Butoconazole
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Ammu Susheela, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Butoconazole is an antifungal that is FDA approved for the treatment of vulvovaginal candidiasis (infections caused by Candida). Common adverse reactions include vulvar/vaginal burning, itching, soreness and swelling, pelvic or abdominal pain or cramping.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
- GYNAZOLE • 1® Butoconazole Nitrate Vaginal Cream USP, 2% is indicated for the local treatment of vulvovaginal candidiasis (infections caused by Candida). The diagnosis should be confirmed by KOH smears and/or cultures.
- Note: GYNAZOLE • 1® Butoconazole Nitrate Vaginal Cream USP, 2% is safe and effective in non-pregnant women; however, the safety and effectiveness of this product in pregnant women has not been established.
- Of the 314 patients treated with GYNAZOLE • 1® Butoconazole Nitrate Vaginal Cream USP, 2% for 1 day in controlled clinical trials, 18 patients (5.7%) reported complaints such as vulvar/vaginal burning, itching, soreness and swelling, pelvic or abdominal pain or cramping, or a combination of two or more of these symptoms. In 3 patients (1%) these complaints were considered treatment-related. Five of the 18 patients reporting adverse events discontinued the study because of them.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Butoconazole in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Butoconazole in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Butoconazole in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Butoconazole in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Butoconazole in pediatric patients.
Contraindications
- GYNAZOLE • 1® Butoconazole Nitrate Vaginal Cream USP, 2% is contraindicated in patients with a history of hypersensitivity to any of the components of the product.
Warnings
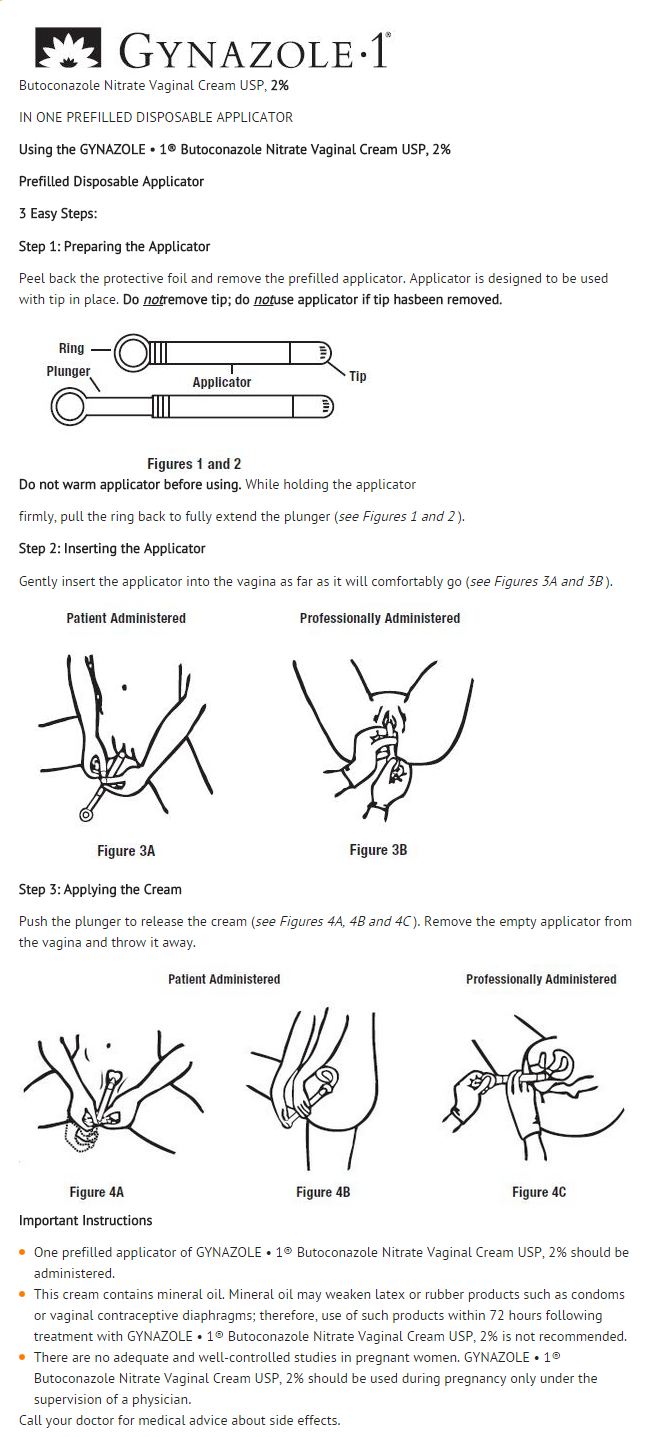
- This cream contains mineral oil. Mineral oil may weaken latex or rubber products such as condoms or vaginal contraceptive diaphragms; therefore, use of such products within 72 hours following treatment with GYNAZOLE • 1® Butoconazole Nitrate Vaginal Cream USP, 2% is not recommended.
