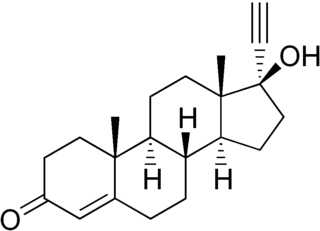
Ethisterone
 | |
 | |
| Clinical data | |
|---|---|
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C21H28O2 |
| Molar mass | 312.446 g/mol |
| 3D model (JSmol) | |
| |
| |
| | |
|
WikiDoc Resources for Ethisterone |
|
Articles |
|---|
|
Most recent articles on Ethisterone Most cited articles on Ethisterone |
|
Media |
|
Powerpoint slides on Ethisterone |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Ethisterone at Clinical Trials.gov Clinical Trials on Ethisterone at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Ethisterone
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Ethisterone Discussion groups on Ethisterone Patient Handouts on Ethisterone Directions to Hospitals Treating Ethisterone Risk calculators and risk factors for Ethisterone
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Ethisterone |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Ethisterone (pregneninolone, 17α-ethynyltestosterone or 19–norandrostane) is a progestin. It is the 17α-ethynyl analog of testosterone, and was synthesized in 1938 by Hans Herloff Inhoffen, Willy Logemann, Walter Hohlweg, and Arthur Serini at Schering AG in Berlin and marketed in Germany in 1939 as Proluton C and by Schering in the U.S. in 1945 as Pranone. It was the first orally-active progestin.
Ethisterone was also marketed in the U.S. from the 1950s into the 1960s under a variety of trade names by other pharmaceutical companies that had been members of the pre-World War II European hormone cartel (Ciba, Organon, Roussel).
Synthesis
Ethisterone is made by the ethynylation of androstenolone with acetylene and successive oxidation of the hydroxyl group at C3 of the steroid system.
References
- Pages with script errors
- Articles with changed CASNo identifier
- Articles with changed EBI identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Drugs with no legal status
- Drugboxes which contain changes to verified fields
- Drug