Epinephrine (aerosol)
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Adeel Jamil, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Epinephrine (aerosol) is an adrenergic, sympathomimetic and bronchodilator agent that is FDA approved for the treatment of for temporary relief of shortness of breath, tightness of chest and wheezing due to bronchial asthma. Common adverse reactions include nausea, vomiting, dizziness, headache, anxiety, sweating and palpitation.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
- For temporary relief of shortness of breath, tightness of chest and wheezing due to bronchial asthma. Eases breathing for asthma patients by reducing spasms of bronchial muscles. For the symptomatic control of bronchial asthma.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Epinephrine (aerosol) in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Epinephrine (aerosol) in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding Epinephrine (aerosol) FDA-Labeled Indications and Dosage (Pediatric) in the drug label.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Epinephrine (aerosol) in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Epinephrine (aerosol) in pediatric patients.
Contraindications
There is limited information regarding Epinephrine (aerosol) Contraindications in the drug label.
Warnings
- Contains dichlorodifluoromethane, dichlorotetrafluoroethane and trichloromonofluoromethane, substances which harm public health and environment by destroying ozone in the upper atmosphere.
- For oral inhalation therapy only
- Do not use this product unless a diagnosis of asthma has been made by a physician. Do not use this product if you have heart disease, high blood pressure, thyroid disease, diabetes or difficulty in urination due to enlargement of the prostate gland, unless directed by a physician. Do not use this product if you have ever been hospitalized for asthma or if you are taking any prescription drug for asthma unless directed by a physician.
- MEDIHALER-EPI is intended for oral inhalation only. Do not use this product more frequently or at higher doses than recommended unless directed by a physician. Excessive use may cause nervousness and rapid heart beat, and, possibly adverse effects on the heart. Do not continue to use this product, but seek medical assistance immediately if symptoms are not relieved within 20 minutes or become worse.
- As with any drug, if you are pregnant or nursing a baby, seek the advise of a health professional before using this product. Keep this and all drugs out of the reach of children. In case of accidental overdose, seek professional assistance or contact a poison control center immediately.
Adverse Reactions
Clinical Trials Experience
- Nausea, vomiting, dizziness, headache, anxiety, sweating and palpitation
Postmarketing Experience
There is limited information regarding Epinephrine (aerosol) Postmarketing Experience in the drug label.
Drug Interactions
- Do not use this product if you are presently taking a prescription drug for high blood pressure or depression without first consulting your physician.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA):
There is no FDA guidance on usage of Epinephrine (aerosol) in women who are pregnant.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Epinephrine (aerosol) in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Epinephrine (aerosol) during labor and delivery.
Nursing Mothers
There is no FDA guidance on the use of Epinephrine (aerosol) in women who are nursing.
Pediatric Use
There is no FDA guidance on the use of Epinephrine (aerosol) in pediatric settings.
Geriatic Use
There is no FDA guidance on the use of Epinephrine (aerosol) in geriatric settings.
Gender
There is no FDA guidance on the use of Epinephrine (aerosol) with respect to specific gender populations.
Race
There is no FDA guidance on the use of Epinephrine (aerosol) with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Epinephrine (aerosol) in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Epinephrine (aerosol) in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Epinephrine (aerosol) in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Epinephrine (aerosol) in patients who are immunocompromised.
Administration and Monitoring
Administration
There is limited information regarding Epinephrine (aerosol) Administration in the drug label.
Monitoring
There is limited information regarding Epinephrine (aerosol) Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Epinephrine (aerosol) and IV administrations.
Overdosage
There is limited information regarding Epinephrine (aerosol) overdosage. If you suspect drug poisoning or overdose, please contact the National Poison Help hotline (1-800-222-1222) immediately.
Pharmacology
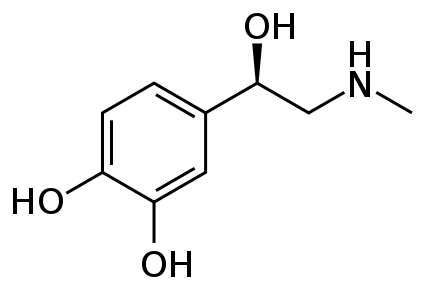
Lua error in Module:Redirect at line 65: could not parse redirect on page "Adrenaline".
