Kaposi's sarcoma-associated herpesvirus
| style="background:#Template:Taxobox colour;"|Template:Taxobox name | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| style="background:#Template:Taxobox colour;" | Virus classification | ||||||||||
|
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
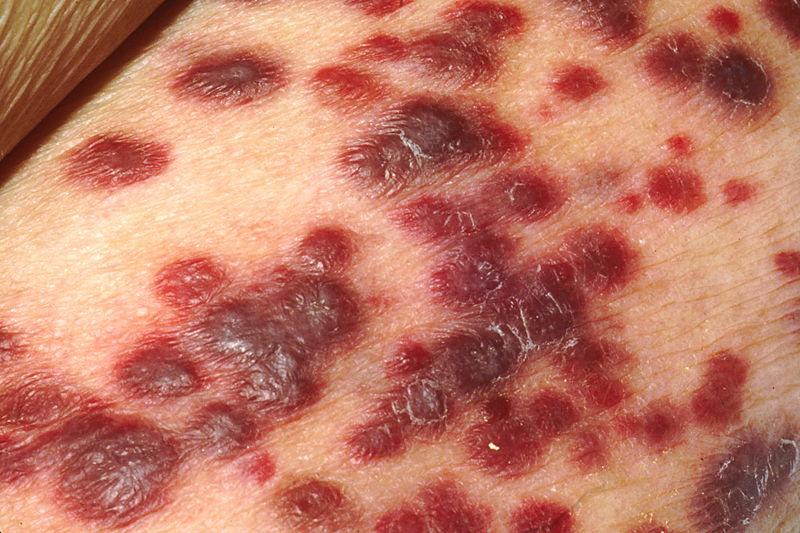
Kaposi's sarcoma-associated herpesvirus (KSHV) is the eighth human herpesvirus; its formal name according to the International Committee on Taxonomy of Viruses is HHV-8. This virus causes Kaposi's sarcoma, a cancer commonly occurring in AIDS patients, as well as primary effusion lymphoma and some types of multicentric Castleman's disease.
History
In 1872, Moritz Kaposi described a blood vessel tumor (originally called "idiopathic multiple pigmented sarcoma of the skin") that has since been eponymously named Kaposi's sarcoma (KS). KS was thought to be an uncommon tumor of Jewish and Mediterranean populations until it was realized that it is actually quite common throughout sub-Saharan Africa. This led to the first suggestions in the 1950s that this tumor might be caused by a virus. With the onset of the AIDS epidemic in the early 1980s, there was a sudden epidemic resurgence of KS affecting primarily gay and bisexual AIDS patients with up to 50% of reported AIDS patients having this tumor--an extraordinary rate of cancer predisposition. Careful analysis of epidemiologic data by Valerie Beral, Thomas Peterman and Harold Jaffe, led these investigators to propose that KS is caused by an unknown sexually transmitted virus that rarely causes tumors unless the host becomes immunosuppressed, as in AIDS.
It was first found by Yuan Chang and Patrick S. Moore, a wife and husband team at Columbia University in 1994, by isolating DNA fragments of a herpesvirus from a Kaposi's sarcoma (KS) tumor in an AIDS patient. As early as 1984, scientists had reported seeing herpesvirus-like structures in KS tumors by electron microscopy. Scientists by then had been searching for the agent causing Kaposi's sarcoma and over 20 agents had been described as the possible cause of KS, including cytomegalovirus and HIV itself. Chang and Moore used representational difference analysis, or RDA, to find KSHV by comparing KS tumor tissue from an AIDS patient to his own unaffected tissue. The idea behind this experiment was that if a virus causes KS, the genomic DNA in the two samples should be precisely identical except for DNA belonging to the virus. In their initial RDA experiment, they isolated two small DNA fragments that represented less than 1% of the actual viral genome. These fragments were similar to but distinct from known herpevirus sequences indicating the presence of a new virus. Starting from these fragments, this research team was then able to sequence the entire genome of the virus less than two years later.
The discovery of this herpesvirus sparked considerable controversy and scientific in-fighting until sufficient data had been collected to show that indeed KSHV was the causative agent of Kaposi's sarcoma. The virus is now known to be a widespread infection of people living in sub-Saharan Africa; intermediate levels of infection occur in Mediterranean populations (including Israel, Saudi Arabia, Italy and Greece) and low levels of infection occur in most Northern European and North American populations. Gay and bi-sexual men are more susceptible to infection (through still unknown routes of sexual transmission) whereas the virus is transmitted through non-sexual routes in developing countries.
Virology
KSHV is a herpesvirus, and is a large double-stranded DNA virus with a protein covering that packages its nucleic acids, called the virion, which is then surrounded by an amorphous protein layer called the tegument, and finally enclosed in a lipid envelope derived in part from the cell membrane. KSHV has a genome which is approximately 165,000 nucleic acid bases in length. It is a rhadinovirus, and is remarkable since it has stolen numerous genes from host cells including genes that encode for complement-binding protein, IL-6, BCL-2, cyclin-D, a G protein-coupled receptor, interferon regulatory factor and Fas-ligand inhibitory protein (FLIP), as well as DNA synthesis proteins including dihydrofolate reductase, thymidine kinase, thymidylate synthetase, DNA polymerase and many others. While no other human tumor virus possesses these same genes, many of these genes target the same cellular pathways targeted by other tumor viruses illustrating that at a basic level, all tumor viruses appear to attack the same cellular control pathways, so-called tumor suppressor pathways.
