Imipenem-Cilastatin
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Kiran Singh, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Imipenem-Cilastatin is an antibiotic that is FDA approved for the treatment of lower respiratory tract infections,urinary tract infections,intra-abdominal infections, gynecologic infections, bacterial septicemia,bone and joint infections, skin and skin structure infections,endocarditis and Polymicrobic infections. Common adverse reactions include hypotension,injection site pain, pruritus,rash,urticaria' nausea , vomiting,phlebitis,dizziness ,somnolence and fever.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- Imipenem and Cilastatin for Injection (I.V.) is indicated for the treatment of serious infections caused by susceptible strains of the designated microorganisms in the conditions listed below:
(1) Lower respiratory tract infections. Staphylococcus aureus (penicillinase-producing strains), Acinetobacter species, Enterobacter species, Escherichia coli, Haemophilus influenzae, Haemophilus parainfluenzae*, Klebsiella species, Serratia marcescens
(2) Urinary tract infections (complicated and uncomplicated). Enterococcus faecalis, Staphylococcus aureus (penicillinase-producing strains)*, Enterobacter species, Escherichia coli, Klebsiella species, Morganella morganii*, Proteus vulgaris*, Providencia rettgeri*, Pseudomonas aeruginosa
(3) Intra-abdominal infections. Enterococcus faecalis, Staphylococcus aureus (penicillinase-producing strains)*, Staphylococcus epidermidis, Citrobacter species, Enterobacter species, Escherichia coli, Klebsiella species, Morganella morganii*, Proteus species, Pseudomonas aeruginosa, Bifidobacterium species, Clostridium species, Eubacterium species, Peptococcus species, Peptostreptococcus species, Propionibacterium species*, Bacteroides species including B. fragilis, Fusobacterium species
(4) Gynecologic infections. Enterococcus faecalis, Staphylococcus aureus (penicillinase-producing strains)*, Staphylococcus epidermidis, Streptococcus agalactiae (Group B streptococci), Enterobacter species*, Escherichia coli, Gardnerella vaginalis, Klebsiella species*, Proteus species, Bifidobacterium species*, Peptococcus species*, Peptostreptococcus species, Propionibacterium species*, Bacteroides species including B. fragilis*
(5) Bacterial septicemia. Enterococcus faecalis, Staphylococcus aureus (penicillinase-producing strains), Enterobacter species, Escherichia coli, Klebsiella species, Pseudomonas aeruginosa, Serratia species*, Bacteroides species including B. fragilis*
(6) Bone and joint infections. Enterococcus faecalis, Staphylococcus aureus (penicillinase-producing strains), Staphylococcus epidermidis, Enterobacter species, Pseudomonas aeruginosa
(7) Skin and skin structure infections. Enterococcus faecalis, Staphylococcus aureus (penicillinase-producing strains), Staphylococcus epidermidis, Acinetobacter species, Citrobacter species, Enterobacter species, Escherichia coli, Klebsiella species, Morganella morganii, Proteus vulgaris, Providencia rettgeri*, Pseudomonas aeruginosa, Serratia species, Peptococcus species, Peptostreptococcus species, Bacteroides species including B. fragilis, Fusobacterium species*
(8) Endocarditis. Staphylococcus aureus (penicillinase-producing strains)
(9) Polymicrobic infections. Imipenem and Cilastatin for Injection (I.V.) is indicated for polymicrobic infections including those in which S. pneumoniae (pneumonia, septicemia), S. pyogenes (skin and skin structure), or nonpenicillinase-producing S. aureus is one of the causative organisms. However, monobacterial infections due to these organisms are usually treated with narrower spectrum antibiotics, such as penicillin G.
- Imipenem and Cilastatin for Injection (I.V.) is not indicated in patients with meningitis because safety and efficacy have not been established.
- Because of its broad spectrum of bactericidal activity against gram-positive and gram-negative aerobic and anaerobic bacteria, Imipenem and Cilastatin for Injection (I.V.) is useful for the treatment of mixed infections and as presumptive therapy prior to the identification of the causative organisms.
- Although clinical improvement has been observed in patients with cystic fibrosis, chronic pulmonary disease, and lower respiratory tract infections caused by Pseudomonas aeruginosa, bacterial eradication may not necessarily be achieved.
- As with other beta-lactam antibiotics, some strains of Pseudomonas aeruginosa may develop resistance fairly rapidly during treatment with Imipenem and Cilastatin for Injection (I.V.) During therapy of Pseudomonas aeruginosa infections, periodic susceptibility testing should be done when clinically appropriate.
- Infections resistant to other antibiotics, for example, cephalosporins, penicillin, and aminoglycosides, have been shown to respond to treatment with Imipenem and Cilastatin for Injection (I.V.).
- To reduce the development of drug-resistant bacteria and maintain the effectiveness of Imipenem and Cilastatin for Injection (I.V.) and other antibacterial drugs, Imipenem and Cilastatin for Injection (I.V.) should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Dosage
Adults
- The dosage recommendations for Imipenem and Cilastatin for Injection (I.V.) represent the quantity of imipenem to be administered. An equivalent amount of cilastatin is also present in the solution. Each 125 mg, 250 mg, or 500 mg dose should be given by intravenous administration over 20 to 30 minutes. Each 750 mg or 1000 mg dose should be infused over 40 to 60 minutes. In patients who develop nausea during the infusion, the rate of infusion may be slowed.
- The total daily dosage for Imipenem and Cilastatin for Injection (I.V.) should be based on the type or severity of infection and given in equally divided doses based on consideration of degree of susceptibility of the pathogen(s), renal function, and body weight. Adult patients with impaired renal function, as judged by creatinine clearance ≤70 mL/min/1.73 m2, require adjustment of dosage as described in the succeeding section of these guidelines.
- Intravenous Dosage Schedule for Adults with Normal Renal Function and Body Weight ≥70 kg
- Doses cited in TABLE 3 are based on a patient with normal renal function and a body weight of 70 kg. These doses should be used for a patient with a creatinine clearance of ≥71 mL/min/1.73 m2 and a body weight of ≥70 kg. A reduction in dose must be made for a patient with a creatinine clearance of ≤70 mL/min/1.73 m2 and/or a body weight less than 70 kg. (See Tables 4 and 5.)
