during Covid-19 pushed MCV1 coverage to 81% (lowest since 2008), with only partial recovery to 83% in 2022–2023; coverage is lowest in low-income countries (64%) and 86% in middle-income countries[4].
- Current case burden (2024–2025). All WHO regions have reported increases since 2024; 395,521 laboratory-confirmed cases were reported in 2024 and 16,147 in the first 2 months of 2025; >50% of reported patients were hospitalized, implying substantial under-ascertainment[5][6].
- European region, 2024. The WHO European Region recorded its highest measles case count in >25 years in 2024, accounting for ~20% of global cases[5].
Historical mortality reductions (Measles Initiative) according to the World Health Organization (WHO). Global measles deaths fell ~60% (from ~873,000 in 1999 to ~345,000 in 2005). In Africa, deaths declined ~75% (from ~506,000 to ~126,000 in 5 years).
Transmission geography, elimination, and importations
- Importations sustain outbreaks in elimination settings. Countries with interrupted endemic transmission (e.g., United States, elimination declared 2000) still see outbreaks seeded by imported cases, with spread in undervaccinated pockets[7][2].
- Global elimination setbacks. Since 2019, no WHO region has achieved and sustained measles elimination[7].
Coverage gaps, susceptibility, and demographics
- Sub-national heterogeneity (U.S. example). County-level analyses in 37 U.S. states showed MMR coverage <95% in 990/1501 counties and <74% in 70 counties, identifying large susceptible clusters[8].
- Infants. Maternal antibody waning earlier than in past decades leaves most infants seronegative by 6 months, amplifying risk during periods of community transmission[9][10][11][12].
- Adolescents/young adults. Immunity gaps are documented among persons ~13–30 years (and some >40 years) who received the standard two-dose schedule in childhood; these gaps account for some adult cases during outbreaks in well-vaccinated countries[10][13][14].
Burden and clinical impact (selected metrics relevant to epi)
- Hospitalization share as severity proxy. In 2024–2025, >50% of reported cases required hospitalization, which the NEJM authors note implies undercounting of true infections[5].
- Complications and fatality gradients (context for demographic risk and setting): case-fatality 1–3/1000 in high-income settings vs 9–16/1000 in middle-/low-income settings; up to 180/1000 in humanitarian crises[15][16][17].
Region-specific notes
Europe
- 2019 surge and 2024 peak. >100,000 European cases in 2019 amid global resurgence; in 2024, the region reached the highest burden in >25 years (~20% of global cases)[1][5].
United States
Pre-elimination Era
- In the decade prior to the licensure of live measles vaccine in 1963, an average of 549,000 measles cases and 495 measles deaths were reported annually.[18]
- Almost every American was affected by measles during their lifetime; it is estimated that 3–4 million measles cases occurred each year.
- Following implementation of the one dose measles vaccine program, there was significant reduction in the reported incidence in the United States by 1988 resulting in decline in measles-related hospitalizations and death.
- During 1989–1991, a resurgence of measles occurred when over 55,000 cases and 123 deaths were reported.
- The epidemiology during the resurgence was characterized mainly by cases in unvaccinated preschool-age children who had not been vaccinated on time with one dose of measles vaccine.
- Outbreaks were reported among highly vaccinated school-age children who received one dose of measles-containing vaccine.
- In 1989, a second-dose vaccination schedule was recommended by the Advisory Committee on Immunization Practices (ACIP), the American Academy of Pediatrics (AAP), and the American Academy of Family Physicians (AAFP).
- In 1998, the ACIP recommended that states ensure second dose coverage of children in all grades by 2001.
- Following the resurgence, improved implementation of the timely administration of the first dose of MMR vaccine and increased implementation of two doses among school-age children led to a dramatic decline in measles cases.
- In 2000, endemic measles was declared “eliminated” from the United States.
- Elimination and ongoing importations. The U.S. declared elimination in 2000; present-day cases reflect importations and spread within undervaccinated communities[7][2].
