Leuprolide
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Vignesh Ponnusamy, M.B.B.S. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Leuprolide is a gonadatropin releasing hormone (GnRH) agonist that is FDA approved for the treatment of advanced prostate cancer. Common adverse reactions include malaise, fatigue, hot flashes/sweats, and testicular atrophy.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Palliative Treatment Of Advanced Prostate Cancer
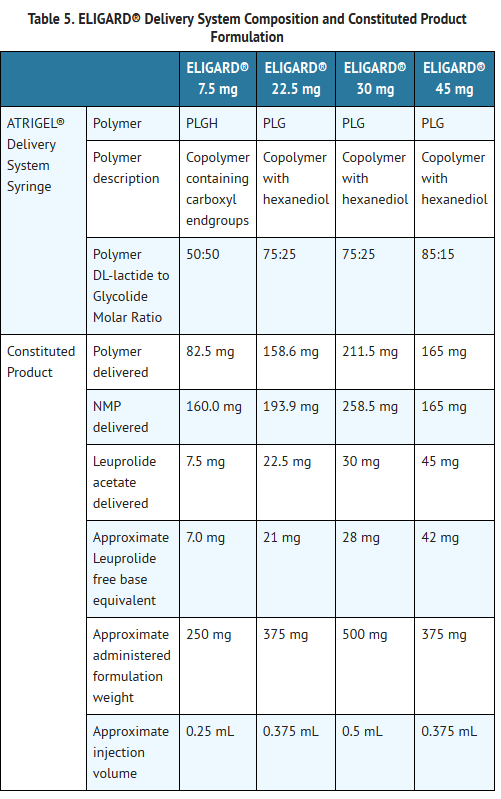
- ELIGARD® is administered subcutaneously and provides continuous release of leuprolide acetate over a one-, three-, four-, or six-month treatment period (TABLE 1). The injection delivers the dose of leuprolide acetate incorporated in a polymer formulation.
- Once mixed, ELIGARD® should be discarded if not administered within 30 minutes.
- As with other drugs administered by subcutaneous injection, the injection site should vary periodically. The specific injection location chosen should be an area with sufficient soft or loose subcutaneous tissue. In clinical trials, the injection was administered in the upper- or mid-abdominal area. Avoid areas with brawny or fibrous subcutaneous tissue or locations that could be rubbed or compressed (i.e., with a belt or clothing waistband).
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Leuprolide in adult patients.
Non–Guideline-Supported Use
Breast cancer
- Leuprolide 3.75 mg 1-month depot formulation.[1]
In vitro fertilization
- Leuprolide 1 mg (0.25 to 0.5 mg for those with lower body mass) subcutaneously either on day 1 of the menstrual cycle and continued each morning until the day of human chorionic gonadotropin (hCG) administration (flare protocol), or on day 21 of the prior cycle (down-regulation protocol).[2]
Ovarian cancer
- Leuprolide acetate, injected intramuscularly monthly at a dose of 7.5 mg.[3]
Premenstrual syndrome
- 3 monthly IM injections of either leuprolide depot 3.75 mg.[4]
Prostate cancer
- Leuprolide IM (7.5 mg/month) or 22.5 mg every 3 months.[5]
Prostate cancer, Neoadjuvant treatment
- Monthly IM injections of leuprolide 7.5 mg.[6]
Uterine leiomyoma
- Leuprolide acetate 0.5 mg daily for 24 weeks.[7]
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Leuprolide in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Leuprolide in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Leuprolide in pediatric patients.
Contraindications
- Hypersensitivity
- ELIGARD® is contraindicated in patients with hypersensitivity to GnRH, GnRH agonist analogs or any of the components of ELIGARD®. Anaphylactic reactions to synthetic GnRH or GnRH agonist analogs have been reported in the literature.
