Hydrogen selenide
Template:Chembox new Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Hydrogen selenide is H2Se, the simplest hydride of selenium. H2Se is a colorless, flammable gas under standard conditions. It is the most toxic selenium compound with an exposure limit: 0.3 ppm over an 8 hour period. This compound has a very irritating smell of decayed horseradish.
Structure
H2Se adopts a "bent" structure with a H-Se-H bond angle of 91°. Consistent with this structure, three IR-active vibrational bands are observed: 2358, 2345, and 1034 cm−1.
Properties
The properties of H2S and H2Se are similar, although the selenide is more acidic with pKa = 3.89, and the second pKa = 11.0 at 25°C. Reflecting its acidity, H2Se is soluble in water.
Preparation
H2Se can be purchased in cylinders. A number of routes to H2Se have been reported, which are suitable for both large and small scale preparations.
- H2Se is usually prepared by the action of water on Al2Se3, concomitant with formation of hydrated alumina. A related reaction involves the acid hydrolysis of FeSe.
- Al2Se3 + 6 H2O Template:Unicode 2 Al(OH)3 + 3 H2Se
- According to the Sonoda method, H2Se is generated from the reaction of H2O and CO on Se in the presence of Et3N.
- H2Se can by synthesized by the direct reaction of H2 with elemental selenium at >350°C.
Selenium metal can be recovered from H2Se through a reaction with aqueous sulfur dioxide (SO2).
- 2 H2Se + SO2 Template:Unicode 2 H2O + Se + S
Applications
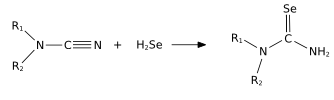
- H2Se is commonly used in the synthesis of Se-containing compounds; it adds across alkenes. Illustrative is the synthesis of selenoureas from nitriles.
- H2Se gas is used to dope semiconductors with selenium.
References
Additional Resources
- Patnaik, Pradyot Handbook of Inorganic Chemicals. McGraw-Hill Handbooks New York (2003).
- V.I. Cohen, Synthesis 1:60-3 (1980).
- Greenwood, N. N., Earnshaw, A. Chemistry of the Elements Second Ed. Butterworth-Heinemann, Boston (1997).
- CRC Handbook of Chemistry and Physics Ed. 82. Chapman and Hall (2001-2002).
- Féher, F. In "Handbook of Preparative Inorganic Chemistry"; Brauer, E., Ed.; Academic: New York, 1963; 1, p 418.
- Sonoda, N.; Kondo K.; Nagano, K.; Kambe, N.; Morimoto, F. Angewandte Chemie, International Edition English 1980, vol. 19, page 308
External links
de:Selenwasserstoff it:Seleniuro di idrogeno