Eflornithine
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Alberto Plate [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Eflornithine is a hair growth retardant, ornithine decarboxylase inhibitor that is FDA approved for the treatment of reduction of unwanted facial hair in women. Common adverse reactions include acne and stinging of skin.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
- Apply a thin layer of Eflornithine cream, 13.9% to affected areas of the face and adjacent involved areas under the chin and rub in thoroughly. Do not wash treated area for at least 4 hours. Use twice daily at least 8 hours apart or as directed by a physician. The patient should continue to use hair removal techniques as needed in conjunction with Eflornithine (Eflornithine should be applied at least 5 minutes after hair removal.) Cosmetics or sunscreens may be applied over treated areas after cream has dried.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Eflornithine in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Eflornithine in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
- Apply a thin layer of Eflornithine cream, 13.9% to affected areas of the face and adjacent involved areas under the chin and rub in thoroughly. Do not wash treated area for at least 4 hours. Use twice daily at least 8 hours apart or as directed by a physician. The patient should continue to use hair removal techniques as needed in conjunction with Eflornithine (Eflornithine should be applied at least 5 minutes after hair removal.) Cosmetics or sunscreens may be applied over treated areas after cream has dried.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Eflornithine in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Eflornithine in pediatric patients.
Contraindications
- Contraindicated in patients with a history of hypersensitivity to any components of the preparation.
Warnings
Discontinue use if hypersensitivity occurs.
Precautions
General
- For external use only.
- Transient stinging or burning sensation may occur when applied to abraded or broken skin.
Information for Patients
Patients using Eflornithine should receive the following information and instructions:
- This medication is not a depilatory, but rather appears to retard hair growth to improve the condition and the patient's appearance. Patients will likely need to continue using a hair removal method (e.g., shaving, plucking, etc.) in conjunction with Eflornithine cream, 13.9%.
- Onset of improvement was seen after as little as 4-8 weeks of treatment in the 24-week clinical trials. The condition may return to pretreatment levels 8 weeks after discontinuing treatment.
- If skin irritation or intolerance develops, direct the patient to temporarily reduce the frequency of application (e.g., once a day). If irritation continues, the patient should discontinue use of the product.
- Refer to the Patient Information Leaflet for additional important information and instructions.
Adverse Reactions
Clinical Trials Experience
- Adverse events reported for most body systems occurred at similar frequencies in Eflornithine cream, 13.9% and vehicle control groups. The most frequent adverse events related to treatment with Eflornithine were skin-related. The following table notes the percentage of adverse events associated with the use of Eflornithine or its vehicle that occurred at greater than 1% in both the vehicle-controlled studies and the open-label safety studies up to 1 year of continuous use.
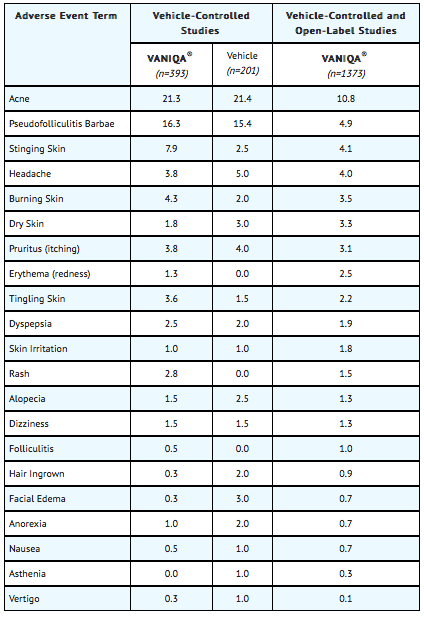
- Treatment-related skin adverse events that occurred in less than 1% of the subjects treated with Eflornithine are: bleeding skin, cheilitis, contact dermatitis, swelling of lips, herpes simplex, numbness, and rosacea.
- Adverse events were primarily mild in intensity and generally resolved without medical treatment or discontinuation of Eflornithine Only 2% of subjects discontinued studies due to an adverse event related to use of Eflornithine.