- Pregnancy
- ELIGARD may cause fetal harm when administered to a pregnant woman. Expected hormonal changes that occur with ELIGARD treatment increase the risk for pregnancy loss and fetal harm when administered to a pregnant women. ELIGARD is contraindicated in women who are or may become pregnant. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
Warnings
Precautions
- Tumor Flare
- ELIGARD® 7.5 mg 22.5 mg 30 mg, like other GnRH agonists, causes a transient increase in serum concentrations of testosterone during the first week of treatment. ELIGARD® 45 mg causes a transient increase in serum concentrations of testosterone during the first two weeks of treatment. Patients may experience worsening of symptoms or onset of new signs and symptoms during the first few weeks of treatment, including bone pain, neuropathy, hematuria, or bladder outlet obstruction.
- Cases of ureteral obstruction and/or spinal cord compression, which may contribute to paralysis with or without fatal complications, have been observed in the palliative treatment of advanced prostate cancer using GnRH agonists.
- Patients with metastatic vertebral lesions and/or with urinary tract obstruction should be closely observed during the first few weeks of therapy. If spinal cord compression or ureteral obstruction develops, standard treatment of these complications should be instituted.
- Laboratory Tests
- Response to ELIGARD® should be monitored by measuring serum concentrations of testosterone and prostate specific antigen periodically.
- In the majority of patients, testosterone levels increased above Baseline during the first week, declining thereafter to Baseline levels or below by the end of the second or third week. Castrate levels were generally reached within two to four weeks.
- Castrate testosterone levels were maintained for the duration of the treatment with ELIGARD® 7.5 mg. No increases to above the castrate level occurred in any of the patients.
- Castrate levels were generally maintained for the duration of treatment with ELIGARD® 22.5 mg.
- Once castrate levels were achieved with ELIGARD® 30 mg, most (86/89) patients remained suppressed throughout the study.
- Once castrate levels were achieved with ELIGARD® 45 mg, only one patient (< 1%) experienced a breakthrough, with testosterone levels > 50 ng/dL.
- Results of testosterone determinations are dependent on assay methodology. It is advisable to be aware of the type and precision of the assay methodology to make appropriate clinical and therapeutic decisions.
- Drug/Laboratory Test Interactions: Therapy with leuprolide acetate results in suppression of the pituitary-gonadal system. Results of diagnostic tests of pituitary gonadotropic and gonadal functions conducted during and after leuprolide therapy may be affected.
- Hyperglycemia and Diabetes
- Hyperglycemia and an increased risk of developing diabetes have been reported in men receiving GnRH agonists. Hyperglycemia may represent development of diabetes mellitus or worsening of glycemic control in patients with diabetes. Monitor blood glucose and/or glycosylated hemoglobin (HbA1c) periodically in patients receiving a GnRH agonist and manage with current practice for treatment of hyperglycemia or diabetes.
- Cardiovascular Diseases
- Increased risk of developing myocardial infarction, sudden cardiac death and stroke has been reported in association with use of GnRH agonists in men. The risk appears low based on the reported odds ratios, and should be evaluated carefully along with cardiovascular risk factors when determining a treatment for patients with prostate cancer. Patients receiving a GnRH agonist should be monitored for symptoms and signs suggestive of development of cardiovascular disease and be managed according to current clinical practice.
Adverse Reactions
Clinical Trials Experience
- The safety of all ELIGARD® formulations was evaluated in clinical trials involving patients with advanced prostate cancer. In addition, the safety of ELIGARD® 7.5 mg was evaluated in 8 surgically castrated males (TABLE 4). ELIGARD®, like other GnRH analogs, caused a transient increase in serum testosterone concentrations during the first one to two weeks of treatment. Therefore, potential exacerbations of signs and symptoms of the disease during the first weeks of treatment are of concern in patients with vertebral metastases and/or urinary obstruction or hematuria. If these conditions are aggravated, it may lead to neurological problems such as weakness and/or paresthesia of the lower limbs or worsening of urinary symptoms.
- During the clinical trials, injection sites were closely monitored. Refer to TABLE 3 for a summary of reported injection site events.
- These localized adverse events were non-recurrent over time. No patient discontinued therapy due to an injection site adverse event.
- The following possibly or probably related systemic adverse events occurred during clinical trials with ELIGARD®, and were reported in > 2% of patients (TABLE 7). Often, causality is difficult to assess in patients with metastatic prostate cancer. Reactions considered not drug-related are excluded.