After infection, the virus enters into lymphocytes where it remains in a latent ("quiet") state. The virus exists as a naked circular piece of DNA called an episome and uses the cellular replication machinery to replicate itself. Various signals such as inflammation may provoke the virus to enter into "lytic replication". When this occurs, the viral episome starts replicating itself in the form of linear DNA molecules that are packaged into virus particles which are expelled from the cell, to infect new cells or to be transmitted to a new host. When the virus enters into lytic replication, thousands of virus particles can be made from a single cell, which usually results in cell death.
Epidemiology
KSHV is an uncommon infection in the United States and in northern Europe, where less than 2% of the general population is infected with the virus. Gay and bisexual men, however, are at high risk for infection with up to 60% of gay men found to be infected in some studies. Among gay men, increasing numbers of sex partners increases the risk for infection. Unlike HIV, it is not clear whether the virus is transmitted through unprotected anal intercourse. Instead, it is likely that it is transmitted between sex partners by oral secretions (e.g. saliva). The exact reasons why gay men are at high risk for infection compared to heterosexuals remains obscure, but may be due to specific sexual behaviors such as deep-kissing or use of saliva as a sexual lubricant during anal intercourse.
In African countries, in contrast, infection is commonly spread through non-sexual routes that remain poorly understood. Young children can be infected (although direct transmission from mothers to their children during pregnancy is uncommon), and rates of infection can continue to increase throughout adulthood. A form of Kaposi's sarcoma occurring in young African children due to this infection is almost uniformly and rapidly fatal. Infection with the KSHV, however, is usually without symptoms in most persons.
In addition to gay men and African populations, this infection is of particular concern to persons receiving organ transplants. Not only is there a risk of transmitting the virus from the donated organ, but recipients are immunosuppressed to avoid organ rejection and are at high risk for developing KS if they are infected. Some studies found that up to 50% of transplant recipients who are infected with KSHV develop Kaposi's sarcoma, which can be life-threatening and can cause loss of the donated organ.
Infection with this virus is thought to be life-long, but a healthy immune system will keep the virus in check. Kaposi's sarcoma occurs when someone who has been infected with KSHV becomes immunocompromised due to AIDS, old age or medical treatment. Since persons infected with KSHV will asymptomatically shed the virus, caution should be used by sex partners in having unprotected sex and activities where saliva might be shared during sexual activity. Avoiding deep-kissing (i.e. tonguing the back of a partner's pharynx) and use of saliva as a sexual lubricant is prudent. Blood tests to detect infection exist, but they are largely restricted to research universities, since biomedical companies have not found it economical to develop these tests.
Treatment
Kaposi's sarcoma is usually a localized tumor that can be treated either surgically or through local irradiation. Chemotherapy with drugs such as liposomal anthracyclines or paclitaxel may be used, particularly for invasive disease. Antiviral drugs, such as ganciclovir, that target the replication of herpesviruses such as KSHV have been used to successfully prevent development of Kaposi's sarcoma, although once the tumor develops these drugs are of little or no use. For patients with AIDS-KS, the most effective therapy is highly active antiretroviral therapy to reduce HIV infection. AIDS patients receiving adequate anti-HIV treatment may have up to a 90% reduction in Kaposi's sarcoma occurrence.
References
- Chang Y, Cesarman E, Pessin M, et al. Identification of herpesvirus-like DNA sequences in AIDS-associated Kaposi's sarcoma. Science 1994;266:1865-9.
- Kaposi M. Idiopathic multiple pigmented sarcoma of the skin. Arch. Dermatol. Syphil. 1872; 4:265-73.
- Beral V, Peterman TA, Berkelman RL, Jaffe HW. Kaposi's sarcoma among persons with AIDS: a sexually transmitted infection? Lancet 1990; 335:123-128.
- Antman K, Chang Y. Kaposi's sarcoma. New Engl J Med 2000;342(14):1027-38.
- Moore PS, Chang Y. Detection of herpesvirus-like DNA sequences in Kaposi's sarcoma in patients with and without HIV infection. N Engl J Med 1995;332(18):1181-5.
- Cesarman E, Chang Y, Moore PS, Said JW, Knowles DM. Kaposi's sarcoma-associated herpesvirus-like DNA sequences in AIDS-related body-cavity-based lymphomas. N Engl J Med 1995;332(18):1186-91.
- Boshoff C, Weiss R. AIDS-related malignancies. Nat Rev Cancer 2002;2(5):373-82.
- Yarchoan R, Tosato G, Little RF. Therapy insight: AIDS-related malignancies - the influence of antiviral therapy on pathogenesis and management. Nature Clin Prac Oncology 2005;2(8):406-15.
- Edelman,D.C. Human herpesvirus 8--a novel human pathogen. Virol. J. 2, 78 (2005)
External link
- Human Herpesvirus-8: Related Resources HIV InSite
- ucsf.edu Image of KSHV episome, latency genes in green.