Laboratory Test Abnormalities
- No laboratory test abnormalities have been consistently found to be associated with Eflornithine. In an open-label study, some patients showed an increase in their transaminases; however, the clinical significance of these findings is not known.
Postmarketing Experience
There is limited information regarding Eflornithine Postmarketing Experience in the drug label.
Drug Interactions
- It is not known if Eflornithine has any interaction with other topically applied drug products.
Use in Specific Populations
Pregnancy
- In the first dermal embryo-fetal development study in rats treated with eflornithine hydrochloride cream, 13.9% (in which no precautions were taken to prevent ingestion of drug from application sites), maternal toxicity and fetal effects including reduced numbers of live fetuses, decreased fetal weights, and delayed ossification and development of the viscera were observed at doses of 225 and 450 mg/kg (15X and 29X the MRHD based on BSA, respectively). When the study was repeated under conditions that avoided ingestion from application sites, no maternal, fetal or teratogenic effects were observed at doses up to 450 mg/kg (29X the MRHD based on BSA). In the first study in which no precautions were taken to prevent ingestion, circulating plasma levels were 11- to 14-fold higher than in the second study in which ingestion was prevented. In a dermal embryo-fetal development study in rabbits treated with Eflornithine cream, 13.9% no adverse maternal or fetal effects occurred at doses up to 90 mg/kg (11X the MRHD based on BSA). Significant dermal irritation, as well as possible ingestion of Eflornithine occurred at 300 mg/kg/day (36X the MRHD based on BSA) and was associated with maternal deaths, abortions, increased fetal resorptions, and reduced fetal weights. Fetotoxicity in the absence of maternal toxicity has been reported in oral studies with eflornithine with fetal no-effect doses of 80 mg/kg in rats and 45 mg/kg in rabbits. In these studies, no evidence of teratogenicity was observed in rats given up to 200 mg/kg or in rabbits given up to 135 mg/kg.
- Although Eflornithine was not formally studied in pregnant patients, 22 pregnancies occurred during the trials. Nineteen of these pregnancies occurred while patients were using Eflornithine Of the 19 pregnancies, there were 9 healthy infants, 4 spontaneous abortions, 5 induced/elective abortions, and 1 birth defect (Down's Syndrome to a 35-year-old). *Because there are no adequate and well-controlled studies in pregnant women, the risk/benefit ratio of using Eflornithine in women with unwanted facial hair who are pregnant should be weighed carefully with serious consideration for either not implementing or discontinuing use of Eflornithine.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Eflornithine in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Eflornithine during labor and delivery.
Nursing Mothers
- It is not known whether or not eflornithine hydrochloride is excreted in human milk. Caution should be exercised when Eflornithine is administered to a nursing woman.
Pediatric Use
- The safety and effectiveness of this product have not been established in pediatric patients less than 12 years of age
Geriatic Use
- Of the 1373 patients on active treatment in clinical studies of Eflornithine approximately 7% were 65 years or older and approximately 1% were 75 or older. No apparent differences in safety were observed between older patients and younger patients.
Gender
There is no FDA guidance on the use of Eflornithine with respect to specific gender populations.
Race
There is no FDA guidance on the use of Eflornithine with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Eflornithine in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Eflornithine in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Eflornithine in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Eflornithine in patients who are immunocompromised.
Administration and Monitoring
Administration
There is limited information regarding Eflornithine Administration in the drug label.
Monitoring
There is limited information regarding Eflornithine Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Eflornithine and IV administrations.
Overdosage
- Overdosage information with Eflornithine is unavailable. Given the low percutaneous penetration of this drug, overdosage via the topical route is not expected. However, should very high topical doses (e.g., multiple tubes per day) or oral ingestion be encountered (a 30 g tube contains 4.2 g of eflornithine hydrochloride), the patient should be monitored, and appropriate supportive measures administered as necessary.