- In addition, the following possibly or probably related systemic adverse events were reported by < 2% of the patients treated with ELIGARD® in these clinical studies.
General
Sweating, insomnia, syncope, rigors, weakness, lethargy
Gastrointestinal
Flatulence, constipation, dyspepsia
Hematologic
Decreased red blood cell count, hematocrit and hemoglobin
Metabolic
Musculoskeletal
Tremor, backache, joint pain, muscle atrophy, limb pain
Nervous
Disturbance of smell and taste, depression, vertigo
Psychiatric
Insomnia, depression, loss of libido*
Renal/Urinary
Difficulties with urination, pain on urination, scanty urination, bladder spasm, blood in urine, urinary retention, urinary urgency, incontinence, nocturia, nocturia aggravated
Reproductive/ Urogenital
Testicular soreness/pain, impotence*, decreased libido*, gynecomastia*, breast soreness/tenderness*, testicular atrophy*, erectile dysfunction, penile disorder*, reduced penis size
Skin
Alopecia, clamminess, night sweats*, sweating increased*
Vascular
-*Expected pharmacological consequences of testosterone suppression.
- Changes in Bone Density
- Decreased bone density has been reported in the medical literature in men who have had orchiectomy or who have been treated with a GnRH agonist analog. It can be anticipated that long periods of medical castration in men will have effects on bone density.
Postmarketing Experience
- During post-marketing surveillance, rare cases of pituitary apoplexy (a clinical syndrome secondary to infarction of the pituitary gland) have been reported after the administration of gonadotropin-releasing hormone agonists. In a majority of these cases, a pituitary adenoma was diagnosed with a majority of pituitary apoplexy cases occurring within 2 weeks of the first dose, and some within the first hour. In these cases, pituitary apoplexy has presented as sudden headache, vomiting, visual changes, ophthalmoplegia, altered mental status, and sometimes cardiovascular collapse. Immediate medical attention has been required.
- Convulsions have also been reported in the postmarketing setting.
Drug Interactions
- No pharmacokinetic drug-drug interaction studies were conducted with ELIGARD®.
Use in Specific Populations
Pregnancy
- Pregnancy Category X
- ELIGARD® is contraindicated in women who are or may become pregnant while receiving the drug. Expected hormonal changes that occur with ELIGARD® treatment increase the risk for pregnancy loss. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus and the potential risk for pregnancy loss.
- In non-clinical studies in rats, major fetal abnormalities were observed after administration of leuprolide acetate throughout gestation. There were increased fetal mortality and decreased fetal weights in rats and rabbits. The effects of fetal mortality are expected consequences of the alterations in hormonal levels brought about by this drug. The possibility exists that spontaneous abortion may occur.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Leuprolide in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Leuprolide during labor and delivery.
Nursing Mothers
- ELIGARD® is not indicated for use in women [see INDICATIONS AND USAGE (1)]. It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from Eligard, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
- The safety and effectiveness of ELIGARD® in pediatric patients have not been established.
Geriatic Use
- The majority of the patients (approximately 70%) studied in the clinical trials were age 70 and older.
Gender
There is no FDA guidance on the use of Leuprolide with respect to specific gender populations.
Race
There is no FDA guidance on the use of Leuprolide with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Leuprolide in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Leuprolide in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Leuprolide in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Leuprolide in patients who are immunocompromised.
Administration and Monitoring
Administration
- Subcutaneous
Monitoring
There is limited information regarding Monitoring of Leuprolide in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Leuprolide in the drug label.
Overdosage
- In clinical trials using daily subcutaneous injections of leuprolide acetate in patients with prostate cancer, doses as high as 20 mg/day for up to two years caused no adverse effects differing from those observed with the 1 mg/day dose.
Pharmacology
Mechanism of Action
- Leuprolide acetate, a gonadotropin releasing hormone (GnRH) agonist, acts as a potent inhibitor of gonadotropin secretion when given continuously in therapeutic doses. Animal and human studies indicate that after an initial stimulation, chronic administration of leuprolide acetate results in suppression of testicular and ovarian steroidogenesis. This effect is reversible upon discontinuation of drug therapy.