Post-elimination Era
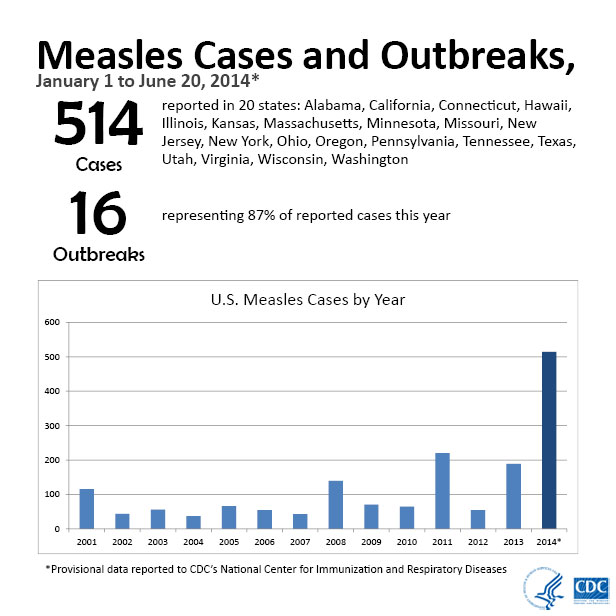
- In 2017, a measles outbreak began in Minnesota among the Somali American community. The number of cases of measles in Minnesota by May 2017 exceeded the annual rate in the entire United States in 2016.
- During 2001–2011, 911 measles cases were reported. [18]
- The median number of measles cases reported per year was 62 (range: 37–220 cases/year).
- Measles incidence has continuously remained below one case per million since 1997. The majority of measles cases were unvaccinated (65%) or had unknown vaccination status (20%).
- Of the 911 reported measles cases, 372 (40%) were importations (on average 34 importations/year), 239 (26%) were epidemiologically linked to these importations, 190 (21%) either had virologic evidence of importation or had been linked to those cases with virologic evidence of importation, and 110 (12%) had unknown source (unknown source cases represent cases where epidemiologic- or virologic-link to an imported case was not detected0
- The highest incidence of measles cases in recent years occurred in 2008 (0.48 cases/million) and 2011 (0.72 cases/million):
- The epidemiology of measles in 2008 was characterized by (1) a high proportion (95%) of cases among U.S. residents who were unvaccinated or who had unknown vaccination status, most of whom were U.S. school-age children whose parents had religious or philosophical objections to vaccination, and (2) more spread from imported cases than other years.
- In 2011, 220 measles cases were reported, the highest number of reported measles cases since 1996; 80 (36%) were importations, 144 (65%) were unvaccinated, and 47 (21%) had unknown vaccination status. Most of the importations were the result of unvaccinated U.S. travelers who had traveled to measles endemic countries, mainly Western Europe and India.
- Although measles elimination has been achieved in the United States, importation of measles will continue to occur as measles remains endemic in many other parts of the world.
- 2025 situation (as of May 30). 1,088 confirmed cases and 3 deaths; ~96% unvaccinated/unknown status; ~12% hospitalized; current counts are ~4× the 2024 total; loss of U.S. elimination status is possible if transmission persists >12 months[20].
- Domestic determinants. Misinformation (e.g., now-disproven autism claims) and vaccine hesitancy contribute to under-vaccination; modeling suggests that a 10% drop in MMR could yield ~11.1 million U.S. measles cases over 25 years[21][22].
Developed countries
- Routine immunization commonly at ~12–18 months as part of MMR; importation-linked outbreaks still occur when pockets of susceptibility exist; Japan experienced a notable 2007 surge; broad MMR eradication proposals have circulated but are not prioritized before global polio eradication.
South America
- Regional initiatives to eliminate rubella by 2010; as of 2006, endemic measles still reported in Bolivia, Brazil, Colombia, Guatemala, Mexico, Peru, Venezuela; immunization campaigns ongoing (e.g., Dominican Republic)[23].
- The Americas were formally declared free of endemic rubella and congenital rubella syndrome (CRS) in April 2015, and PAHO notes the region has maintained rubella/CRS elimination in subsequent years (despite measles flare-ups)[24].