(Note: Use of an intravenous formulation of eflornithine hydrochloride at high doses (400 mg/kg/day or approximately 24 g/day) for the treatment of Trypanosoma brucei gambiense infection (African sleeping sickness) has been associated with adverse events and laboratory abnormalities. Adverse events in this setting have included hair loss, facial swelling, seizures, hearing impairment, stomach upset, loss of appetite, headache, weakness and dizziness. A variety of hematological toxicities, including anemia, thrombocytopenia and leukopenia have also been observed, but these were usually reversible upon discontinuation of treatment.)
Pharmacology
| |
Eflornithine
| |
| Systematic (IUPAC) name | |
| (RS)-2,5-diamino-2-(difluoromethyl)pentanoic acid | |
| Identifiers | |
| CAS number | |
| ATC code | D11 P01CX03 (WHO) |
| PubChem | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 182.2 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | 100% (Intravenous) Negligible (Dermal) |
| Metabolism | Not metabolised |
| Half life | 8 hours |
| Excretion | Renal |
| Therapeutic considerations | |
| Licence data |
, |
| Pregnancy cat. |
Category C for dermal cream |
| Legal status |
Template:Unicode Prescription only |
| Routes | Intravenous (discontinued) Dermal |
Mechanism of Action
There is limited information regarding Eflornithine Mechanism of Action in the drug label.
Structure
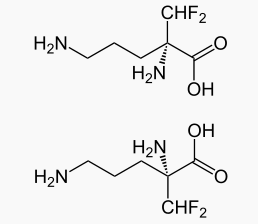
- Chemically, eflornithine hydrochloride is (±) -2-(difluoromethyl) ornithine monohydrochloride monohydrate, with the empirical formula C6H12F2N2O2• HCl•H2O, a molecular weight of 236.65 and the following structural formula:

- Anhydrous eflornithine hydrochloride has an empirical formula C6H12F2N2O2• HCl and a molecular weight of 218.65.
Pharmacodynamics
- There are no studies examining the inhibition of the enzyme ornithine decarboxylase ([ODC]]) in human skin following the application of topical eflornithine. However, there are studies in the literature that report the inhibition of ODC activity in skin following oral eflornithine. It is postulated that topical eflornithine hydrochloride irreversibly inhibits skin ODC activity. This enzyme is necessary in the synthesis of polyamines. Animal data indicate that inhibition of ornithine decarboxylase inhibits cell division and synthetic functions, which affect the rate of hair growth. Eflornithine cream, 13.9% has been shown to retard the rate of hair growth in non-clinical and clinical studies.
Pharmacokinetics
- The mean percutaneous absorption of eflornithine in women with unwanted facial hair, from a 13.9% w/w cream formulation, is less than 1% of the radioactive dose, following either single or multiple doses under conditions of clinical use, that included shaving within 2 hours before radiolabeled dose application in addition to other forms of cutting or plucking, and tweezing to remove facial hair. Steady state was reached within four days of twice-daily application. The apparent steady-state plasma t1/2 of eflornithine was approximately 8 hours. Following twice-daily application of 0.5 g of the cream (total dose 1 g/day; 139 mg as anhydrous eflornithine hydrochloride), under conditions of clinical use in women with unwanted facial hair (n=10), the steady-state Cmax, Ctrough and AUC12hr were approximately 10 ng/mL, 5 ng/mL, and 92 ng•hr/mL, respectively, expressed in terms of the anhydrous free base of eflornithine hydrochloride. At steady state, the dose-normalized peak concentrations (Cmax) and the extent of daily systemic exposure (AUC) of eflornithine following twice-daily application of 0.5 g of the cream (total dose 1 g/day) is estimated to be approximately 100- and 60-fold lower, when compared to 370 mg/day once-daily oral doses. This compound is not known to be metabolized and is primarily excreted unchanged in the urine.