- In humans, administration of leuprolide acetate results in an initial increase in circulating levels of luteinizing hormone (LH) and follicle stimulating hormone (FSH), leading to a transient increase in levels of the gonadal steroids (testosterone and dihydrotestosterone in males, and estrone and estradiol in premenopausal females). However, continuous administration of leuprolide acetate results in decreased levels of LH and FSH. In males, testosterone is reduced to below castrate threshold (≤ 50 ng/dL). These decreases occur within two to four weeks after initiation of treatment. Long-term studies have shown that continuation of therapy with leuprolide acetate maintains testosterone below the castrate level for up to seven years.
Structure
- ELIGARD® is a sterile polymeric matrix formulation of leuprolide acetate, a GnRH agonist, for subcutaneous injection. It is designed to deliver leuprolide acetate at a controlled rate over a one-, three-, four- or six-month therapeutic period.
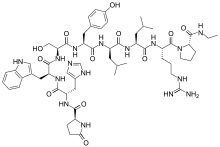
- Leuprolide acetate is a synthetic nonapeptide analog of naturally occurring gonadotropin releasing hormone (GnRH) that, when given continuously, inhibits pituitary gonadotropin secretion and suppresses testicular and ovarian steroidogenesis. The analog possesses greater potency than the natural hormone. The chemical name is 5-oxo-L-prolyl-L-histidyl-L-tryptophyl-L-seryl-L-tyrosyl-D-leucyl-L-leucyl-L-arginyl-N-ethyl-L-prolinamide acetate (salt) with the following structural formula:
- ELIGARD® is prefilled and supplied in two separate, sterile syringes whose contents are mixed immediately prior to administration. The two syringes are joined and the single dose product is mixed until it is homogenous. ELIGARD® is administered subcutaneously, where it forms a solid drug delivery depot.
- One syringe contains the ATRIGEL® Delivery System and the other contains leuprolide acetate. ATRIGEL® is a polymeric (non-gelatin containing) delivery system consisting of a biodegradable poly (DL-lactide-co-glycolide) (PLGH or PLG) polymer formulation dissolved in a biocompatible solvent, N-methyl-2-pyrrolidone (NMP).
- Refer to TABLE 5 for the delivery system composition and constituted product formulation for each ELIGARD® product.
Pharmacodynamics
- Following the first dose of ELIGARD®, mean serum testosterone concentrations transiently increased, then fell to below castrate threshold (≤ 50 ng/dL) within three weeks for all ELIGARD® concentrations.
- Continued monthly treatment with ELIGARD® 7.5 mg maintained castrate testosterone suppression throughout the study. No breakthrough of testosterone concentrations above castrate threshold (> 50 ng/dL) occurred at any time during the study once castrate suppression was achieved (FIGURE 11).
- One patient received less than a full dose of ELIGARD® 22.5 mg at baseline, never suppressed and withdrew from the study at Day 73. Of the 116 patients remaining in the study, 115 (99%) had serum testosterone levels below the castrate threshold by Month 1 (Day 28). By Day 35, 116 (100%) had serum testosterone levels below the castrate threshold. Once testosterone suppression was achieved, one patient (< 1%) demonstrated breakthrough (concentrations > 50 ng/dL after achieving castrate levels) following the initial injection; that patient remained below the castrate threshold following the second injection (FIGURE 12).
- One patient withdrew from the ELIGARD® 30 mg study at Day 14. Of the 89 patients remaining in the study, 85 (96%) had serum testosterone levels below the castrate threshold by Month 1 (Day 28). By Day 42, 89 (100%) of patients attained castrate testosterone suppression. Once castrate testosterone suppression was achieved, three patients (3%) demonstrated breakthrough (concentrations > 50 ng/dL after achieving castrate levels) (FIGURE 13).
