Gepirone
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | 2-3 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C19H29N5O2 |
| Molar mass | 359.46586 g/mol |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
|
WikiDoc Resources for Gepirone |
|
Articles |
|---|
|
Most recent articles on Gepirone |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Gepirone at Clinical Trials.gov Clinical Trials on Gepirone at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Gepirone
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Directions to Hospitals Treating Gepirone Risk calculators and risk factors for Gepirone
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Gepirone |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Gepirone (Ariza, Variza; BMY-13,805, ORG-13,011) is an antidepressant and anxiolytic of the azapirone class. Like other azapirones, it acts as a selective partial agonist of the 5-HT1A receptor. Gepirone was under development in the U.S. in an extended release form, but despite completing phase III clinical trials, it failed to pass the drug approval process. As a result, gepirone, despite already having brand names picked out, did not manage to reach the pharmaceutical market.
Gepirone was originally developed by Bristol-Myers Squibb, but was out-licensed to Fabre-Kramer in 1993. The U.S. Food and Drug Administration (FDA) rejected approval for gepirone in 2004. It was submitted for the preregistration (NDA) phase again in May 2007 after adding additional information from clinical trials as the FDA required in 2004. However, in 2007 it once again failed to convince the FDA of its qualities for treating anxiety and depression. There have been no updates on gepirone since.
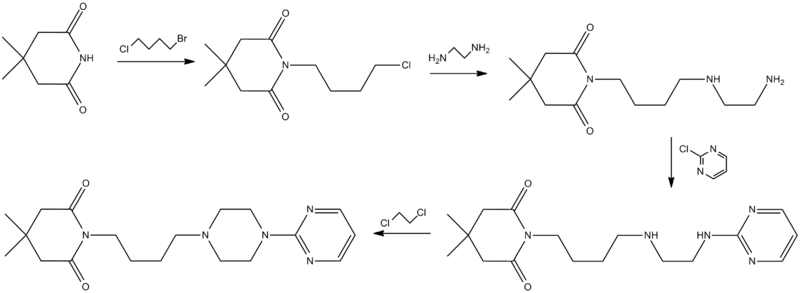
Synthesis
Ormaza, V. A.; 1986, ES 8606333.