- Dosage regimens in column A of TABLE 3 are recommended for infections caused by fully susceptible organisms which represent the majority of pathogenic species. Dosage regimens in column B of TABLE 3 are recommended for infections caused by organisms with moderate susceptibility to imipenem, primarily some strains of P. aeruginosa.
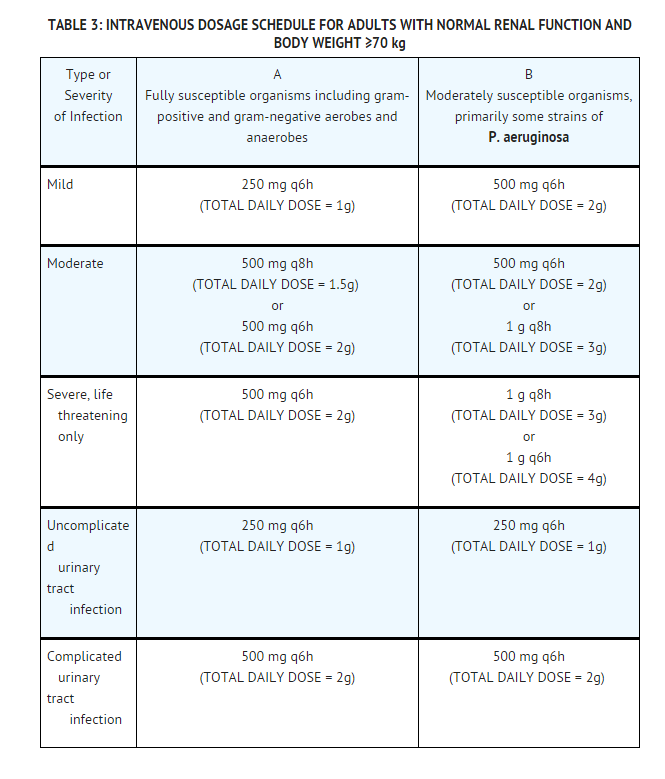
- Due to the high antimicrobial activity of Imipenem and Cilastatin for Injection (I.V.), it is recommended that the maximum total daily dosage not exceed 50 mg/kg/day or 4 g/day, whichever is lower. There is no evidence that higher doses provide greater efficacy. However, patients over twelve years of age with cystic fibrosis and normal renal function have been treated with Imipenem and Cilastatin for Injection (I.V.) at doses up to 90 mg/kg/day in divided doses, not exceeding 4 g/day.
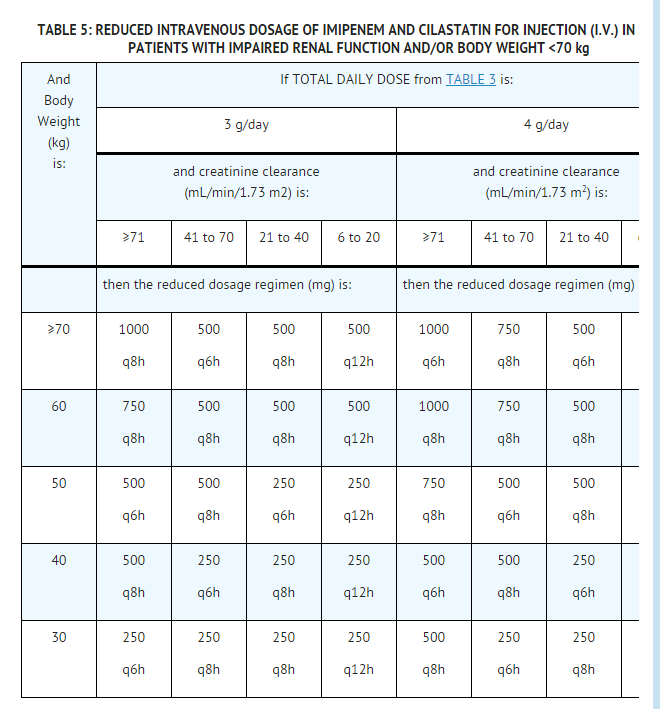
- Reduced Intravenous Schedule for Adults with Impaired Renal Function and/or Body Weight <70 kg
- Patients with creatinine clearance of ≤70 mL/min/1.73 m2 and/or body weight less than 70 kg require dosage reduction of Imipenem and Cilastatin for Injection (I.V.) as indicated in the tables below. Creatinine clearance may be calculated from serum creatinine concentration by the following equation:
- Tcc (Males) = (wt. in kg) (140 – age)
(72) (creatinine in mg/dL)
- Tcc (Females) = 0.85 × above value
- To determine the dose for adults with impaired renal function and/or reduced body weight:
- Choose a total daily dose from TABLE 3 based on infection characteristics.
a) If the total daily dose is 1 g, 1.5 g, or 2 g, use the appropriate subsection of TABLE 4 and continue with step 3.
b) If the total daily dose is 3 g or 4 g, use the appropriate subsection of TABLE 5 and continue with step 3.
- From Table 4 or 5:
a) Select the body weight on the far left which is closest to the patient's body weight (kg).
b) Select the patient's creatinine clearance category.
c) Where the row and column intersect is the reduced dosage regimen.
