Ethionamide
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Kiran Singh, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Ethionamide is an antitubercular that is FDA approved for the treatment of tuberculosis. Common adverse reactions include abdominal pain, diarrhea, metallic taste, nausea, stomatitis, vomiting.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- Trecator is primarily indicated for the treatment of active tuberculosis in patients with M. tuberculosis resistant to isoniazid or rifampin, or when there is intolerance on the part of the patient to other drugs. Its use alone in the treatment of tuberculosis results in the rapid development of resistance. It is essential, therefore, to give a suitable companion drug or drugs, the choice being based on the results of susceptibility tests. If the susceptibility tests indicate that the patient's organism is resistant to one of the first-line antituberculosis drugs (i.e., isoniazid or rifampin) yet susceptible to ethionamide, ethionamide should be accompanied by at least one drug to which the M. tuberculosis isolate is known to be susceptible.3 If the tuberculosis is resistant to both isoniazid and rifampin, yet susceptible to ethionamide, ethionamide should be accompanied by at least two other drugs to which the M. tuberculosis isolate is known to be susceptible.3
- Patient nonadherence to prescribed treatment can result in treatment failure and in the development of drug-resistant tuberculosis, which can be life-threatening and lead to other serious health risks. It is, therefore, essential that patients adhere to the drug regimen for the full duration of treatment. Directly observed therapy is recommended for all patients receiving treatment for tuberculosis. Patients in whom drug-resistant M. tuberculosis organisms are isolated should be managed in consultation with an expert in the treatment of drug-resistant tuberculosis.
Dosage
In the treatment of tuberculosis, a major cause of the emergence of drug-resistant organisms, and thus treatment failure, is patient nonadherence to prescribed treatment. Treatment failure and drug-resistant organisms can be life-threatening and may result in other serious health risks. It is, therefore, important that patients adhere to the drug regimen for the full duration of treatment. Directly observed therapy is recommended when patients are receiving treatment for tuberculosis. Consultation with an expert in the treatment of drug-resistant tuberculosis is advised for patients in whom drug-resistant tuberculosis is suspected or likely. Ethionamide should be administered with at least one, sometimes two, other drugs to which the organism is known to be susceptible (see INDICATIONS AND USAGE).
- Trecator is administered orally. The usual adult dose is 15 to 20 mg/kg/day, administered once daily or, if patient exhibits poor gastrointestinal tolerance, in divided doses, with a maximum daily dosage of 1 gram.
- Trecator tablets have been reformulated from a sugar-coated tablet to a film-coated tablet. Patients should be monitored and have their dosage retitrated when switching from the sugar-coated tablet to the film-coated tablet.
- Therapy should be initiated at a dose of 250 mg daily, with gradual titration to optimal doses as tolerated by the patient. A regimen of 250 mg daily for 1 or 2 days, followed by 250 mg twice daily for 1 or 2 days with a subsequent increase to 1 gm in 3 or 4 divided doses has been reported.4,5 Thus far, there is insufficient evidence to indicate the lowest effective dosage levels. Therefore, in order to minimize the risk of resistance developing to the drug or to the companion drug, the principle of giving the highest tolerated dose (based on gastrointestinal intolerance) has been followed. In the adult this would seem to be between 0.5 and 1.0 gm daily, with an average of 0.75 gm daily.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Ethionamide in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Ethionamide in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Indications For Pediatric,12 Yrs And older
- Trecator is primarily indicated for the treatment of active tuberculosis in patients with M. tuberculosis resistant to isoniazid or rifampin, or when there is intolerance on the part of the patient to other drugs. Its use alone in the treatment of tuberculosis results in the rapid development of resistance. It is essential, therefore, to give a suitable companion drug or drugs, the choice being based on the results of susceptibility tests. If the susceptibility tests indicate that the patient's organism is resistant to one of the first-line antituberculosis drugs (i.e., isoniazid or rifampin) yet susceptible to ethionamide, ethionamide should be accompanied by at least one drug to which the M. tuberculosis isolate is known to be susceptible.3 If the tuberculosis is resistant to both isoniazid and rifampin, yet susceptible to ethionamide, ethionamide should be accompanied by at least two other drugs to which the M. tuberculosis isolate is known to be susceptible.3
- Patient nonadherence to prescribed treatment can result in treatment failure and in the development of drug-resistant tuberculosis, which can be life-threatening and lead to other serious health risks. It is, therefore, essential that patients adhere to the drug regimen for the full duration of treatment. Directly observed therapy is recommended for all patients receiving treatment for tuberculosis. Patients in whom drug-resistant M. tuberculosis organisms are isolated should be managed in consultation with an expert in the treatment of drug-resistant tuberculosis.