Nonclinical Toxicology
- In a 12-month photocarcinogenicity study in hairless albino mice, animals treated with the vehicle alone showed an increased incidence of skin tumors induced by exposure to ultraviolet (UVA/UVB) light, whereas mice treated topically with Eflornithine at doses up to 600 mg/kg [19X the Maximum Recommended Human Dose (MRHD) based on body surface area (BSA)] showed an incidence of skin tumors equivalent to untreated-control animals.
- A 2-year dermal carcinogenicity study in CD-1 mice treated with Eflornithine revealed no evidence of carcinogenicity at daily doses up to 600 mg/kg (950X the MRHD based on AUC comparisons).
- Eflornithine did not elicit mutagenic effects in an Ames reverse-mutation assay or clastogenicity in primary human lymphocytes, with and without metabolic activation. In a dermal micronucleus assay, eflornithine hydrochloride cream, 13.9%, at doses up to 900 mg/kg (58X the MRHD based on BSA) in rats yielded no evidence of genotoxicity.
- In a dermal fertility and early embryonic development study in rats treated with Eflornithine, there were no adverse reproductive effects at doses up to 450 mg/kg (29X the MRHD based on BSA). In a peri- and postnatal study in rats, eflornithine administered in the drinking water was associated with maternal toxicity and reduced pup weights at doses of at least 625 mg/kg (40X the MRHD based on BSA) and a slightly reduced fertility index, which was considered to be of questionable biological significance, at 1698 mg/kg (110X the MRHD based on BSA). No effects were seen with an oral dose of 223 mg/kg (14X the MRHD based on BSA). In the latter study, the multiples of the human exposure are likely much higher, since eflornithine is well absorbed orally in rats, whereas minimal absorption occurs in humans treated topically.
Clinical Studies
- Results of topical dermal studies for contact sensitization, photocontact sensitization, and photocontact irritation reveal that under conditions of clinical use, Eflornithine is not expected to cause contact sensitization, phototoxic, or photosensitization reactions. Results of the topical dermal study for contact irritation did reveal that Eflornithine could cause irritation reactions in clinical use in susceptible individuals or under conditions of exaggerated use.
- Two randomized double-blind studies involving 594 female patients (393 treated with Eflornithine 201 with vehicle) treated twice daily for up to 24 weeks evaluated the efficacy of Eflornithine in the reduction of unwanted facial hair in women. Women in the trial had a customary frequency of removal of facial hair two or more times per week. Women with facial conditions such as severe inflammatory acne, women who were pregnant, and nursing mothers were excluded from the studies. Physicians assessed the improvement or worsening from the baseline condition (Physician's Global Assessment [PGA]), 48 hours after shaving, of all treated areas. Statistically significant improvement for Eflornithine cream, 13.9% versus vehicle was seen in each of these studies for "marked improvement" or greater response (24-week time point; p≤ 0.001). Marked improvement was seen consistently at 8 weeks after initiation of treatment and continued throughout the 24 weeks of treatment. Hair growth approached pretreatment levels within 8 weeks of treatment withdrawal. The success rate over time is graphically presented below for each pivotal trial.
