Halcinonide
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Rabin Bista, M.B.B.S. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Halcinonide is a topical corticosteroid that is FDA approved for the treatment of corticosteroid-responsive dermatoses. Common adverse reactions include Dry skin, Pruritus, Burning sensation, Irritation.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- HALOG (Halcinonide Cream, USP) 0.1% is indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses.
Dosage
- Apply the 0.1% HALOG (Halcinonide Cream, USP) to the affected area two to three times daily. Rub in gently.
Occlusive Dressing Technique
- Occlusive dressings may be used for the management of psoriasis or other recalcitrant conditions. Gently rub a small amount of cream into the lesion until it disappears. Reapply the preparation leaving a thin coating on the lesion, cover with a pliable nonporous film, and seal the edges. If needed, additional moisture may be provided by covering the lesion with a dampened clean cotton cloth before the nonporous film is applied or by briefly wetting the affected area with water immediately prior to applying the medication. The frequency of changing dressings is best determined on an individual basis. It may be convenient to apply HALOG under an occlusive dressing in the evening and to remove the dressing in the morning (i.e., 12-hour occlusion). When utilizing the 12-hour occlusion regimen, additional cream should be applied, without occlusion, during the day. Reapplication is essential at each dressing change.
- If an infection develops, the use of occlusive dressings should be discontinued and appropriate antimicrobial therapy instituted.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Halcinonide in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Halcinonide in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Halcinonide in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Halcinonide in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Halcinonide in pediatric patients.
Contraindications
- Topical corticosteroids are contraindicated in those patients with a history of hypersensitivity to any of the components of the preparations.
Warnings
Precautions
General
- Systemic absorption of topical corticosteroids has produced reversible hypothalamic-pituitary-adrenal (HPA) axis suppression, manifestations of Cushing’s syndrome, hyperglycemia, and glucosuria in some patients.
- Conditions which augment systemic absorption include the application of the more potent steroids, use over large surface areas, prolonged use, and the addition of occlusive dressings.
- Therefore, patients receiving a large dose of any potent topical steroid applied to a large surface area or under an occlusive dressing should be evaluated periodically for evidence of HPA axis suppression by using the urinary free cortisol and ACTH stimulation tests, and for impairment of thermal homeostasis. If HPA axis suppression or elevation of the body temperature occurs, an attempt should be made to withdraw the drug, to reduce the frequency of application, substitute a less potent steroid, or use a sequential approach when utilizing the occlusive technique.
- Recovery of HPA axis function and thermal homeostasis are generally prompt and complete upon discontinuation of the drug. Infrequently, signs and symptoms of steroid withdrawal may occur, requiring supplemental systemic corticosteroids. Occasionally, a patient may develop a sensitivity reaction to a particular occlusive dressing material or adhesive and a substitute material may be necessary.
- Children may absorb proportionally larger amounts of topical corticosteroids and thus be more susceptible to systemic toxicity.
- If irritation develops, topical corticosteroids should be discontinued and appropriate therapy instituted.
- In the presence of dermatological infections, the use of an appropriate antifungal or antibacterial agent should be instituted. If a favorable response does not occur promptly, the corticosteroid should be discontinued until the infection has been adequately controlled.
- This preparation is not for ophthalmic, oral, or intravaginal use.
Laboratory Tests
- A urinary free cortisol test and ACTH stimulation test may be helpful in evaluating HPA axis suppression.
Adverse Reactions
Clinical Trials Experience
- The following local adverse reactions are reported infrequently with topical corticosteroids, but may occur more frequently with the use of occlusive dressings (reactions are listed in an approximate decreasing order of occurrence): burning, itching, irritation, dryness, folliculitis, hypertrichosis, acneiform eruptions, hypopigmentation, perioral dermatitis, Allergic contact dermatitis, maceration of the skin, Secondary infection, skin atrophy, striae, and miliaria.
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Halcinonide in the drug label.
Drug Interactions
There is limited information regarding Halcinonide Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
- Corticosteroids are generally teratogenic in laboratory animals when administered systemically at relatively low dosage levels. The more potent corticosteroids have been shown to be teratogenic after dermal application in laboratory animals. There are no adequate and well-controlled studies in pregnant women on teratogenic effects from topically applied corticosteroids. Therefore, topical corticosteroids should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Drugs of this class should not be used extensively on pregnant patients, in large amounts, or for prolonged periods of time.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Halcinonide in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Halcinonide during labor and delivery.
Nursing Mothers
- It is not known whether topical administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in breast milk. Systemically administered corticosteroids are secreted into breast milk in quantities not likely to have a deleterious effect on the infant. Nevertheless, caution should be exercised when topical corticosteroids are administered to a nursing woman.
Pediatric Use
- Pediatric patients may demonstrate greater susceptibility to topical corticosteroid-induced HPA axis suppression and Cushing’s syndrome than mature patients because of a larger skin surface area to body weight ratio.
- HPA axis suppression, Cushing’s syndrome, and intracranial hypertension have been reported in children receiving topical corticosteroids. Manifestations of adrenal suppression in children include linear Growth retardation, delayed weight gain, low plasma cortisol levels, and absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches, and bilateral papilledema.
- Administration of topical corticosteroids to children should be limited to the least amount compatible with an effective therapeutic regimen. Chronic corticosteroid therapy may interfere with the growth and development of children.