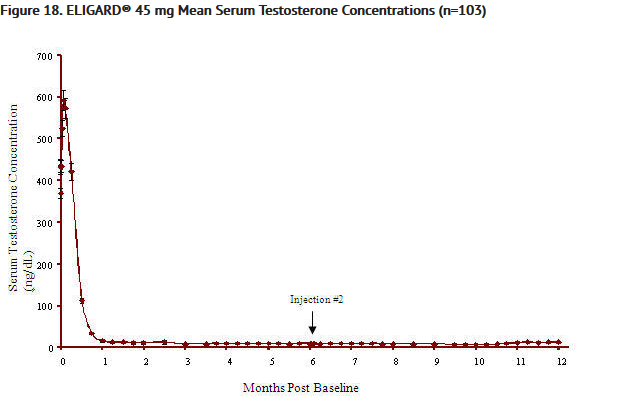
- One patient at Day 1 and another patient at Day 29 were withdrawn from the ELIGARD® 45 mg study. Of the 109 patients remaining in the study, 108 (99.1%) had serum testosterone levels below the castrate threshold by Month 1 (Day 28). One patient did not achieve castrate suppression and was withdrawn from the study at Day 85. Once castrate testosterone suppression was achieved, one patient (< 1%) demonstrated breakthrough (concentrations > 50 ng/dL after achieving castrate levels) (FIGURE 14).
- Leuprolide acetate is not active when given orally.
Pharmacokinetics
- Absorption
- ELIGARD® 7.5 mg
- The pharmacokinetics/pharmacodynamics observed during three once-monthly injections in 20 patients with advanced prostate cancer is shown in Figure 11. Mean serum leuprolide concentrations following the initial injection rose to 25.3 ng/mL (Cmax) at approximately 5 hours after injection. After the initial increase following each injection, serum concentrations remained relatively constant (0.28 – 2.00 ng/mL).
- ELIGARD® 22.5 mg
- The pharmacokinetics/pharmacodynamics observed during two injections every three months (ELIGARD® 22.5 mg) in 22 patients with advanced prostate cancer is shown in Figure 12. Mean serum leuprolide concentrations rose to 127 ng/mL and 107 ng/mL at approximately 5 hours following the initial and second injections, respectively. After the initial increase following each injection, serum concentrations remained relatively constant (0.2 – 2.0 ng/mL).
- ELIGARD® 30 mg
- The pharmacokinetics/pharmacodynamics observed during injections administered initially and at four months (ELIGARD® 30 mg ) in 24 patients with advanced prostate cancer is shown in Figure 13. Mean serum leuprolide concentrations following the initial injection rose rapidly to 150 ng/mL (Cmax) at approximately 3.3 hours after injection. After the initial increase following each injection, mean serum concentrations remained relatively constant (0.1 – 1.0 ng/mL).
- ELIGARD® 45 mg
- The pharmacokinetics/pharmacodynamics observed during injections administered initially and at six months (ELIGARD® 45 mg) in 27 patients with advanced prostate cancer is shown in Figure 14. Mean serum leuprolide concentrations rose to 82 ng/mL and 102 ng/mL (Cmax) at approximately 4.5 hours following the initial and second injections, respectively. After the initial increase following each injection, mean serum concentrations remained relatively constant (0.2 – 2.0 ng/mL).
- Distribution
- The mean steady-state volume of distribution of leuprolide following intravenous bolus administration to healthy male volunteers was 27 L. In vitro binding to human plasma proteins ranged from 43% to 49%.
- Metabolism
- In healthy male volunteers, a 1-mg bolus of leuprolide administered intravenously revealed that the mean systemic clearance was 8.34 L/h, with a terminal elimination half-life of approximately 3 hours based on a two compartment model.
- No drug metabolism study was conducted with ELIGARD®. Upon administration with different leuprolide acetate formulations, the major metabolite of leuprolide acetate is a pentapeptide (M-1) metabolite.
- Excretion
- No drug excretion study was conducted with ELIGARD®.