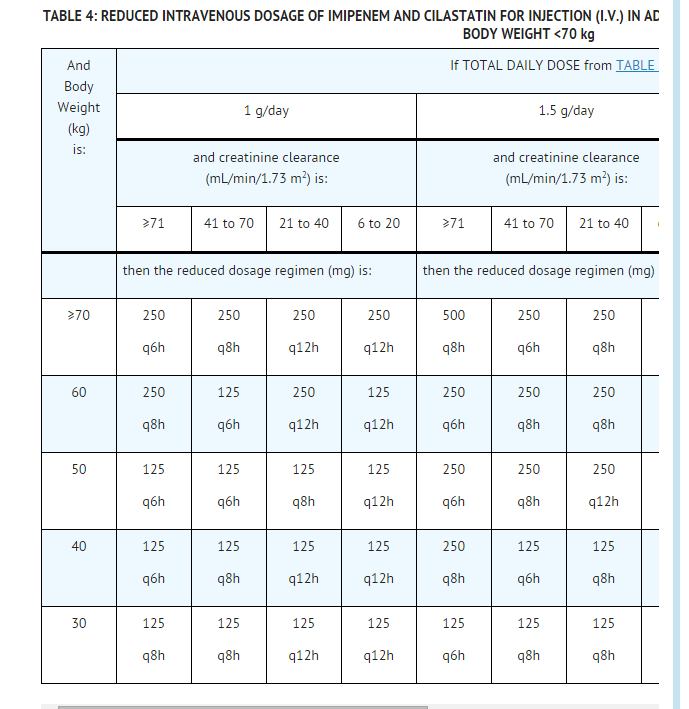
- Patients with creatinine clearances of 6 to 20 mL/min/1.73 m2 should be treated with Imipenem and Cilastatin for Injection (I.V.) 125 mg or 250 mg every 12 hours for most pathogens. There may be an increased risk of seizures when doses of 500 mg every 12 hours are administered to these patients.
- Patients with creatinine clearance ≤5 mL/min/1.73 m2 should not receive Imipenem and Cilastatin for Injection (I.V.) unless hemodialysis is instituted within 48 hours. There is inadequate information to recommend usage of Imipenem and Cilastatin for Injection (I.V.) for patients undergoing peritoneal dialysis.
Hemodialysis
- When treating patients with creatinine clearances of ≤5 mL/min/1.73 m2 who are undergoing hemodialysis, use the dosage recommendations for patients with creatinine clearances of 6 to 20 mL/min/1.73 m2. (See REDUCED INTRAVENOUS DOSAGE SCHEDULE FOR ADULTS WITH IMPAIRED RENAL FUNCTION AND/OR BODY WEIGHT <70 KG.) Both imipenem and cilastatin are cleared from the circulation during hemodialysis. The patient should receive Imipenem and Cilastatin for Injection (I.V.) after hemodialysis and at 12 hour intervals timed from the end of that hemodialysis session. Dialysis patients, especially those with background CNS disease, should be carefully monitored; for patients on hemodialysis, Imipenem and Cilastatin for Injection (I.V.) is recommended only when the benefit outweighs the potential risk of seizures.
PREPARATION OF SOLUTION
- After constitution it is to be solubilized in a suitable parenteral fluid prior to intravenous infusion.
Vials
- Contents of the vials must be suspended and transferred to 100 mL of an appropriate infusion solution.
- A suggested procedure is to add approximately 10 mL from the appropriate infusion solution (see list of diluents under COMPATIBILITY AND STABILITY) to the vial. Shake well and transfer the resulting suspension to the infusion solution container.
- Benzyl alcohol as a preservative has been associated with toxicity in neonates. While toxicity has not been demonstrated in pediatric patients greater than three months of age, small pediatric patients in this age range may also be at risk for benzyl alcohol toxicity. Therefore, diluents containing benzyl alcohol should not be used when Imipenem and Cilastatin for Injection (I.V.) is constituted for administration to pediatric patients in this age range.
CAUTION: THE SUSPENSION IS NOT FOR DIRECT INFUSION.
- Repeat with an additional 10 mL of infusion solution to ensure complete transfer of vial contents to the infusion solution. The resulting mixture should be agitated until clear.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Imipenem-Cilastatin in adult patients.
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Imipenem-Cilastatin in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Indications
- Imipenem and Cilastatin for Injection (I.V.) is indicated for the treatment of serious infections caused by susceptible strains of the designated microorganisms in the conditions listed below:
(1) Lower respiratory tract infections. Staphylococcus aureus (penicillinase-producing strains), Acinetobacter species, Enterobacter species, Escherichia coli, Haemophilus influenzae, Haemophilus parainfluenzae*, Klebsiella species, Serratia marcescens
(2) Urinary tract infections (complicated and uncomplicated). Enterococcus faecalis, Staphylococcus aureus (penicillinase-producing strains)*, Enterobacter species, Escherichia coli, Klebsiella species, Morganella morganii*, Proteus vulgaris*, Providencia rettgeri*, Pseudomonas aeruginosa
(3) Intra-abdominal infections. Enterococcus faecalis, Staphylococcus aureus (penicillinase-producing strains)*, Staphylococcus epidermidis, Citrobacter species, Enterobacter species, Escherichia coli, Klebsiella species, Morganella morganii*, Proteus species, Pseudomonas aeruginosa, Bifidobacterium species, Clostridium species, Eubacterium species, Peptococcus species, Peptostreptococcus species, Propionibacterium species*, Bacteroides species including B. fragilis, Fusobacterium species
(4) Gynecologic infections. Enterococcus faecalis, Staphylococcus aureus (penicillinase-producing strains)*, Staphylococcus epidermidis, Streptococcus agalactiae (Group B streptococci), Enterobacter species*, Escherichia coli, Gardnerella vaginalis, Klebsiella species*, Proteus species, Bifidobacterium species*, Peptococcus species*, Peptostreptococcus species, Propionibacterium species*, Bacteroides species including B. fragilis*
(5) Bacterial septicemia. Enterococcus faecalis, Staphylococcus aureus (penicillinase-producing strains), Enterobacter species, Escherichia coli, Klebsiella species, Pseudomonas aeruginosa, Serratia species*, Bacteroides species including B. fragilis*
(6) Bone and joint infections. Enterococcus faecalis, Staphylococcus aureus (penicillinase-producing strains), Staphylococcus epidermidis, Enterobacter species, Pseudomonas aeruginosa
(7) Skin and skin structure infections. Enterococcus faecalis, Staphylococcus aureus (penicillinase-producing strains), Staphylococcus epidermidis, Acinetobacter species, Citrobacter species, Enterobacter species, Escherichia coli, Klebsiella species, Morganella morganii, Proteus vulgaris, Providencia rettgeri*, Pseudomonas aeruginosa, Serratia species, Peptococcus species, Peptostreptococcus species, Bacteroides species including B. fragilis, Fusobacterium species*
(8) Endocarditis. Staphylococcus aureus (penicillinase-producing strains)
(9) Polymicrobic infections. Imipenem and Cilastatin for Injection (I.V.) is indicated for polymicrobic infections including those in which S. pneumoniae (pneumonia, septicemia), S. pyogenes (skin and skin structure), or nonpenicillinase-producing S. aureus is one of the causative organisms. However, monobacterial infections due to these organisms are usually treated with narrower spectrum antibiotics, such as penicillin G.