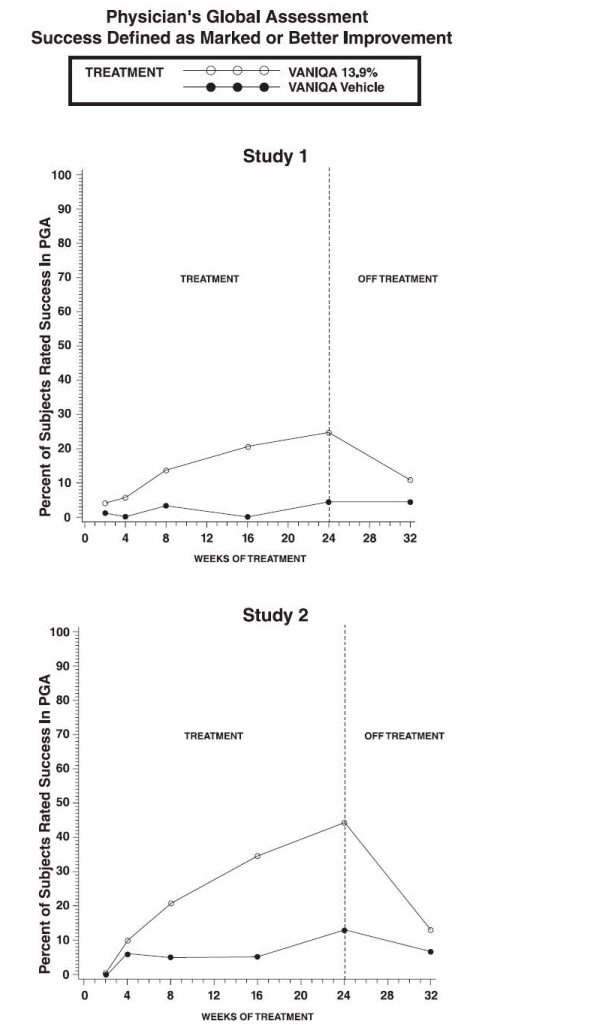
- Approximately 32% of patients showed marked improvement or greater (protocol definition of clinical success) after 24 weeks of treatment with Eflornithine (eflornithine hydrochloride) cream, 13.9%, compared to 8% with the vehicle. Combined results of these two trials through 24 weeks are presented below.
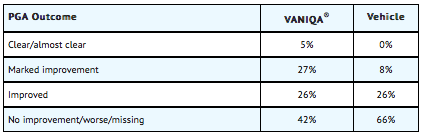
- Subgroup analyses appeared to suggest greater benefit for Whites than non-Whites (37% versus 22% success, respectively; p=0.017). However, non-Whites, mostly Black subjects, did have significant treatment benefit with 22% graded as success on Eflornithine compared to 5% on vehicle.
- About 12% of women in the clinical trials were postmenopausal. Significant improvement in PGA outcome versus vehicle was seen in postmenopausal women (38% compared to 0%, p≤ 0.001).
- Eflornithine statistically significantly reduced how bothered patients felt by their facial hair and by the time spent removing, treating, or concealing facial hair. These patient-observable differences were seen as early as 8 weeks after initiating treatment. Hair growth approached pretreatment levels within 8 weeks of treatment withdrawal.
- Clinical trials with Eflornithine involved over 1370 women with unwanted facial hair of skin types I-VI, of whom 68% were White, 17% Black, 11% Hispanic-Latino, 2% Asian-Pacific Islander, 0.6% American Native, and 1.3% other.
How Supplied
- Eflornithine hydrochloride cream, 13.9% is available as:
- 45 gram tube (NDC 0023-4857-45)
Storage
- Store at 25°C (77°F); excursions permitted to 15°-30°C (59°-86°F)
Images
Drug Images
{{#ask: Page Name::Eflornithine |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
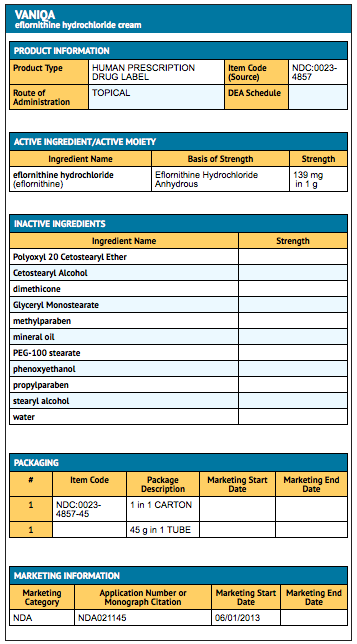
{{#ask: Label Page::Eflornithine |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Eflornithine Patient Counseling Information in the drug label.
Precautions with Alcohol
- Alcohol-Eflornithine interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
Look-Alike Drug Names
There is limited information regarding Eflornithine Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Label Page=Eflornithine |Label Name=Vaniqua package.png
}}