Dosage For Pediatrics 12 Yrs And Older
The optimum dosage for pediatric patients has not been established. However, pediatric dosages of 10 to 20 mg/kg p.o. daily in 2 or 3 divided doses given after meals or 15 mg/kg/24 hrs as a single daily dose have been recommended.1,2 As with adults, ethionamide may be administered to pediatric patients once daily. It should be noted that in patients with concomitant tuberculosis and HIV infection, malabsorption syndrome may be present. Drug malabsorption should be suspected in patients who adhere to therapy, but who fail to respond appropriately. In such cases, consideration should be given to therapeutic drug monitoring.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Ethionamide in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Ethionamide in pediatric patients.
Contraindications
- Ethionamide is contraindicated in patients with severe hepatic impairment and in patients who are hypersensitive to the drug.
Warnings
- The use of Trecator alone in the treatment of tuberculosis results in rapid development of resistance. It is essential, therefore, to give a suitable companion drug or drugs, the choice being based on the results of susceptibility testing. However, therapy may be initiated prior to receiving the results of susceptibility tests as deemed appropriate by the physician. Ethionamide should be administered with at least one, sometimes two, other drugs to which the organism is known to be susceptible. Drugs which have been used as companion agents are rifampin, ethambutol, pyrazinamide, cycloserine, kanamycin, streptomycin, and isoniazid. The usual warnings, precautions, and dosage regimens for these companion drugs should be observed.
- Patient compliance is essential to the success of the antituberculosis therapy and to prevent the emergence of drug-resistant organisms. Therefore, patients should adhere to the drug regimen for the full duration of treatment. It is recommended that directly observed therapy be practiced when patients are receiving antituberculous medication. Additional consultation from experts in the treatment of drug-resistant tuberculosis is recommended when patients develop drug-resistant organisms.
Adverse Reactions
Clinical Trials Experience
Gastrointestinal: The most common side effects of ethionamide are gastrointestinal disturbances including nausea, vomiting, diarrhea, abdominal pain, excessive salivation, metallic taste, stomatitis, anorexia and weight loss. Adverse gastrointestinal effects appear to be dose related, with approximately 50% of patients unable to tolerate 1 gm as a single dose. Gastrointestinal effects may be minimized by decreasing dosage, by changing the time of drug administration, or by the concurrent administration of an antiemetic agent.
Nervous System: Psychotic disturbances (including mental depression), drowsiness, dizziness, restlessness, headache, and postural hypotension have been reported with ethionamide. Rare reports of peripheral neuritis, optic neuritis, diplopia, blurred vision, and a pellagra-like syndrome also have been reported. Concurrent administration of pyridoxine has been recommended to prevent or relieve neurotoxic effects.
Hepatic: Transient increases in serum bilirubin, SGOT, SGPT; Hepatitis (with or without jaundice).
Other: Hypersensitivity reactions including rash, photosensitivity, thrombocytopenia and purpura have been reported rarely. Hypoglycemia, hypothyroidism, gynecomastia, impotence, and acne also have occurred. The management of patients with diabetes mellitus may become more difficult in those receiving ethionamide.
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Ethionamide in the drug label.
Drug Interactions
- Trecator has been found to temporarily raise serum concentrations of isoniazid. Trecator may potentiate the adverse effects of other antituberculous drugs administered concomitantly. In particular, convulsions have been reported when ethionamide is administered with cycloserine and special care should be taken when the treatment regimen includes both of these drugs. Excessive ethanol ingestion should be avoided because a psychotic reaction has been reported.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): Teratogenic Effects: Pregnancy Category C
- Animal studies conducted with Trecator indicate that the drug has teratogenic potential in rabbits and rats. The doses used in these studies on a mg/kg basis were considerably in excess of those recommended in humans. There are no adequate and well-controlled studies in pregnant women. Because of these animal studies, however, it must be recommended that Trecator be withheld from women who are pregnant, or who are likely to become pregnant while under therapy, unless the prescribing physician considers it to be an essential part of the treatment.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Ethionamide in women who are pregnant.
Labor and Delivery
- The effect of Trecator on labor and delivery in pregnant women is unknown.
Nursing Mothers
- Because no information is available on the excretion of ethionamide in human milk, Trecator should be administered to nursing mothers only if the benefits outweigh the risks. Newborns who are breast-fed by mothers who are taking Trecator should be monitored for adverse effects.
Pediatric Use
- Due to the fact that pulmonary tuberculosis resistant to primary therapy is rarely found in neonates, infants, and children, investigations have been limited in these age groups. At present, the drug should not be used in pediatric patients under 12 years of age except when the organisms are definitely resistant to primary therapy and systemic dissemination of the disease, or other life-threatening complications of tuberculosis, is judged to be imminent.
Geriatic Use
There is no FDA guidance on the use of Ethionamide with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Ethionamide with respect to specific gender populations.
Race
There is no FDA guidance on the use of Ethionamide with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Ethionamide in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Ethionamide in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Ethionamide in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Ethionamide in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
Monitoring
There is limited information regarding Monitoring of Ethionamide in the drug
IV Compatibility
There is limited information regarding IV Compatibility of Ethionamide in the drug label.