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| [[Regulation of therapeutic goods |Template:Engvar data]] |
|
| Pregnancy category |
|
| Addiction liability | None |
| Routes of administration | IV, IM, endotracheal, IC, Nasal Ophthalmic |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | adrenergic synapse (MAO and COMT) |
| Elimination half-life | 2 minutes |
| Excretion | Urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C9H13NO3 |
| Molar mass | 183.204 g/mol |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Mechanism of Action
There is limited information regarding Epinephrine (aerosol) Mechanism of Action in the drug label.
Structure
There is limited information regarding Epinephrine (aerosol) Structure in the drug label.
Pharmacodynamics
There is limited information regarding Epinephrine (aerosol) Pharmacodynamics in the drug label.
Pharmacokinetics
There is limited information regarding Epinephrine (aerosol) Pharmacokinetics in the drug label.
Nonclinical Toxicology
There is limited information regarding Epinephrine (aerosol) Nonclinical Toxicology in the drug label.
Clinical Studies
There is limited information regarding Epinephrine (aerosol) Clinical Studies in the drug label.
How Supplied
There is limited information regarding Epinephrine (aerosol) How Supplied in the drug label.
Storage
- Do not puncture or incinerate canister. Do not store near heat.
- Exposure to temperatures above 120˚F may cause bursting.
- Store at controlled room temperature between 15˚C and 30˚C (59˚F and 86˚F).
Images
Drug Images
{{#ask: Page Name::Epinephrine (aerosol) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Epinephrine (aerosol) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
DIRECTIONS=
- Each inhalation contains the equivalent of 0.16 milligrams epinephrine delivered at the mouthpiece. Inhalation dosage for adults and children 4 years of age and older: start with one inhalation, then wait at least one minute. If not relieved, use MEDIHALER-EPI once more. Do not use again for at least three hours. The use of this product by children should be supervised by an adult. Children under 4 years of age: consult a physician.
- MEDIHALER-EPI delivers the same dose every time. It does not depend on the strength with which the canister is pressed against the oral adapter or on the time the canister is held down. The canister must be released before it can be depressed again for another dose.
- MEDIHALER-EPI is designed to deliver a sufficient dose for most people in one or occasionally two inhalations. It is not necessary to use several applications in rapid succession. Use only when you actually need relief. Do no get into the habit of using MEDIHALER-EPI indiscriminately.
- Before each use, remove dust cap and inspect oral adapter for foreign objects. Shake MADIHALER-EPI well. To get the medication deep into the lungs, follow these three simple steps which can be completed within five seconds:
- Breath out fully and place mouthpiece well into the mouth, aimed at the back of the throat.
- As you begin to breathe in deeply, press the canister firmly down into the adapter with the index finger, and continue to inhale. This releases one dose.
- Release pressure on canister and remove unit from the mouth. Hold the breath as long as possible, then breathe out slowly.
- Replace dust cap after each use.
- MEDIHALER-EPI is packaged in a shatterproof aluminum canister fitted with a specially designed valve. It can be used only with the MEDIHALER-EPI oral adapter, a non-breakable inhaler device.
DESCRIPTION
- For speed of relief and convenience, MEDIHALER-EPI oral inhaler comes ready to use. It consists of two parts:
- A. An aluminum canister, containing the medication, with a single dose valve, which fits into
- B. The white plastic oral adapter. The canister can be used only with the MEDIHALER-EPI oral adapter.
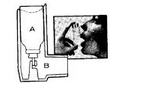
- The plastic oral adapter should be cleaned daily. Simply remove canister, wash adapter with soap and hot water, and rinse thoroughly. Dry adapter and replace the canister and dust cap.
Precautions with Alcohol
Alcohol-Epinephrine (aerosol) interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Epinephrine (aerosol) Brand Names in the drug label.
Look-Alike Drug Names
There is limited information regarding Epinephrine (aerosol) Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- Pages with script errors
- Use dmy dates from March 2015
- Articles with invalid date parameter in template
- Template:drugs.com link with non-standard subpage
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Drugboxes which contain changes to watched fields