Crown ether
Crown ethers are heterocyclic chemical compounds that, in their simplest form, are cyclic oligomers of ethylene oxide. The essential repeating unit of any simple crown ether is ethyleneoxy, i.e., -CH2CH2O-, which repeats twice in dioxane and six times in 18-crown-6. In general, macrocycles of the (-CH2CH2O-)n type in which n ≥ 4 are referred to as "crown" ethers rather than by their lengthier systematic names: for instance, the systematic name of 18-crown-6 is "1,4,7,10,13,16-hexaoxacyclooctadecane". "Crown" refers to the resemblance between the structure of a crown ether bound to a cation, and a crown sitting on a head; the first number in a crown ether's name refers to the number of atoms in the cycle, and the second number refers to the number of those atoms which are oxygen.
Crown ethers are known for their ability to strongly solvate cations. In other words, the equilibrium is strongly towards the complex. The oxygen atoms are ideally situated to coordinate with a cation in the interior of the ring, whereas the exterior of the ring is hydrophobic. The result is that the complexed cation is soluble in nonpolar solvents. The size of the interior of the crown ether determines the size of the cation it can solvate. Therefore, 18-crown-6 has high affinity for potassium cation, 15-crown-5 for sodium cation and 12-crown-4 for lithium cation. The high affinity of 18-crown-6 for potassium ions contributes towards its toxicity in humans.
| 12-crown-4 chemical fourmula | 15-crown-5 chemical fourmula | 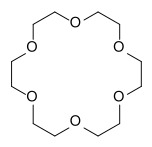
|
| 12-crown-4 | 15-crown-5 | 18-crown-6 |
Crown ethers are not the only macrocyclic ligands that have affinity for the potassium cation. Ionophores such as nonactin and valinomycin also display a marked preference for the potassium cation over other cations.
Early reports of crown ethers concentrated on synthetic methods for their production; only later were their properties and the fundamental theoretical implications thereof realized.
In 1967, Charles Pedersen, who was a chemist working at DuPont, discovered a simple method of synthesizing a crown ether when he was trying to prepare a complexing agent for divalent cations [1]. His strategy was to link two catechols through one hydroxyl on each molecule. This would give him a compound that could partially envelop the cation and, by ionization of the phenolic hydroxyls, neutralize the bound dication. He was surprised to isolate a by-product that bound or complexed with potassium cation but had no ionizable hydroxyl group. Citing earlier work on the dissolution of potassium in 16-crown-4 [2] [3], he realized that the cyclic polyethers represented a new class of complexing agents that were capable of binding alkali metal cations. He proceeded to report systematic studies of the synthesis and binding properties of crown ethers in a seminal series of papers. The fields of anionic synthetic reagents, phase transfer catalysts, biological ion transfer, and other emerging disciplines benefited profoundly from the discovery of crown ether.
Pedersen shared the 1987 Nobel Prize in Chemistry for the discovery of the synthetic routes to, and binding properties, of crown ethers.
Apart from its high affinity for potassium cations, 18-crown-6 can also bind to protonated amines and form very stable complexes in both solution and the gas phase. Some amino acids, such as lysine, contain a primary amine on their side chains. Those protonated amino groups can bind to the cavity of 18-crown-6 and form stable complexes in the gas phase. Hydrogen-bonds are formed between the three hydrogen atoms of protonated amines and three oxygen atoms of 18-crown-6. These hydrogen-bonds make the complex a stable adduct.
Aza- analogues of crown ethers exist as well, in particular cyclen.
- ↑ C. J. Pedersen, J. Am. Chem. Soc., 1967, 89, 7017.
- ↑ D. G. Stewart. D. Y. Waddan and E. T. Borrows, British Patent 785,229, Oct. 23, 1957.
- ↑ J. L. Down, J. Lewis, B. Moore and G. W. Wilkinson, Proc. Chem. Soc., 1959, 209; J. Chem. Soc., 1959, 3767.