Homatropine
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Rabin Bista, M.B.B.S. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Homatropine is a anticholinergic agent that is FDA approved for the treatment of inflammatory conditions of the uveal tract, for cycloplegic refraction and as an optical aid in some cases of axial lens opacities. Common adverse reactions include stinging and burning sensation in eye.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- A moderately long-acting mydriatic and cycloplegic for cycloplegic refraction and in the treatment of inflammatory conditions of the uveal tract. For pre- and postoperative states when mydriasis is required. Use as an optical aid in some cases of axial lens opacities.
Dosage
- For refraction, instill one or two drops topically in the eye(s). May be repeated in five to ten minutes if necessary. For uveitis, instill one or two drops topically up to every three to four hours. Individuals with heavily pigmented irides may require larger doses. Only the 2% strength should be used in pediatric patients.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Homatropine in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Homatropine in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Homatropine in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Homatropine in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Homatropine in pediatric patients.
Contraindications
- Contraindicated in persons with primary glaucoma or a tendency toward glaucoma, e.g., narrow anterior chamber angle, and in those persons showing hypersensitivity to any component of this preparation.
Warnings
- FOR TOPICAL OPHTHALMIC USE ONLY - NOT FOR INJECTION. Risk-benefit should be considered when the following medical problems exist: keratoconus (homatropine may produce fixed dilated pupil); Down’s syndrome, children with brain damage and the elderly (increased susceptibility). In infants and small children, use with extreme caution.
Precautions
General
- To avoid excessive systemic absorption, the lacrimal sac should be compressed by digital pressure for two to three minutes after instillation. To avoid inducing angle closure glaucoma, an estimation of the depth of the angle of the anterior chamber should be made. Excessive topical use of this drug can potentially lead to a confusional state characterized by delirium, agitation, and rarely coma. This state is more apt to occur in the pediatric and geriatric age groups. The specific antidote for this systemic anticholinergic syndrome is injectable physostigmine salicylate.
Adverse Reactions
Clinical Trials Experience
- Transient symptoms of stinging and burning] may occur. Prolonged use may produce local irritation characterized by follicular conjunctivitis, vascular congestion, edema, exudate, and an eczematoid dermatitis. Thirst or dryness of mouth, eye irritation not present before therapy, or increased sensitivity of eyes to light may occur.
Overdose for Homatropine
- Systemic homatropine toxicity is manifested by flushing and dryness of the skin (a rash may be present in children), blurred vision, a rapid and irregular pulse, fever, abdominal distention in infants, mental aberration (hallucinosis) and loss of neuro-muscular coordination. Atropine poisoning, although distressing, is rarely fatal even with large doses of atropine, and is self-limited if the cause is recognized and the homatropine medication is discontinued. Treatment includes supportive measures including maintaining a patent airway and assisting respiration if needed. Treat hyperthermia, coma and seizures if they occur (1). In infants and children, the body surface must be kept moist. Excitement may be controlled by diazepam or a short-acting barbiturate. For ingestion, activated charcoal can be used to prevent drug absorption. If necessary, ipecac or another cathartic may be useful for drug removal during initial treatment (1, 2). Physostigmine is used as an antidote to the systemic effects of atropine and may be administered parenterally to provide more prompt relief of intoxication. Parenteral physostigmine may be particularly useful in cases of pronounced hallucinations, agitation in which a patient may be dangerous to himself or others, arrhythmias resulting in uncontrolled hemodynamic instability, and intractable seizures.
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Homatropine in the drug label.
Drug Interactions
There is limited information regarding Homatropine Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
- Animal reproduction studies have not been conducted with homatropine hydrobromide. It is also not known whether homatropine hydrobromide can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Homatropine hydrobromide should be given to a pregnant woman only if clearly needed.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Homatropine in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Homatropine during labor and delivery.
Nursing Mothers
- It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when homatropine hydrobromide is administered to a nursing woman.
Pediatric Use
- Safety and effectiveness in pediatric patients have not been established.
Geriatic Use
- No overall differences in safety or effectiveness have been observed between elderly and younger patients.
Gender
There is no FDA guidance on the use of Homatropine with respect to specific gender populations.
Race
There is no FDA guidance on the use of Homatropine with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Homatropine in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Homatropine in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Homatropine in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Homatropine in patients who are immunocompromised.
Administration and Monitoring
Administration
- topical eye drops
Monitoring
There is limited information regarding Monitoring of Homatropine in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Homatropine in the drug label.
Overdosage
Overdose for Homatropine
- Systemic homatropine toxicity is manifested by flushing and dryness of the skin (a rash may be present in children), blurred vision, a rapid and irregular pulse, fever, abdominal distention in infants, mental aberration (hallucinosis) and loss of neuro-muscular coordination. Atropine poisoning, although distressing, is rarely fatal even with large doses of atropine, and is self-limited if the cause is recognized and the homatropine medication is discontinued. Treatment includes supportive measures including maintaining a patent airway and assisting respiration if needed. Treat hyperthermia, coma and seizures if they occur (1). In infants and children, the body surface must be kept moist. Excitement may be controlled by diazepam or a short-acting barbiturate. For ingestion, activated charcoal can be used to prevent drug absorption. If necessary, ipecac or another cathartic may be useful for drug removal during initial treatment (1, 2). Physostigmine is used as an antidote to the systemic effects of atropine and may be administered parenterally to provide more prompt relief of intoxication. Parenteral physostigmine may be particularly useful in cases of pronounced hallucinations, agitation in which a patient may be dangerous to himself or others, arrhythmias resulting in uncontrolled hemodynamic instability, and intractable seizures.