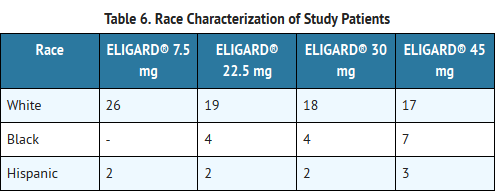
- Race
- In patients studied, mean serum leuprolide concentrations were similar regardless of race. Refer to TABLE 6 for distribution of study patients by race.
- Renal and Hepatic Insufficiency
- The pharmacokinetics of ELIGARD® in hepatically and renally impaired patients have not been determined.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
- Two-year carcinogenicity studies were conducted with leuprolide acetate in rats and mice. In rats, a dose-related increase of benign pituitary hyperplasia and benign pituitary adenomas was noted at 24 months when the drug was administered subcutaneously at high daily doses (0.6 to 4 mg/kg). There was a significant but not dose-related increase of pancreatic islet-cell adenomas in females and of testicular interstitial cell adenomas in males (highest incidence in the low dose group). In mice, no leuprolide acetate-induced tumors or pituitary abnormalities were observed at a dose as high as 60 mg/kg for two years. Patients have been treated with leuprolide acetate for up to three years with doses as high as 10 mg/day and for two years with doses as high as 20 mg/day without demonstrable pituitary abnormalities. No carcinogenicity studies have been conducted with ELIGARD®.
- Mutagenicity studies have been performed with leuprolide acetate using bacterial and mammalian systems and with ELIGARD® 7.5 mg in bacterial systems. These studies provided no evidence of a mutagenic potential.
Clinical Studies
- One open-label, multicenter study was conducted with each ELIGARD® formulation (7.5 mg, 22.5 mg, 30 mg, and 45 mg) in patients with Jewett stage A though D prostate cancer who were treated with at least a single injection of study drug (TABLE 7). These studies evaluated the achievement and maintenance of castrate serum testosterone suppression over the duration of therapy (FIGURES 15-18).
- During the AGL9904 study using ELIGARD® 7.5 mg, once testosterone suppression was achieved, no patients (0%) demonstrated breakthrough (concentration >50 ng/dL) at any time in the study.
- During the AGL9909 study using ELIGARD® 22.5 mg, once testosterone suppression was achieved, only one patient (< 1%) demonstrated breakthrough following the initial injection; that patient remained below the castrate threshold following the second injection.
- During the AGL0001 study using ELIGARD® 30 mg, once testosterone suppression was achieved, three patients (3%) demonstrated breakthrough. In the first of these patients, a single serum testosterone concentration of 53 ng/dL was reported on the day after the second injection. In this patient, castrate suppression was reported for all other timepoints. In the second patient, a serum testosterone concentration of 66 ng/dL was reported immediately prior to the second injection. This rose to a maximum concentration of 147 ng/dL on the second day after the second injection. In this patient, castrate suppression was again reached on the seventh day after the second injection and was maintained thereafter. In the final patient, serum testosterone concentrations > 50 ng/dL were reported at 2 and at 8 hours after the second injection. Serum testosterone concentration rose to a maximum of 110 ng/dL on the third day after the second injection. In this patient, castrate suppression was again reached eighteen days after the second injection and was maintained until the final day of the study, when a single serum testosterone concentration of 55 ng/dL was reported.
- During the AGL0205 study using ELIGARD® 45 mg, once testosterone suppression was achieved, one patient (<1%) demonstrated breakthrough. This patient reached castrate suppression at Day 21 and remained suppressed until Day 308 when his testosterone level rose to 112 ng/dL. At Month 12 (Day 336), his testosterone was 210 ng/dL.
- Serum PSA decreased in all patients in all studies whose Baseline values were elevated above the normal limit. Refer to TABLE 8 for a summary of the effectiveness of ELIGARD® in reducing serum PSA values.
- Other secondary efficacy endpoints evaluated included WHO performance status, bone pain, urinary pain and urinary signs and symptoms. Refer to TABLE 9 for a summary of these endpoints.