- Recurrent vaginal yeast infections, especially those that are difficult to eradicate, can be an early sign of infection with the human immunodeficiency virus (HIV) in women who are considered at risk for HIV infection.
Adverse Reactions
Clinical Trials Experience
There is limited information regarding Clinical Trial Experience of Butoconazole in the drug label.
Postmarketing Experience
- Of the 314 patients treated with GYNAZOLE • 1® Butoconazole Nitrate Vaginal Cream USP, 2% for 1 day in controlled clinical trials, 18 patients (5.7%) reported complaints such as vulvar/vaginal burning, itching, soreness and swelling, pelvic or abdominal pain or cramping, or a combination of two or more of these symptoms. In 3 patients (1%) these complaints were considered treatment-related. Five of the 18 patients reporting adverse events discontinued the study because of them.Of the 314 patients treated with GYNAZOLE • 1® Butoconazole Nitrate Vaginal Cream USP, 2% for 1 day in controlled clinical trials, 18 patients (5.7%) reported complaints such as vulvar/vaginal burning, itching, soreness and swelling, pelvic or abdominal pain or cramping, or a combination of two or more of these symptoms. In 3 patients (1%) these complaints were considered treatment-related. Five of the 18 patients reporting adverse events discontinued the study because of them.
Drug Interactions
There is limited information regarding Butoconazole Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
- In pregnant rats administered 6 mg/kg/day of butoconazole nitrate intravaginally during the period of organogenesis, there was an increase in resorption rate and decrease in litter size; however, no teratogenicity was noted. This dose represents a 130- to 353-fold margin of safety based on serum levels achieved in rats following intravaginal administration compared to the serum levels achieved in humans following intravaginal administration of the recommended therapeutic dose of butoconazole nitrate.
- Butoconazole nitrate has no apparent adverse effect when administered orally to pregnant rats throughout organogenesis at dose levels up to 50 mg/kg/day (5 times the human dose based on mg/m2). Daily oral doses of 100, 300 or 750 mg/kg/day (10, 30 or 75 times the human dose based on mg/m2 respectively) resulted in fetal malformations (abdominal wall defects, cleft palate), but maternal stress was also evident at these higher dose levels. There were, however, no adverse effects on litters of rabbits who received butoconazole nitrate orally, even at maternally stressful dose levels (e.g., 150 mg/kg, 24 times the human dose based on mg/m2).
- Butoconazole nitrate, like other azole antifungal agents, causes dystocia in rats when treatment is extended through parturition. However, this effect was not apparent in rabbits treated with as much as 100 mg/kg/day orally (16 times the human dose based on mg/m2).
- There are, however, no adequate and well-controlled studies in pregnant women. GYNAZOLE • 1® Butoconazole Nitrate Vaginal Cream USP, 2% should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Butoconazole in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Butoconazole during labor and delivery.
Nursing Mothers
- It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when butoconazole nitrate is administered to a nursing woman.
Pediatric Use
There is no FDA guidance on the use of Butoconazole with respect to pediatric patients.
Geriatic Use
There is no FDA guidance on the use of Butoconazole with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Butoconazole with respect to specific gender populations.
Race
There is no FDA guidance on the use of Butoconazole with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Butoconazole in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Butoconazole in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Butoconazole in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Butoconazole in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
- Intravenous
Monitoring
There is limited information regarding Monitoring of Butoconazole in the drug label.
- Description
IV Compatibility
There is limited information regarding IV Compatibility of Butoconazole in the drug label.
Overdosage
There is limited information regarding Butoconazole overdosage. If you suspect drug poisoning or overdose, please contact the National Poison Help hotline (1-800-222-1222) immediately.