Overdosage
- No specific information is available on the treatment of overdosage with Trecator. If it should occur, standard procedures to evacuate gastric contents and to support vital functions should be employed.
Pharmacology
Mechanism of Action
- Ethionamide may be bacteriostatic or bactericidal in action, depending on the concentration of the drug attained at the site of infection and the susceptibility of the infecting organism. The exact mechanism of action of ethionamide has not been fully elucidated, but the drug appears to inhibit peptide synthesis in susceptible organisms.
Structure
- Trecator® (ethionamide tablets, USP) is used in the treatment of tuberculosis. The chemical name for ethionamide is 2-ethylthioisonicotinamide with the following structural formula:
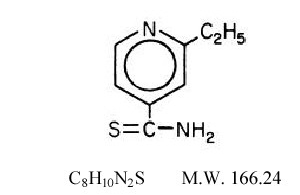
- Ethionamide is a yellow crystalline, nonhygroscopic compound with a faint to moderate sulfide odor and a melting point of 162°C. It is practically insoluble in water and ether, but soluble in methanol and ethanol. It has a partition coefficient (octanol/water) Log P value of 0.3699. Trecator tablets contain 250 mg of ethionamide. The inactive ingredients present are croscarmellose sodium, FD&C Yellow #6, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, silicon dioxide, talc, and titanium dioxide.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Ethionamide in the drug label.
Pharmacokinetics
Absorption
- Ethionamide is essentially completely absorbed following oral administration and is not subjected to any appreciable first pass metabolism. Ethionamide tablets may be administered without regard to the timing of meals.
- The pharmacokinetic parameters of ethionamide following single oral-dose administration of 250 mg of Trecator film-coated tablets under fasted conditions to 40 healthy adult volunteers are provided below.
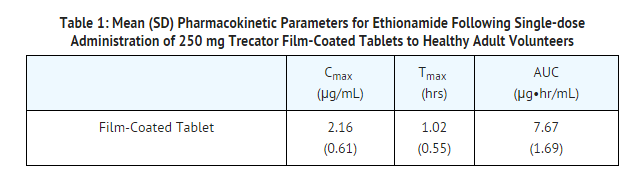
- Trecator tablets have been reformulated from a sugar-coated tablet to a film-coated tablet. The Cmax for the film-coated tablets (2.16 µg/mL) was significantly higher than that of sugar-coated tablets (1.48 µg/mL).
Distribution
- Ethionamide is rapidly and widely distributed into body tissues and fluids following administration of a sugar-coated tablet, with concentrations in plasma and various organs being approximately equal. Significant concentrations are also present in cerebrospinal fluid following administration of a sugar-coated tablet. Distribution of ethionamide into the same body tissues and fluids, including cerebrospinal fluid following administration of the film-coated tablet, has not been studied, but is not expected to differ significantly from that of the sugar-coated tablet. The drug is approximately 30% bound to proteins. The mean (SD) apparent oral volume of distribution observed in 40 healthy volunteers following a 250 mg oral dose of film-coated tablets was 93.5 (19.2) L.
Metabolism
- Ethionamide is extensively metabolized to active and inactive metabolites. Metabolism is presumed to occur in the liver and thus far 6 metabolites have been isolated: 2-ethylisonicotinamide, carbonyl-dihydropyridine, thiocarbonyl-dihydropyridine, S-oxocarbamoyl dihydropyridine, 2-ethylthioiso-nicotinamide, and ethionamide sulphoxide. The sulphoxide metabolite has been demonstrated to have antimicrobial activity against Mycobacterium tuberculosis.
Elimination
- The mean (SD) half-life observed in 40 healthy volunteers following a 250 mg oral dose of film-coated tablets was 1.92 (0.27) hours. Less than 1% of the oral dose is excreted as ethionamide in urine.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Ethionamide in the drug label.
Clinical Studies
There is limited information regarding Clinical Studies of Ethionamide in the drug label.
How Supplied
- Trecator® (ethionamide tablets, USP) are supplied in bottles of 100 tablets as follows:
- 250 mg, orange film-coated tablet marked "W" on one side and "4117" on reverse side, NDC 0008-4117-01.
Storage
- Store at controlled room temperature 20° to 25°C (68° to 77°F). Dispense in a tight container.
Images
Drug Images
{{#ask: Page Name::Ethionamide |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Ethionamide |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Patients should be advised to consult their physician should blurred vision or any loss of vision, with or without eye pain, occur during treatment.
- Excessive ethanol ingestion should be avoided because a psychotic reaction has been reported.
Precautions with Alcohol
- Alcohol-Ethionamide interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- TRECATOR
Look-Alike Drug Names
There is limited information regarding Ethionamide Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Page Name=Ethionamide
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Ethionamide |Label Name=Ethionamide image.jpg
}}
{{#subobject:
|Label Page=Ethionamide |Label Name=Ethionamide ingredients and appearance.png
}}