Developing countries
- Programmatic setbacks (e.g., early-2000s northern Nigeria) with vaccine refusals led to large outbreaks and child deaths[25].
- In developing countries, measles remains common. Unvaccinated populations are at risk for the disease.
- After vaccination rates dropped in northern Nigeria in the early 2000s due to religious and political objections, the number of cases rose significantly, and hundreds of children died.[25]
- A 2005 measles outbreak in Indiana was attributed to children whose parents refused vaccination.[26]
- 2016→2019 global surge (major LMIC impact). Reported measles cases climbed to 869,770 in 2019 (highest since 1996), with deaths up ~50% from 2016 to ~207,500 in 2019; large outbreaks were concentrated in countries with chronic under-immunization and health-system gaps (DRC, Madagascar, Samoa, Ukraine, others)[4][27].
Flagship LMIC outbreaks:
- DRC, 2019–2020: “World’s worst measles epidemic” with >6,000 deaths by Jan 2020; drivers included low coverage, malnutrition, weak systems, other epidemics, access insecurity[28].
- Madagascar, 2018–2019: explosive nationwide epidemic; >100,000 cases over first 6 months[29][30].
- Pacific (Samoa et al.), 2019: severe outbreaks with emergency declarations; extremely low MMR coverage preceded the surge[31].
- WHO Europe spillover affecting lower-income settings (2019): Regional spike, majority of cases in Ukraine early 2019 (vaccine gaps)[32].
- Pandemic shock and recovery lag in LMICs. First-dose measles coverage (MCV1) fell to ~81% (2021) and only partly recovered to ~83% (2022), below 2019’s 86%; WHO estimates ~35.2 million additional children at risk from service disruptions[4][33].
- Ongoing high burden post-COVID. CDC estimates ~10.3 million infections in 2023 and outbreaks in every region; LMICs bear disproportionate morbidity/mortality[34].
- Surveillance & lab capacity improvements relevant to LMICs.
- Field confirmation: Rapid diagnostic tests (RDTs) for measles IgM (capillary blood/oral fluid) facilitate real-time confirmation where EIA/RT-PCR capacity is limited. Genes can be amplified from dried IgM-positive RDTs to support genotyping. (Appendix section “Measles diagnostic and genotypes monitoring”)[35][36].
- Genomic surveillance: 24 genotypes (N-450 window) remain the basis for global tracking; since 2018, B3, D4, D8, H1 are identified, with B3/D8/H1 dominating 2024–2025 outbreaks; Nanopore enables low-cost near-full genomes for chain tracking in resource-limited labs[37][38].
- Antigenic stability & vaccine breadth: Despite genotype diversity, H and F have remained antigenically stable for decades; H-specific antibodies dominate protection; vaccine lineage (genotype A) continues to protect across circulating genotypes[39][40][41][42].
- Programmatic shifts & evidence relevant to LMIC policy.
- Early-age vaccination discussions: WHO technical consultations and multiple studies examine <9-month dosing (maternal antibody interference vs. outbreak control), waning dynamics, and boost strategies — critically relevant for LMIC settings with high infant exposure risk[43][44][45].
- Vitamin A in severe measles (still pertinent in LMICs): WHO clinical guidance and evidence syntheses reaffirm adjunct vitamin A for complicated measles (malnutrition, eye complications), reflecting persistent LMIC risk profiles[46][47][48].
- Financing headwinds (program risk). Recent reporting raised concerns about potential U.S. cuts to Gavi; Gavi stated no official termination notice had been received, but any reduction would jeopardize LMIC immunization[49].
References
- ↑ 1.0 1.1 Patel, M.K. et al. (2020) “Progress toward regional measles elimination - worldwide, 2000-2019,” MMWR. Morbidity and mortality weekly report, 69(45), pp. 1700–1705. Available at: https://doi.org/10.15585/mmwr.mm6945a6.
- ↑ 2.0 2.1 2.2 Hotez, P.J., Nuzhath, T. and Colwell, B. (2020) “Combating vaccine hesitancy and other 21st century social determinants in the global fight against measles,” Current opinion in virology, 41, pp. 1–7. Available at: https://doi.org/10.1016/j.coviro.2020.01.001.