How Supplied
- ELIGARD® is available in a single use kit of a two syringe-mixing system in the following strengths:
- ELIGARD® 7.5 mg – NDC 0024-0793-75
- ELIGARD® 22.5 mg – NDC 0024-0222-05
- ELIGARD® 30 mg – NDC 0024-0610-30
- ELIGARD® 45 mg – NDC 0024-0605-45
- Storage:
- Store at 2 – 8 °C (35.6 – 46.4 °F)
Storage
There is limited information regarding Leuprolide Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Leuprolide |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Leuprolide |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- As with other GnRH agonists, patients may experience hot flashes. During the first few weeks of treatment, patients may also experience increased bone pain, increased difficulty in urinating, and the onset or aggravation of weakness or paralysis. Patients should notify their doctor if they develop new or worsened symptoms after beginning ELIGARD® treatment. Patients should be told about the injection site related adverse reactions, such as transient burning/stinging, pain, bruising, and redness. These injection site reactions are usually mild and reversible. If they do not resolve, patients should tell their doctor. If the patient experiences an allergic reaction, they should contact their doctor immediately.
Precautions with Alcohol
- Alcohol-Leuprolide interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- ELIGARD®[8]
Look-Alike Drug Names
There is limited information regarding Leuprolide Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ Boccardo F, Rubagotti A, Amoroso D, Agostara B, Amadori D, Gallo L; et al. (1999). "Endocrinological and clinical evaluation of two depot formulations of leuprolide acetate in pre- and perimenopausal breast cancer patients". Cancer Chemother Pharmacol. 43 (6): 461–6. doi:10.1007/s002800050924. PMID 10321505.
- ↑ Liu HC, Lai YM, Davis O, Berkeley AS, Graf M, Grifo J; et al. (1992). "Improved pregnancy outcome with gonadotropin releasing hormone agonist (GnRH-a) stimulation is due to the improvement in oocyte quantity rather than quality". J Assist Reprod Genet. 9 (4): 338–44. PMID 1472811.
- ↑ Fishman A, Kudelka AP, Tresukosol D, Edwards CL, Freedman RS, Kaplan AL; et al. (1996). "Leuprolide acetate for treating refractory or persistent ovarian granulosa cell tumor". J Reprod Med. 41 (6): 393–6. PMID 8799913.
- ↑ Schmidt PJ, Nieman LK, Danaceau MA, Adams LF, Rubinow DR (1998). "Differential behavioral effects of gonadal steroids in women with and in those without premenstrual syndrome". N Engl J Med. 338 (4): 209–16. doi:10.1056/NEJM199801223380401. PMID 9435325.
- ↑ D'Amico AV, Manola J, Loffredo M, Renshaw AA, DellaCroce A, Kantoff PW (2004). "6-month androgen suppression plus radiation therapy vs radiation therapy alone for patients with clinically localized prostate cancer: a randomized controlled trial". JAMA. 292 (7): 821–7. doi:10.1001/jama.292.7.821. PMID 15315996.
- ↑ Gleave ME, Goldenberg SL, Chin JL, Warner J, Saad F, Klotz LH; et al. (2001). "Randomized comparative study of 3 versus 8-month neoadjuvant hormonal therapy before radical prostatectomy: biochemical and pathological effects". J Urol. 166 (2): 500–6, discussion 506-7. PMID 11458055.
- ↑ Friedman AJ, Barbieri RL, Doubilet PM, Fine C, Schiff I (1988). "A randomized, double-blind trial of a gonadotropin releasing-hormone agonist (leuprolide) with or without medroxyprogesterone acetate in the treatment of leiomyomata uteri". Fertil Steril. 49 (3): 404–9. PMID 2963759.
- ↑ "ELIGARD leuprolide acetate kit".
{{#subobject:
|Page Name=Leuprolide
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Leuprolide |Label Name=Leuprolide10.png
}}
{{#subobject:
|Label Page=Leuprolide |Label Name=Leuprolide11.png
}}
{{#subobject:
|Label Page=Leuprolide |Label Name=Leuprolide12.png
}}
{{#subobject:
|Label Page=Leuprolide |Label Name=Leuprolide13.png
}}
{{#subobject:
|Label Page=Leuprolide |Label Name=Leuprolide14.png
}}