Geriatic Use
- Of approximately 3000 patients included in clinical studies of 0.1% HALOG CREAM, 14% were 60 years or older, while 4% were 70 years or older. No overall differences in safety were observed between these patients and younger patients. Efficacy data have not been evaluated for differences between elderly and younger patients. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Gender
There is no FDA guidance on the use of Halcinonide with respect to specific gender populations.
Race
There is no FDA guidance on the use of Halcinonide with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Halcinonide in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Halcinonide in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Halcinonide in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Halcinonide in patients who are immunocompromised.
Administration and Monitoring
Administration
- topical
Monitoring
- patients receiving a large dose of any potent topical steroid applied to a large surface area or under an occlusive dressing should be evaluated periodically for evidence of HPA axis suppression by using the urinary free cortisol and ACTH stimulation tests, and for impairment of thermal homeostasis.
IV Compatibility
There is limited information regarding IV Compatibility of Halcinonide in the drug label.
Overdosage
- Topically applied corticosteroids can be absorbed in sufficient amounts to produce systemic effects
Pharmacology
| |
Halcinonide
| |
| Systematic (IUPAC) name | |
| (4aS,4bR,5S,6aS,6bS,9aR,10aS,10bS)-6b-(chloroacetyl)-4b-fluoro-5-hydroxy-4a,6a,8,8-tetramethyl-3,4,4a,4b,5,6,6a,6b,9a,10,10a,10b,11,12-tetradecahydro-2H-naphtho[2',1':4,5]indeno[1,2-d][1,3]dioxol-2-one | |
| Identifiers | |
| CAS number | |
| ATC code | D07 |
| PubChem | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 454.959 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | Topical |
Mechanism of Action
- Topical corticosteroids share anti-inflammatory, antipruritic and vasoconstrictive actions.
- The mechanism of anti-inflammatory activity of the topical corticosteroids is unclear. Various laboratory methods, including vasoconstrictor assays, are used to compare and predict potencies and/or clinical efficacies of the topical corticosteroids. There is some evidence to suggest that a recognizable correlation exists between vasoconstrictor potency and therapeutic efficacy in man.
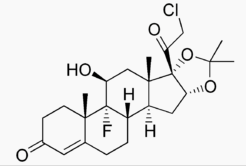
Structure
- The topical corticosteroids constitute a class of primarily synthetic steroids used as anti-inflammatory and antipruritic agents. The steroids in this class include halcinonide. Halcinonide is designated chemically as 21-Chloro-9-fluoro-11β,16α, 17-trihydroxypregn-4-ene-3,20-dione cyclic 16,17-acetal with acetone. Graphic formula:
- Each gram of 0.1% HALOG (Halcinonide Cream, USP) contains 1 mg halcinonide, USP in a specially formulated cream base consisting of cetyl alcohol, dimethicone 350, glyceryl monostearate , isopropyl palmitate, polysorbate 60, propylene glycol, purified water, and titanium dioxide.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Halcinonide in the drug label.
Pharmacokinetics
- The extent of percutaneous absorption of topical corticosteroids is determined by many factors including the vehicle, the integrity of the epidermal barrier, and the use of occlusive dressings.
- Topical corticosteroids can be absorbed from normal intact skin. Inflammation and/or other disease processes in the skin increase percutaneous absorption. Occlusive dressings substantially increase the percutaneous absorption of topical corticosteroids. Thus, occlusive dressings may be a valuable therapeutic adjunct for treatment of resistant dermatoses.
- Once absorbed through the skin, topical corticosteroids are handled through pharmacokinetic pathways similar to systemically administered corticosteroids. Corticosteroids are bound to plasma proteins in varying degrees. Corticosteroids are metabolized primarily in the liver and are then excreted by the kidneys. Some of the topical corticosteroids and their metabolites are also excreted into the bile.
Nonclinical Toxicology
Carcinogenesis and Mutagenesis and Impairment of Fertility
- Long-term animal studies have not been performed to evaluate the carcinogenic potential or the effect on fertility of topical corticosteroids.
- Studies to determine mutagenicity with prednisolone and hydrocortisone showed negative results.
Clinical Studies
There is limited information regarding Clinical Studies of Halcinonide in the drug label.
How Supplied
- HALOG ® (Halcinonide Cream, USP) 0.1% is smooth, soft homogeneous white to off-white cream, essentially free of foreign matter and is supplied as:
- NDC 10631-094-20 Tubes containing 30 g
- NDC 10631-094-30 Tubes containing 60 g
- NDC 10631-094-76 Jars containing 216 g
Storage
- Store at room temperature; avoid excessive heat (104° F).
- To report SUSPECTED ADVERSE REACTIONS, contact the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
- RANBAXY
- Jacksonville, FL 32257 USA
- 140025
- Revised March 2013
Images
Drug Images
{{#ask: Page Name::Halcinonide |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Ingredients and Appearance
{{#ask: Label Page::Halcinonide |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Patients using topical corticosteroids should receive the following information and instructions:
1. This medication is to be used as directed by the physician. It is for dermatologic use only. Avoid contact with the eyes.
2. Patients should be advised not to use this medication for any disorder other than for which it was prescribed.
3. The treated skin area should not be bandaged or otherwise covered or wrapped as to be occlusive unless directed by the physician.
4. Patients should report any signs of local adverse reactions especially under occlusive dressing.
5. Parents of pediatric patients should be advised not to use tight-fitting diapers or plastic pants on a child being treated in the diaper area, as these garments may constitute occlusive dressings.
Precautions with Alcohol
- Alcohol-Halcinonide interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Halog®[1]
Look-Alike Drug Names
There is limited information regarding Halcinonide Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.