- ↑ Larson, H.J., Gakidou, E. and Murray, C.J.L. (2022) “The vaccine-hesitant moment,” The New England journal of medicine, 387(1), pp. 58–65. Available at: https://doi.org/10.1056/NEJMra2106441.
- ↑ 4.0 4.1 4.2 Minta, A.A. et al. (2024) “Progress toward measles elimination - worldwide, 2000-2023,” MMWR. Morbidity and mortality weekly report, 73(45), pp. 1036–1042. Available at: https://doi.org/10.15585/mmwr.mm7345a4.
- ↑ 5.0 5.1 5.2 5.3 WHO Immunization Data portal - All Data (no date) Immunization Data. Available at: https://immunizationdata.who.int/global (Accessed: September 24, 2025).
- ↑ “In Japan, there are concerns that measles infections will spread due to the Osaka/Kansai World Expo in April 2025” (2025). Available at: https://www.bmj.com/content/388/bmj.r528/rr (Accessed: September 24, 2025).
- ↑ 7.0 7.1 7.2 Do, L.A.H. and Mulholland, K. (2025) “Measles 2025,” The New England journal of medicine [Preprint], (NEJMra2504516). Available at: https://doi.org/10.1056/NEJMra2504516.
- ↑ Rader, B. et al. (2025) “Revising US MMR vaccine recommendations amid changing domestic risks,” JAMA: the journal of the American Medical Association, 333(14), pp. 1201–1202. Available at: https://doi.org/10.1001/jama.2025.3867.
- ↑ Guerra, F.M. et al. (2018) “Waning of measles maternal antibody in infants in measles elimination settings - A systematic literature review,” Vaccine, 36(10), pp. 1248–1255. Available at: https://doi.org/10.1016/j.vaccine.2018.01.002.
- ↑ 10.0 10.1 Schenk, J. et al. (2021) “Immunogenicity and persistence of trivalent measles, mumps, and rubella vaccines: a systematic review and meta-analysis,” The Lancet infectious diseases, 21(2), pp. 286–295. Available at: https://doi.org/10.1016/S1473-3099(20)30442-4.
- ↑ Science M, Savage R, Severini A, et al. Measles antibody levels in young infants. Pediatrics 2019;144(6):e20190630.
- ↑ Bokop, C. et al. (2025) “Sero-epidemiology of measles immunoglobulin G antibodies among newborns from South-East Asia and sub-Saharan Africa: an observational, multicenter study,” International journal of infectious diseases : IJID : official publication of the International Society for Infectious Diseases, 154. Available at: https://doi.org/10.1016/j.ijid.2025.107882.
- ↑ Robert A, Suffel AM, Kucharski AJ. Long-term waning of vaccine-induced im�munity to measles in England: a mathe�matical modelling study. Lancet Public Health 2024;9(10):e766-e775.
- ↑ Mehra, S. et al. (2025) “Unveiling immunity gaps and determining a suitable age for a third dose of the measles-containing vaccine: a strategic approach to accelerating measles elimination,” The Lancet regional health. Southeast Asia, 32(100523), p. 100523. Available at: https://doi.org/10.1016/j.lansea.2024.100523.
- ↑ CDC (2025) Measles Symptoms and Complications, Measles (Rubeola). Available at: https://www.cdc.gov/measles/signs-symptoms/index.html (Accessed: September 24, 2025).
- ↑ Sbarra, A.N. et al. (2023) “Estimating national-level measles case-fatality ratios in low-income and middle-income countries: an updated systematic review and modelling study,” The Lancet. Global health, 11(4), pp. e516–e524. Available at: https://doi.org/10.1016/S2214-109X(23)00043-8.
- ↑ Salama, P. et al. (2001) “Malnutrition, measles, mortality, and the humanitarian response during a famine in Ehiopia,” JAMA: the journal of the American Medical Association, 286(5), pp. 563–571. Available at: https://doi.org/10.1001/jama.286.5.563.
- ↑ 18.0 18.1 "Measles" (PDF).