- Imipenem and Cilastatin for Injection (I.V.) is not indicated in patients with meningitis because safety and efficacy have not been established.
- Because of its broad spectrum of bactericidal activity against gram-positive and gram-negative aerobic and anaerobic bacteria, Imipenem and Cilastatin for Injection (I.V.) is useful for the treatment of mixed infections and as presumptive therapy prior to the identification of the causative organisms.
- Although clinical improvement has been observed in patients with cystic fibrosis, chronic pulmonary disease, and lower respiratory tract infections caused by Pseudomonas aeruginosa, bacterial eradication may not necessarily be achieved.
- As with other beta-lactam antibiotics, some strains of Pseudomonas aeruginosa may develop resistance fairly rapidly during treatment with Imipenem and Cilastatin for Injection (I.V.) During therapy of Pseudomonas aeruginosa infections, periodic susceptibility testing should be done when clinically appropriate.
- Infections resistant to other antibiotics, for example, cephalosporins, penicillin, and aminoglycosides, have been shown to respond to treatment with Imipenem and Cilastatin for Injection (I.V.).
- To reduce the development of drug-resistant bacteria and maintain the effectiveness of Imipenem and Cilastatin for Injection (I.V.) and other antibacterial drugs, Imipenem and Cilastatin for Injection (I.V.) should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Dosage
- For pediatric patients ≥3 months of age, the recommended dose for non-CNS infections is 15 to 25 mg/kg/dose administered every six hours. Based on studies in adults, the maximum daily dose for treatment of infections with fully susceptible organisms is 2 g per day, and of infections with moderately susceptible organisms (primarily some strains of P. aeruginosa) is 4 g/day. Higher doses (up to 90 mg/kg/day in older children) have been used in patients with cystic fibrosis.
- For pediatric patients ≤3 months of age (weighing ≥1,500 g), the following dosage schedule is recommended for non-CNS infections:
- <1 wk of age: 25 mg/kg every 12 hrs
- 1 to 4 wks of age: 25 mg/kg every 8 hrs
- 4 wks to 3 mos. of age: 25 mg/kg every 6 hrs.
- Doses less than or equal to 500 mg should be given by intravenous infusion over 15 to 30 minutes. Doses greater than 500 mg should be given by intravenous infusion over 40 to 60 minutes.
- Imipenem and Cilastatin for Injection (I.V.) is not recommended in pediatric patients with CNS infections because of the risk of seizures.
- Imipenem and Cilastatin for Injection (I.V.) is not recommended in pediatric patients <30 kg with impaired renal function, as no data are available.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Imipenem-Cilastatin in pediatric patients.
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Imipenem-Cilastatin in pediatric patients.
Contraindications
- Imipenem and Cilastatin for Injection (I.V.) is contraindicated in patients who have shown hypersensitivity to any component of this product.
Warnings
- SERIOUS AND OCCASIONALLY FATAL HYPERSENSITIVITY (ANAPHYLACTIC) REACTIONS HAVE BEEN REPORTED IN PATIENTS RECEIVING THERAPY WITH BETA-LACTAMS. THESE REACTIONS ARE MORE APT TO OCCUR IN PERSONS WITH A HISTORY OF SENSITIVITY TO MULTIPLE ALLERGENS.
- THERE HAVE BEEN REPORTS OF PATIENTS WITH A HISTORY OF PENICILLIN HYPERSENSITIVITY WHO HAVE EXPERIENCED SEVERE HYPERSENSITIVITY REACTIONS WHEN TREATED WITH ANOTHER BETA-LACTAM. BEFORE INITIATING THERAPY WITH IMIPENEM AND CILASTATIN FOR INJECTION (I.V.), CAREFUL INQUIRY SHOULD BE MADE CONCERNING PREVIOUS HYPERSENSITIVITY REACTIONS TO PENICILLINS, CEPHALOSPORINS, OTHER BETA-LACTAMS, AND OTHER ALLERGENS. IF AN ALLERGIC REACTION OCCURS, IMIPENEM AND CILASTATIN FOR INJECTION (I.V.) SHOULD BE DISCONTINUED.
- SERIOUS ANAPHYLACTIC REACTIONS REQUIRE IMMEDIATE EMERGENCY TREATMENT WITH EPINEPHRINE. OXYGEN, INTRAVENOUS STEROIDS, AND AIRWAY MANAGEMENT, INCLUDING INTUBATION, MAY ALSO BE ADMINISTERED AS INDICATED.
Seizure Potential
- Seizures and other CNS adverse experiences, such as confusional states and myoclonic activity, have been reported during treatment with Imipenem and Cilastatin for Injection (I.V.).
- Case reports in the literature have shown that co-administration of carbapenems, including imipenem, to patients receiving valproic acid or divalproex sodium results in a reduction in valproic acid concentrations. The valproic acid concentrations may drop below the therapeutic range as a result of this interaction, therefore increasing the risk of breakthrough seizures. Increasing the dose of valproic acid or divalproex sodium may not be sufficient to overcome this interaction. The concomitant use of imipenem and valproic acid/divalproex sodium is generally not recommended. Anti-bacterials other than carbapenems should be considered to treat infections in patients whose seizures are well controlled on valproic acid or divalproex sodium. If administration of Imipenem and Cilastatin for Injection (I.V.) is necessary, supplemental anticonvulsant therapy should be considered.