Pharmacology

| |
Butoconazole
| |
| Systematic (IUPAC) name | |
| 1-[4-(4-Chlorophenyl)-2-(2,6-dichlorophenyl)sulfanylbutyl]imidazole | |
| Identifiers | |
| CAS number | |
| ATC code | G01 |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 411.776 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
C(US) |
| Legal status | |
| Routes | Vaginal cream |
Mechanism of Action
- The exact mechanism of the antifungal action of butoconazole nitrate is unknown; however, it is presumed to function as other imidazole derivatives via inhibition of steroid synthesis. Imidazoles generally inhibit the conversion of lanosterol to ergosterol, resulting in a change in fungal cell membrane lipid composition. This structural change alters cell permeability and, ultimately, results in the osmotic disruption or growth inhibition of the fungal cell.
- Butoconazole nitrate is an imidazole derivative that has fungicidal activity in vitro against Candida spp. and has been demonstrated to be clinically effective against vaginal infections due to Candida albicans. Candida albicanshas been identified as the predominant species responsible for vulvovaginal candidiasis.
Structure
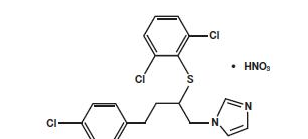
- GYNAZOLE • 1® Butoconazole Nitrate Vaginal Cream USP, 2% contains butoconazole nitrate 2%, an imidazole derivative with antifungal activity and it has the following chemical structure:
- Butoconazole nitrate is a white to off-white crystalline powder with a molecular weight of 474.79. It is sparingly soluble in methanol; slightly soluble in chloroform, methylene chloride, acetone, and ethanol; very slightly soluble in ethyl acetate; and practically insoluble in water. It melts at about 159°C with decomposition.
- GYNAZOLE • 1® Butoconazole Nitrate Vaginal Cream USP, 2% contains 2% butoconazole nitrate in a cream of edentate disodium, glyceryl monoisostearate, methylparaben, mineral oil, polyglyceryl-3 oleate, propylene glycol, propylparaben, colloidal silicon dioxide, sorbitol solution, purified water, and microcrystalline wax.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Butoconazole in the drug label.
Pharmacokinetics
- Following vaginal administration of butoconazole nitrate vaginal cream, 2% to 3 women, 1.7% (range 1.3-2.2%) of the dose was absorbed on average. Peak plasma levels (13.6-18.6 ng radioequivalents/mL of plasma) of the drug and its metabolites are attained between 12 and 24 hours after vaginal administration.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Butoconazole in the drug label.
Clinical Studies
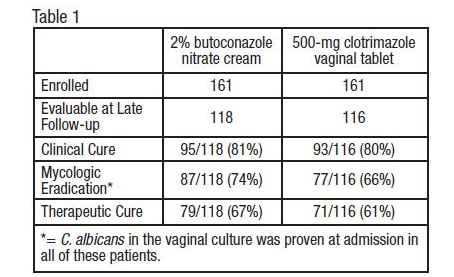
- Vulvovaginal Candidiasis: Two studies were conducted that compared 2% butoconazole nitrate cream with clotrimazole tablets. There were 322 enrolled patients, 161 received 2.0% butoconazole vaginal cream and 161 patients inserted the 500-mg clotrimazole vaginal tablet. At the second follow-up visit (30 days post-therapy), 118 patients in the butoconzole group and 116 in the clotrimazole group were evaluable for efficacy analysis, respectively. All of these patients had infection caused by Candida albicans.
- The efficacy of the study drugs was assessed by evaluating clinical, mycologic and therapeutic cure rates, which are summarized in Table 1.
- The therapeutic cure was defined as complete resolution of signs and symptoms of vaginal candidiasis (clinical cure) along with a negative KOH examination and negative culture for Candida spp. (microbiologic eradication) at the long term follow-up (30 days). The therapeutic cure rate was 67% in the butoconazole group and 61% in the clotrimazole group.
How Supplied
- GYNAZOLE • 1® Butoconazole Nitrate Vaginal Cream USP, 2% is available in cartons containing one single-dose prefilled disposable applicator (NDC 64011-246-01).
Storage
- Store at 25°C (77°F); excursions permitted to 15°-30°C
- (59°-86°F)
- Avoid heat above 30°C (86°F).
Images
Drug Images
{{#ask: Page Name::Butoconazole |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Butoconazole |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Precautions with Alcohol
- Alcohol-Butoconazole interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- GYNAZOLE 1 ®[1]
Look-Alike Drug Names
There is limited information regarding Butoconazole Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Label Page=Butoconazole |Label Name=But 04.png
}}
{{#subobject:
|Label Page=Butoconazole |Label Name=But 03.jpg
}}