- ↑ "Measles Cases and Outbreaks".
- ↑ CDC (2025) Measles Cases and Outbreaks, Measles (Rubeola). Available at: https://www.cdc.gov/measles/data-research/index.html (Accessed: September 24, 2025).
- ↑ DeStefano, F. and Shimabukuro, T.T. (2019) “The MMR vaccine and autism,” Annual review of virology, 6(1), pp. 585–600. Available at: https://doi.org/10.1146/annurev-virology-092818-015515.
- ↑ Kiang, M.V. et al. (2025) “Modeling reemergence of vaccine-eliminated infectious diseases under declining vaccination in the US,” JAMA: the journal of the American Medical Association, 333(24), pp. 2176–2187. Available at: https://doi.org/10.1001/jama.2025.6495.
- ↑ "www.paho.org". Retrieved 2013-02-25.
- ↑ Americas region is declared the world’s first to eliminate rubella (no date) Paho.org. Available at: https://www.paho.org/en/news/29-4-2015-americas-region-declared-worlds-first-eliminate-rubella (Accessed: September 24, 2025).
- ↑ 25.0 25.1 Youth and migration (2007) The New Humanitarian. Available at: https://www.thenewhumanitarian.org/feature/2007/02/06/youth-and-migration (Accessed: September 24, 2025).
- ↑ Parker A, Staggs W, Dayan G; et al. (2006). "Implications of a 2005 measles outbreak in Indiana for sustained elimination of measles in the United States". N Engl J Med. 355 (5): 447–55. PMID 16885548.
- ↑ Worldwide measles deaths climb 50% from 2016 to 2019 claiming over 207 500 lives in 2019 (no date) Who.int. Available at: https://www.who.int/news/item/12-11-2020-worldwide-measles-deaths-climb-50-from-2016-to-2019-claiming-over-207-500-lives-in-2019?utm_source=chatgpt.com (Accessed: September 24, 2025).
- ↑ Deaths from Democratic Republic of the Congo measles outbreak top 6000 (no date) WHO | Regional Office for Africa. Available at: https://www.afro.who.int/news/deaths-democratic-republic-congo-measles-outbreak-top-6000 (Accessed: September 24, 2025).
- ↑ Measles – Madagascar (no date) Who.int. Available at: https://www.who.int/emergencies/disease-outbreak-news/item/17-january-2019-measles-madagascar-en?utm_source=chatgpt.com (Accessed: September 24, 2025).
- ↑ Nimpa, M.M. et al. (2020) “Measles outbreak in 2018-2019, Madagascar: epidemiology and public health implications,” The Pan African medical journal, 35, p. 84. Available at: https://doi.org/10.11604/pamj.2020.35.84.19630.
- ↑ (No date) Who.int. Available at: https://www.who.int/docs/default-source/wpro---documents/dps/outbreaks-and-emergencies/measles-2019/measles-pacific-who-unicef-sitrep-20200103.pdf?utm_source=chatgpt.com (Accessed: September 24, 2025).
- ↑ Measles – European Region (no date) Who.int. Available at: https://www.who.int/emergencies/disease-outbreak-news/item/2019-DON140?utm_source=chatgpt.com (Accessed: September 24, 2025).
- ↑ Childhood immunization begins recovery after COVID-19 backslide (no date) Who.int. Available at: https://www.who.int/news/item/18-07-2023-childhood-immunization-begins-recovery-after-covid-19-backslide?utm_source=chatgpt.com (Accessed: September 24, 2025).
- ↑ CDC (2025) Global Measles Outbreaks, Global Measles Vaccination. Available at: https://www.cdc.gov/global-measles-vaccination/data-research/global-measles-outbreaks/index.html (Accessed: September 24, 2025).
- ↑ Brown, D.W. et al. (2020) “Rapid diagnostic tests to address challenges for global measles surveillance,” Current opinion in virology, 41, pp. 77–84. Available at: https://doi.org/10.1016/j.coviro.2020.05.007.