- Clostridium difficile'' associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including Imipenem and Cilastatin for Injection (I.V.), and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
- C. difficile produces toxins A and B which contribute to the development of CDAD.
- Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
- If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
Adverse Reactions
Clinical Trials Experience
Adults
- Imipenem and Cilastatin for Injection (I.V.) is generally well tolerated. Many of the 1,723 patients treated in clinical trials were severely ill and had multiple background diseases and physiological impairments, making it difficult to determine causal relationship of adverse experiences to therapy with Imipenem and Cilastatin for Injection (I.V.).
Local Adverse Reactions
- Adverse local clinical reactions that were reported as possibly, probably, or definitely related to therapy with Imipenem and Cilastatin for Injection (I.V.) were:
- Phlebitis/thrombophlebitis — 3.1%
- Pain at the injection site — 0.7%
- Erythema at the injection site — 0.4%
- Vein induration — 0.2%
- Infused vein infection — 0.1%
Systemic Adverse Reactions
- The most frequently reported systemic adverse clinical reactions that were reported as possibly, probably, or definitely related to Imipenem and Cilastatin for Injection (I.V.) were nausea (2%), diarrhea (1.8%), vomiting (1.5%), rash (0.9%), fever (0.5%), hypotension (0.4%), seizures (0.4%) , dizziness (0.3%), pruritus (0.3%), urticaria (0.2%), somnolence (0.2%).
- Additional adverse systemic clinical reactions reported as possibly, probably, or definitely drug related occurring in less than 0.2% of the patients or reported since the drug was marketed are listed within each body system in order of decreasing severity:
- Gastrointestinal — pseudomembranous colitis (the onset of pseudomembranous colitis symptoms may occur during or after antibacterial treatment,), hemorrhagic colitis, hepatitis (including fulminant hepatitis), hepatic failure, jaundice, gastroenteritis, abdominal pain, glossitis, tongue papillar hypertrophy, staining of the teeth and/or tongue, heartburn, pharyngeal pain, increased salivation;
- Hematologic — pancytopenia, bone marrow depression, thrombocytopenia, neutropenia, leukopenia, hemolytic anemia;
- CNS — encephalopathy, tremor, confusion, myoclonus, paresthesia, vertigo, headache, psychic disturbances including hallucinations;
- Special Senses — hearing loss, tinnitus, taste perversion;
- Respiratory — chest discomfort, dyspnea, hyperventilation, thoracic spine pain;
- Cardiovascular — palpitations, tachycardia;
- Skin — Stevens-Johnson syndrome, toxic epidermal necrolysis, erythema multiforme, angioneurotic edema, flushing, cyanosis, hyperhidrosis, skin texture changes, candidiasis, pruritus vulvae;
- Body as a whole — polyarthralgia, asthenia/weakness, drug fever;
- Renal — acute renal failure, oliguria/anuria, polyuria, urine discoloration. The role of Imipenem and Cilastatin for Injection (I.V.) in changes in renal function is difficult to assess, since factors predisposing to pre-renal azotemia or to impaired renal function usually have been present.
Adverse Laboratory Changes
- Adverse laboratory changes without regard to drug relationship that were reported during clinical trials or reported since the drug was marketed were:
- Hepatic: Increased ALT (SGPT), AST (SGOT), alkaline phosphatase, bilirubin, and LDH
- Hemic: Increased eosinophils, positive Coombs test, increased WBC, increased platelets, decreased hemoglobin and hematocrit, agranulocytosis, increased monocytes, abnormal prothrombin time, increased lymphocytes, increased basophils
- Electrolytes: Decreased serum sodium, increased potassium, increased chloride
- Renal: Increased BUN, creatinine
- Urinalysis: Presence of urine protein, urine red blood cells, urine white blood cells, urine casts, urine bilirubin, and urine urobilinogen.
Pediatric Patients
- In studies of 178 pediatric patients ≥3 months of age, the following adverse events were noted:
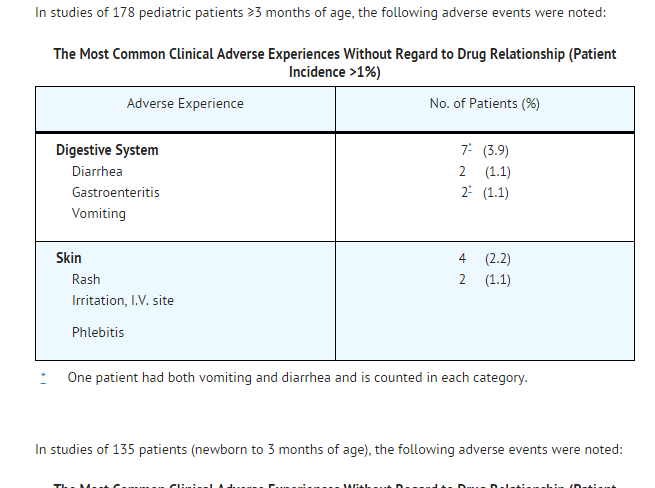
- In studies of 135 patients (newborn to 3 months of age), the following adverse events were noted:
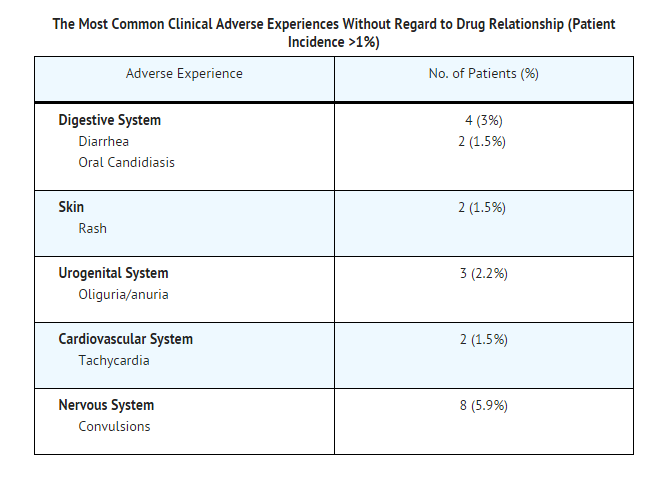
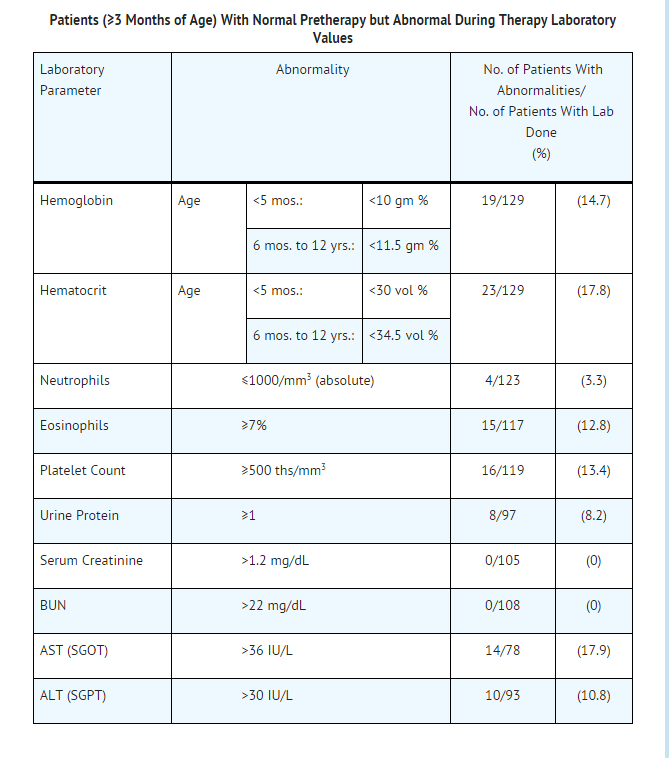

- Examination of published literature and spontaneous adverse event reports suggested a similar spectrum of adverse events in adult and pediatric patients.
Postmarketing Experience
- There is limited information regarding Postmarketing Experience of Imipenem-Cilastatin in the drug label.
Drug Interactions
There is limited information regarding Imipenem-Cilastatin Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): Pregnancy: Teratogenic Effects
Pregnancy Category C:
- Teratology studies with cilastatin sodium at doses of 30, 100, and 300 mg/kg/day administered intravenously to rabbits and 40, 200, and 1000 mg/kg/day administered subcutaneously to rats, up to approximately 1.9 and 3.2 times†† the maximum recommended daily human dose (on a mg/m2 body surface area basis) of the intravenous formulation of imipenem-cilastatin sodium (50 mg/kg/day) in the two species, respectively, showed no evidence of adverse effect on the fetus. No evidence of teratogenicity was observed in rabbits given imipenem at intravenous doses of 15, 30 or 60 mg/kg/day and rats given imipenem at intravenous doses of 225, 450, or 900 mg/kg/day, up to approximately 0.4 and 2.9 times†† the maximum recommended daily human dose (on a mg/m2 body surface area basis) in the two species, respectively.
- Teratology studies with imipenem-cilastatin sodium at intravenous doses of 20 and 80, and a subcutaneous dose of 320 mg/kg/day, up to 0.5 times†† (mice) to approximately equal to (rats) the highest recommended daily intravenous human dose (on a mg/m2 body surface area basis) in pregnant rodents during the period of major organogenesis, revealed no evidence of teratogenicity.
- Imipenem-cilastatin sodium, when administered subcutaneously to pregnant rabbits at dosages equivalent to the usual human dose of the intravenous formulation and higher (1000 to 4000 mg/day), caused body weight loss, diarrhea, and maternal deaths. When comparable doses of imipenem-cilastatin sodium were given to non-pregnant rabbits, body weight loss, diarrhea, and deaths were also observed. This intolerance is not unlike that seen with other beta-lactam antibiotics in this species and is probably due to alteration of gut flora.
- A teratology study in pregnant cynomolgus monkeys given imipenem-cilastatin sodium at doses of 40 mg/kg/day (bolus intravenous injection) or 160 mg/kg/day (subcutaneous injection) resulted in maternal toxicity including emesis, inappetence, body weight loss, diarrhea, abortion, and death in some cases. In contrast, no significant toxicity was observed when non-pregnant cynomolgus monkeys were given doses of imipenem-cilastatin sodium up to 180 mg/kg/day (subcutaneous injection). When doses of imipenem-cilastatin sodium (approximately 100 mg/kg/day or approximately 0.6 times†† the maximum recommended daily human dose of the intravenous formulation) were administered to pregnant cynomolgus monkeys at an intravenous infusion rate which mimics human clinical use, there was minimal maternal intolerance (occasional emesis), no maternal deaths, no evidence of teratogenicity, but an increase in embryonic loss relative to control groups.
- No adverse effects on the fetus or on lactation were observed when imipenem-cilastatin sodium was administered subcutaneously to rats late in gestation at dosages up to 320 mg/kg/day, approximately equal to the highest recommended human dose (on a mg/m2 body surface area basis).
- There are, however, no adequate and well-controlled studies in pregnant women. Imipenem and Cilastatin for Injection (I.V.) should be used during pregnancy only if the potential benefit justifies the potential risk to the mother and fetus.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Imipenem-Cilastatin in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Imipenem-Cilastatin during labor and delivery.
Nursing Mothers
- It is not known whether imipenem-cilastatin sodium is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Imipenem and Cilastatin for Injection (I.V.) is administered to a nursing woman.
