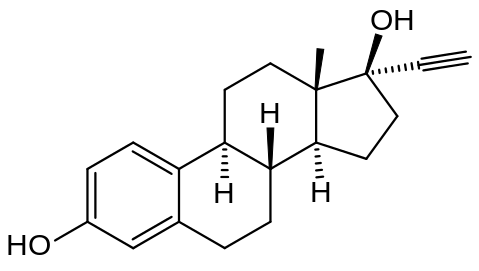
Ethinylestradiol
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| MedlinePlus | a604032 |
| Pregnancy category |
|
| Routes of administration | Oral, transdermal |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 97% is bound |
| Metabolism | Liver |
| Elimination half-life | 36 ± 13 hours |
| Excretion | Feces and Urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C20H24O2 |
| Molar mass | 296.403 g/mol |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
|
WikiDoc Resources for Ethinylestradiol |
|
Articles |
|---|
|
Most recent articles on Ethinylestradiol Most cited articles on Ethinylestradiol |
|
Media |
|
Powerpoint slides on Ethinylestradiol |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Ethinylestradiol at Clinical Trials.gov Trial results on Ethinylestradiol Clinical Trials on Ethinylestradiol at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Ethinylestradiol NICE Guidance on Ethinylestradiol
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Ethinylestradiol Discussion groups on Ethinylestradiol Patient Handouts on Ethinylestradiol Directions to Hospitals Treating Ethinylestradiol Risk calculators and risk factors for Ethinylestradiol
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Ethinylestradiol |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Ethinyl estradiol (EE2) /ˌɛθ[invalid input: 'ɨ']n[invalid input: 'ɨ']lˌiːstrəˈdaɪ.əl/, also sometimes written as 17α-ethinyl estradiol, ethinylestradiol, ethynyl estradiol, or ethinyl œstradiol, is a derivative of 17β-estradiol (E2), the major endogenous estrogen in humans. EE2 is an orally bioactive estrogen used in many formulations of combined oral contraceptive pills. It is one of the most commonly used medications for this purpose.
Transdermal ethinyl estradiol carries a greater risk of clot formation and venous thromboembolism than naturally occurring 17β-estradiol, which some have theorized to be related to different amounts of hepatic metabolism after absorption. The same contraindications and precautions apply for EE2 as with other estrogen medications.
Estinyl was a preparation of EE2 alone that was used for the management of menopausal symptoms and female hypogonadism.[1]
EE2 is released into the environment as a xenoestrogen from the urine and feces of people who take it as a medication.
The major concern with unopposed estrogen (without progestogen) is of endometrial cancer.[citation needed] As such, the medication is generally prescribed with progesterone in the setting of birth control.
History
The first orally active semisynthetic steroidal estrogen, EE2 (17α-ethynylestradiol), the 17α-ethynyl analog of E2, was synthesized in 1938 by Hans Herloff Inhoffen and Walter Hohlweg at Schering AG in Berlin.[2][3][4][5][6]
EE2 was approved by the FDA in the U.S. on June 25, 1943 and marketed by Schering as Estinyl.[7]
The FDA withdrew approval of Estinyl effective June 4, 2004 at the request of Schering, who had discontinued marketing Estinyl.[8]
Pharmacology
While E2 is readily absorbed when taken orally, it is also quickly inactivated by the liver. Substitution at C17 of the estrane steroid with an ethinyl group served to provide an estrogen that is much more resistant to degradation and paved the way for the development of oral contraceptives.
EE2 is absorbed in the small intestine and reaches a serum peak about 2 hours later. It undergoes extensive metabolism in the liver involving the cytochrome P450 3A4 isoenzyme. EE2 and its metabolites are excreted with the bile. Due to the effect of enterohepatic circulation a second peak is seen several hours later. Individually, wide variations exist in the overall absorption process, and can be further modified by drugs (i.e. antibiotics) that affect the enterohepatic circulation or liver enzymes. In circulation EE2 is almost fully bound to plasma albumin. It is metabolized by hydroxylation of the aromatic ring and excreted in both feces and urine, in part as glucuronide and sulfate conjugate.
EE2 is hormonally effective by activating the estrogen receptor and thus is an estrogen. It finds its most common use in the estrogen-progestin combination preparations of oral contraceptives. Over time, formulations have decreased the EE2 dose from as high as 100 μg to as low as 10 μg in LoLoestrin Fe.[9]
See also
- Mestranol
- Estrogen
- Hormonal contraception
- Oral contraceptive formulations
- Hormone replacement therapy
- Diethylstilbestrol
- Estradiol
References
- ↑ RxList.com - Estinyl (ethynyl estradiol)
- ↑ Inhoffen, H. H.; Hohlweg, W. (1938). "Neue per os-wirksame weibliche Keimdrüsenhormon-Derivate: 17-Aethinyl-oestradiol und Pregnen-in-on-3-ol-17 (New female glandular derivatives active per os: 17α-ethynyl-estradiol and pregnen-in-on-3-ol-17)". Naturwissenschaften. 26 (6): 96. doi:10.1007/BF01681040.
- ↑ Maisel, Albert Q. (1965). The Hormone Quest. New York: Random House. OCLC 543168.
- ↑ Petrow, Vladimir (December 1970). "The contraceptive progestagens". Chem Rev. 70 (6): 713–26. doi:10.1021/cr60268a004. PMID 4098492.
- ↑ Sneader, Walter (2005). "Hormone analogues". Drug discovery : a history. Hoboken, NJ: John Wiley & Sons. pp. 188–225. ISBN 0-471-89980-1.
- ↑ Djerassi, Carl (January 2006). "Chemical birth of the pill". American Journal of Obstetrics and Gynecology. 194 (1): 290–8. doi:10.1016/j.ajog.2005.06.010. PMID 16389046.
- ↑ FDA (2007). "Drug details: Estinyl (ethinyl estradiol) NDA 005292". search: Estinyl
- ↑ FDA (May 5, 2004). "Schering Corp. et al.; Withdrawal of Approval of 92 New Drug Applications and 49 Abbreviated New Drug Applications. Notice" (PDF). Federal Register. 69 (87): 25124–30.
- ↑ "LoLoestrin Fe website". Warner Chilcott. Retrieved 15 October 2011.
External links
- Pages with script errors
- Template:drugs.com link with non-standard subpage
- Drugs with non-standard legal status
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Ill-formatted IPAc-en transclusions
- All articles with unsourced statements
- Articles with unsourced statements from November 2014
- Articles with invalid date parameter in template
- Alkynes
- Drug
- Hormonal contraception
- Steroids
- Synthetic estrogens