- ↑ Warrener, L. et al. (2023) “Evaluation of a rapid diagnostic test for measles IgM detection; accuracy and the reliability of visual reading using sera from the measles surveillance programme in Brazil, 2015,” Epidemiology and infection, 151, p. e151. Available at: https://doi.org/10.1017/S0950268823000845.
- ↑ CDC (2024) Genetic Analysis of Measles Viruses, Measles (Rubeola). Available at: https://www.cdc.gov/measles/php/laboratories/genetic-analysis.html (Accessed: September 24, 2025).
- ↑ Namuwulya, P. et al. (2022) “Near-complete genome sequences of measles virus strains from 10 years of Uganda country-wide surveillance,” Microbiology resource announcements, 11(8), p. e0060622. Available at: https://doi.org/10.1128/mra.00606-22.
- ↑ Beaty, S.M. and Lee, B. (2016) “Constraints on the genetic and antigenic variability of measles virus,” Viruses, 8(4), p. 109. Available at: https://doi.org/10.3390/v8040109.
- ↑ Muñoz-Alía, M.Á. et al. (2021) “Serotypic evolution of measles virus is constrained by multiple co-dominant B cell epitopes on its surface glycoproteins,” Cell reports. Medicine, 2(4), p. 100225. Available at: https://doi.org/10.1016/j.xcrm.2021.100225.
- ↑ Tahara, M. et al. (2013) “The receptor-binding site of the measles virus hemagglutinin protein itself constitutes a conserved neutralizing epitope,” Journal of virology, 87(6), pp. 3583–3586. Available at: https://doi.org/10.1128/JVI.03029-12.
- ↑ Muñoz-Alía, M.Á., Muller, C.P. and Russell, S.J. (2017) “Antigenic drift defines a new D4 subgenotype of measles virus,” Journal of virology, 91(11). Available at: https://doi.org/10.1128/JVI.00209-17.
- ↑ Varma, A. et al. (2025) “What is the current evidence base for measles vaccination earlier than 9 months of age?: Report from an informal technical consultation of the World Health Organization,” Vaccine, 57(127187), p. 127187. Available at: https://doi.org/10.1016/j.vaccine.2025.127187.
- ↑ Njie-Jobe, J. et al. (2012) “Immunological impact of an additional early measles vaccine in Gambian children: responses to a boost at 3 years,” Vaccine, 30(15), pp. 2543–2550. Available at: https://doi.org/10.1016/j.vaccine.2012.01.083.
- ↑ Garly, M.-L. et al. (2006) “Prophylactic antibiotics to prevent pneumonia and other complications after measles: community based randomised double blind placebo controlled trial in Guinea-Bissau,” BMJ (Clinical research ed.), 333(7581), p. 1245. Available at: https://doi.org/10.1136/bmj.38989.684178.AE.
- ↑ Stinchfield, P.A. and Orenstein, W.A. (2020) “Vitamin A for the management of measles in the United States,” Infectious diseases in clinical practice (Baltimore, Md.), 28(4), pp. 181–187. Available at: https://doi.org/10.1097/ipc.0000000000000873.
- ↑ Immunization, Vaccines and Biologicals (2020) Guide for clinical case management and infection prevention and control during a measles outbreak, Who.int. World Health Organization. Available at: https://www.who.int/publications/i/item/9789240002869 (Accessed: September 24, 2025).
- ↑ Huiming, Y., Chaomin, W. and Meng, M. (2005) “Vitamin A for treating measles in children,” Cochrane database of systematic reviews, 2005(4), p. CD001479. Available at: https://doi.org/10.1002/14651858.CD001479.pub3.
- ↑ Vaccines group Gavi says U.S. has not confirmed it will end funding (2025) AP News. Available at: https://apnews.com/article/gavi-alliance-vaccines-us-funding-3123781dd776b0019d4f869789e2715e (Accessed: September 24, 2025).
index.php?title=Category:Pediatrics index.php?title=Category:Dermatology index.php?title=Category:Viral diseases index.php?title=Category:Mononegavirales index.php?title=Category:Ophthalmology index.php?title=Category:Otolaryngology index.php?title=Category:Pulmonology index.php?title=Category:Disease