Pediatric Use
- Use of Imipenem and Cilastatin for Injection (I.V.) in pediatric patients, neonates to 16 years of age, is supported by evidence from adequate and well-controlled studies of Imipenem and Cilastatin for Injection (I.V.) in adults and by the following clinical studies and published literature in pediatric patients: Based on published studies of 178** pediatric patients ≥3 months of age (with non-CNS infections), the recommended dose of Imipenem and Cilastatin for Injection (I.V.) is 15 to 25 mg/kg/dose administered every six hours. Doses of 25 mg/kg/dose in patients 3 months to <3 years of age, and 15 mg/kg/dose in patients 3 to 12 years of age were associated with mean trough plasma concentrations of imipenem of 1.1±0.4 mcg/mL and 0.6±0.2 mcg/mL following multiple 60-minute infusions, respectively; trough urinary concentrations of imipenem were in excess of 10 mcg/mL for both doses. These doses have provided adequate plasma and urine concentrations for the treatment of non-CNS infections. Based on studies in adults, the maximum daily dose for treatment of infections with fully susceptible organisms is 2 g per day, and of infections with moderately susceptible organisms (primarily some strains of P. aeruginosa) is 4 g/day. (See DOSAGE AND ADMINISTRATION, TABLE 3.) Higher doses (up to 90 mg/kg/day in older children) have been used in patients with cystic fibrosis.
- Based on studies of 135*** pediatric patients ≤3 months of age (weighing ≥1,500 g), the following dosage schedule is recommended for non-CNS infections:
- <1 wk of age: 25 mg/kg every 12 hrs
- 1 to 4 wks of age: 25 mg/kg every 8 hrs
- 4 wks to 3 mos. of age: 25 mg/kg every 6 hrs.
- In a published dose-ranging study of smaller premature infants (670 to 1,890 g) in the first week of life, a dose of 20 mg/kg q12h by 15 to 30 minutes infusion was associated with mean peak and trough plasma imipenem concentrations of 43 mcg/mL and 1.7 mcg/mL after multiple doses, respectively. However, moderate accumulation of cilastatin in neonates may occur following multiple doses of Imipenem and Cilastatin for Injection (I.V.) The safety of this accumulation is unknown.
- Imipenem and Cilastatin for Injection (I.V.) is not recommended in pediatric patients with CNS infections because of the risk of seizures.
- Imipenem and Cilastatin for Injection (I.V.) is not recommended in pediatric patients <30 kg with impaired renal function, as no data are available.
Geriatic Use
- Of the approximately 3600 subjects ≥18 years of age in clinical studies of Imipenem and Cilastatin for Injection (I.V.), including postmarketing studies, approximately 2800 received Imipenem and Cilastatin for Injection (I.V.) Of the subjects who received Imipenem and Cilastatin for Injection (I.V.), data are available on approximately 800 subjects who were 65 and over, including approximately 300 subjects who were 75 and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
- This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
- No dosage adjustment is required based on age. Dosage adjustment in the case of renal impairment is necessary
Gender
There is no FDA guidance on the use of Imipenem-Cilastatin with respect to specific gender populations.
Race
There is no FDA guidance on the use of Imipenem-Cilastatin with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Imipenem-Cilastatin in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Imipenem-Cilastatin in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Imipenem-Cilastatin in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Imipenem-Cilastatin in patients who are immunocompromised.
Administration and Monitoring
Administration
- Intravenous
Monitoring
There is limited information regarding Monitoring of Imipenem-Cilastatin in the drug label.
- Description
IV Compatibility
There is limited information regarding IV Compatibility of Imipenem-Cilastatin in the drug label.
Overdosage
- The acute intravenous toxicity of imipenem-cilastatin sodium in a ratio of 1:1 was studied in mice at doses of 751 to 1359 mg/kg. Following drug administration, ataxia was rapidly produced and clonic convulsions were noted in about 45 minutes. Deaths occurred within 4 to 56 minutes at all doses.
- The acute intravenous toxicity of imipenem-cilastatin sodium was produced within 5 to 10 minutes in rats at doses of 771 to 1583 mg/kg. In all dosage groups, females had decreased activity, bradypnea, and ptosis with clonic convulsions preceding death; in males, ptosis was seen at all dose levels while tremors and clonic convulsions were seen at all but the lowest dose (771 mg/kg). In another rat study, female rats showed ataxia, bradypnea, and decreased activity in all but the lowest dose (550 mg/kg); deaths were preceded by clonic convulsions. Male rats showed tremors at all doses and clonic convulsions and ptosis were seen at the two highest doses (1130 and 1734 mg/kg). Deaths occurred between 6 and 88 minutes with doses of 771 to 1734 mg/kg.
- In the case of overdosage, discontinue Imipenem and Cilastatin for Injection (I.V.), treat symptomatically, and institute supportive measures as required. Imipenem-cilastatin sodium is hemodialyzable. However, usefulness of this procedure in the overdosage setting is questionable.
Pharmacology
Imipenem-Cilastatin
| |
| Combination of | |
| Imipenem | Carbapenem antibiotic |
| Cilastatin | Dehydropeptidase inhibitor |
| Identifiers | |
| CAS number | |
| ATC code | J01 |
| PubChem | |
| Therapeutic considerations | |
| Pregnancy cat. |
C(US) |
| Legal status |
POM(UK) [[Prescription drug|Template:Unicode-only]](US) |
| Routes | Intravenous |
Mechanism of Action
There is limited information regarding Mechanism of action of Imipenem-Cilastatin in the drug label.
Structure
- Imipenem and Cilastatin for Injection (I.V.) is a sterile formulation of imipenem (a thienamycin antibiotic) and cilastatin sodium (the inhibitor of the renal dipeptidase, dehydropeptidase l), with sodium bicarbonate added as a buffer. Imipenem and Cilastatin for Injection (I.V.) is a potent broad spectrum antibacterial agent for intravenous administration.