Pharmacology
| |
Homatropine
| |
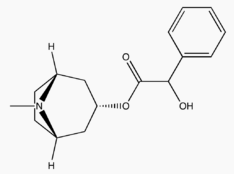
| Systematic (IUPAC) name | |
| (N-methyl-8-azabicyclo[3.2.1]oct-3-yl) 2-hydroxy-2-phenylacetate | |
| Identifiers | |
| CAS number | |
| ATC code | A02 S01FA05 (WHO) |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 356.26 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status |
rx only not FDA approved only sold in UK |
| Routes | ? |
Mechanism of Action
- This anticholinergic preparation blocks the responses of the sphincter muscle of the iris and the accommodative muscle of the ciliary body to cholinergic stimulation, producing pupillary dilation (mydriasis) and paralysis of accommodation (cycloplegia).
Structure
- ISOPTO® Homatropine (homatropine hydrobromide ophthalmic solution) is an anticholinergic prepared as a sterile topical ophthalmic solution supplied in two strengths. The active ingredient is represented by the chemical structure:
- Established name: Homatropine Hydrobromide
- Chemical name: Benzeneacetic acid, α-hydroxy-, 8-methyl-8-azabicyclo[3.2.1]-oct-3-yl ester, hydrobromide, endo-(±)-.
- Each mL contains: Active: homatropine hydrobromide 2.0% or 5.0%. Preservatives: benzalkonium chloride 0.01% in 2% strength, benzethonium chloride 0.005% in 5% strength. Vehicle: hypromellose 0.5%. Inactives: sodium chloride, polysorbate 80 (in 2% strength), sodium hydroxide and/or hydrochloric acid (to adjust pH), purified water.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Homatropine in the drug label.
Pharmacokinetics
There is limited information regarding Pharmacokinetics of Homatropine in the drug label.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
- There have been no long-term studies done using homatropine hydrobromide in animals to evaluate carcinogenic potential.
Clinical Studies
There is limited information regarding Clinical Studies of Homatropine in the drug label.
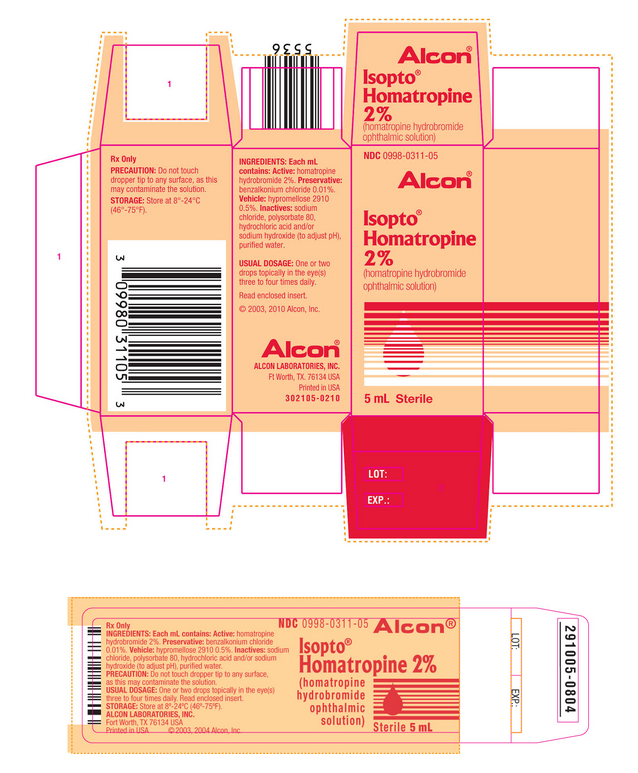
How Supplied
- 5 mL in plastic DROP-TAINER® Dispensers.
- 2% ISOPTO® Homatropine
- 5 mL NDC 0998-0311-05
- 5% ISOPTO® Homatropine
- 5 mL NDC 0998-0315-05
Storage
- Store at 8° - 24°C (46° - 75°F).
- Rx Only
Images
Drug Images
{{#ask: Page Name::Homatropine |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
PRINCIPAL DISPLAY PANEL
NDC 0998-0311-05
Alcon®
Isopto® Homatropine 2% (homatropine hydrobromide ophthalmic solution)
5 mL Sterile
Ingredients and Appearance
{{#ask: Label Page::Homatropine |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Patient should be advised not to drive or engage in other hazardous activities while pupils are dilated. Patient may experience sensitivity to light and should protect eyes in bright illumination during dilation. Parents should be warned not to get this preparation in their child’s mouth and to wash their own hands and the child’s hands following administration. Do not touch dropper tip to any surface, as this may contaminate the solution.
Precautions with Alcohol
- Alcohol-Homatropine interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- ISOPTO HOMATROPINE®[1]
Look-Alike Drug Names
There is limited information regarding Homatropine Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.