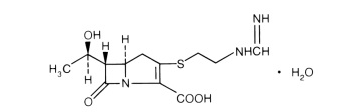
- Imipenem (N-formimidoylthienamycin monohydrate) is a crystalline derivative of thienamycin, which is produced by Streptomyces cattleya. Its chemical name is (5R,6S)-3-2-(formimidoylamino)ethylthio-6-(R)-1-hydroxyethyl-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid monohydrate. It is an off-white, nonhygroscopic crystalline compound with a molecular weight of 317.37. It is sparingly soluble in water and slightly soluble in methanol. Its empirical formula is C12H17N3O4S•H2O, and its structural formula is:
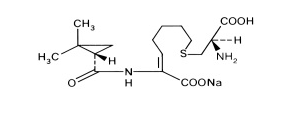
- Cilastatin sodium is the sodium salt of a derivatized heptenoic acid. Its chemical name is sodium(Z)-7(R)-2-amino-2-carboxyethyl]thio]-2-[(S)-2,2-dimethylcyclopropane carboxamido]-2-heptenoate. It is an off-white to yellowish-white, hygroscopic, amorphous compound with a molecular weight of 380.43. It is very soluble in water and in methanol. Its empirical formula is C16H25N2O5SNa, and its structural formula is:
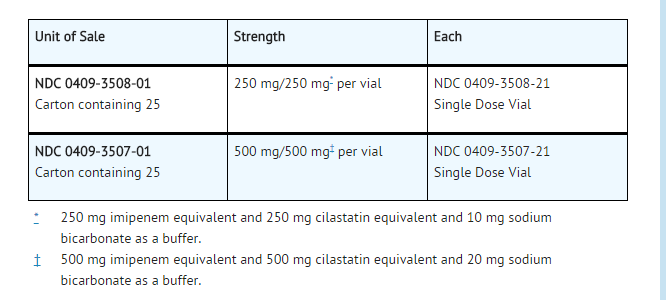
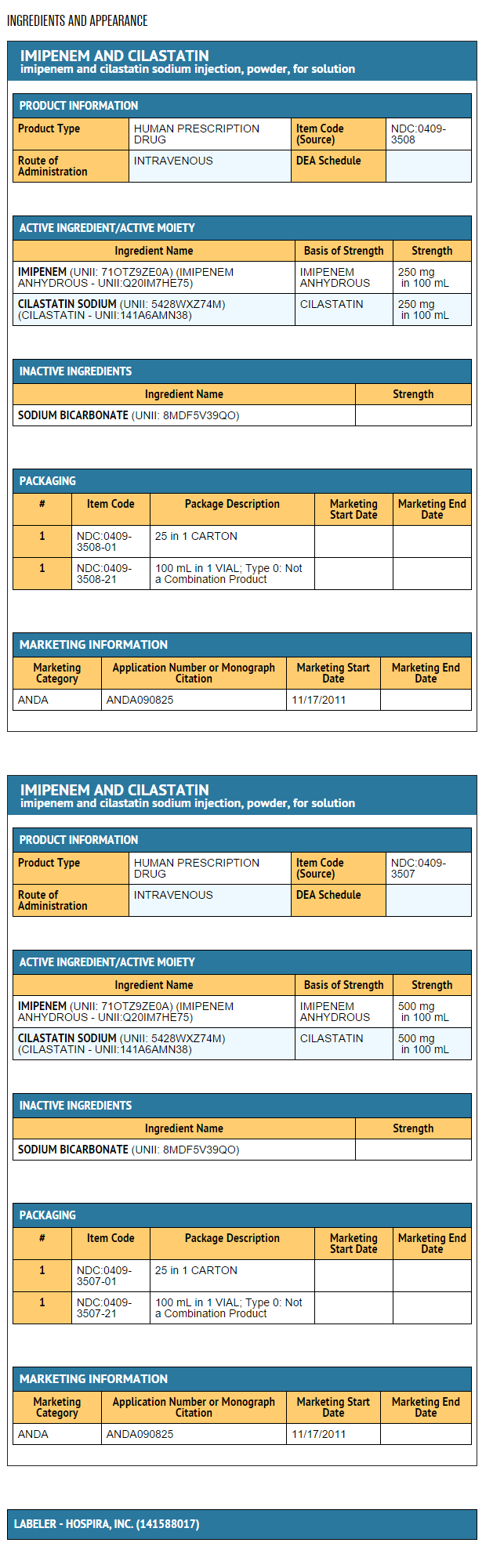
- Imipenem and Cilastatin for Injection (I.V.) is buffered to provide solutions in the pH range of 6.5 to 8.5. There is no significant change in pH when solutions are prepared and used as directed.The 250 mg/250 mg vial contains 250 mg imipenem (anhydrous equivalent) and 250 mg cilastatin as sodium salt. The inactive ingredient is sodium bicarbonate 10 mg as a buffer. The 500 mg/500 mg vial contains 500 mg imipenem (anhydrous equivalent) and 500 mg cilastatin as sodium salt. The inactive ingredient is sodium bicarbonate 20 mg as a buffer. Imipenem and Cilastatin for Injection (I.V.) 250 mg/250 mg contains 18.8 mg of sodium (0.8 mEq) and Imipenem and Cilastatin for Injection (I.V.) 500 mg/500 mg contains 37.5 mg of sodium (1.6 mEq). Solutions of Imipenem and Cilastatin for Injection (I.V.) range from colorless to yellow. Variations of color within this range do not affect the potency of the product.
- After constitution it is to be solubilized in a suitable parenteral fluid prior to intravenous infusion.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Imipenem-Cilastatin in the drug label.
Pharmacokinetics
There is limited information regarding Pharmacokinetics of Imipenem-Cilastatin in the drug label.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Imipenem-Cilastatin in the drug label.
Clinical Studies
- There is limited information regarding Clinical Studies of Imipenem-Cilastatin in the drug label.
How Supplied
- Imipenem and Cilastatin for Injection (I.V.) is supplied as a sterile powder mixture in single dose vials containing imipenem (anhydrous equivalent) and cilastatin sodium as follows:
Storage
There is limited information regarding Imipenem-Cilastatin Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Imipenem-Cilastatin |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel


{{#ask: Label Page::Imipenem-Cilastatin |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Imipenem-Cilastatin in the drug label.
Precautions with Alcohol
- Alcohol-Imipenem-Cilastatin interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- IMIPENEMANDCILASTATIN®<refTemplate:Citewebhttp://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=fe44d24d-55cd-439c-895b-00a38cd59f65</ref>
Look-Alike Drug Names
- A® — B®[1]
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Page Name=Imipenem-Cilastatin
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Imipenem-Cilastatin |Label Name=Imipenem-Cilastatin11.png
}}
{{#subobject:
|Label Page=Imipenem-Cilastatin |Label Name=Imipenem-Cilastatin11.png
}}