Butenafine
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Ammu Susheela, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Butenafine is an antifungal that is FDA approved for the treatment of dermatologic infection, tinea (pityriasis) versicolor due to M. furfur (formerly P. orbiculare). Common adverse reactions include contact dermatitis, erythema, itching, skin irritation.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
- Butenafine HCl Cream, 1% is indicated for the topical treatment of the dermatologic infection, tinea (pityriasis) versicolor due to M. furfur (formerly P. orbiculare). Butenafine HCl cream was not studied in immunocompromised patients.
- Patients with tinea (pityriasis) versicolor should apply Mentax® Cream, 1%, once daily for two weeks. Sufficient Mentax® Cream should be applied to cover affected areas and immediately surrounding skin of patients with tinea versicolor. If a patient shows no clinical improvement after the treatment period, the diagnosis and therapy should be reviewed.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Butenafine in adult patients.
Non–Guideline-Supported Use
- Onychomycosis due to dermatophyte[1]
- Seborrheic dermatitis
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding Butenafine FDA-Labeled Indications and Dosage (Pediatric) in the drug label.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Butenafine in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Butenafine in pediatric patients.
Contraindications
- Butenafine Cream, 1%, is contraindicated in individuals who have known or suspected sensitivity to Mentax® Cream, 1%, or any of its components.
Warnings
- Butenafine Cream, 1%, is not for ophthalmic, oral, or intravaginal use.
Adverse Reactions
Clinical Trials Experience
There is limited information regarding Clinical Trial Experience of Butenafine in the drug label.
Postmarketing Experience
- In controlled clinical trials, 9 (approximately 1%) of 815 patients treated with Mentax® Cream, 1%, reported adverse events related to the skin. These included burning/stinging, itching and worsening of the condition. No patient treated with Mentax® Cream, 1%, discontinued treatment due to an adverse event. In the vehicle-treated patients, 2 of 718 patients discontinued because of treatment site adverse events, one of which was severe burning/stinging and itching at the site of application.
- In uncontrolled clinical trials, the most frequently reported adverse events in patients treated with Mentax® Cream, 1%, were: contact dermatitis, erythema, irritation, and itching, each occurring in less than 2% of patients.
- In provocative testing in over 200 subjects, there was no evidence of allergic-contact sensitization for either cream or vehicle base for Mentax® Cream, 1%.
Drug Interactions
There is limited information regarding Butenafine Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
- Subcutaneous doses of butenafine (dose levels up to 25 mg/kg/day administered during organogenesis) (equivalent to 0.5 times the maximum recommended dose in humans for tinea versicolor based on body surface area comparisons) were not teratogenic in rats. In an oral embryofetal development study in rabbits (dose levels up to 400 mg butenafine HCl/kg/day administered during organogenesis) (equivalent to 16 times the maximum recommended dose in humans for tinea versicolor based on body surface area comparisons), no treatment-related external, visceral, skeletal malformations or variations were observed.
In an oral peri- and post-natal developmental study in rats (dose levels up to 125 mg butenafine HCl/kg/day) (equivalent to 2.5 times the maximum recommended dose in humans for tinea versicolor based on body surface area comparisons), no treatment-related effects on postnatal survival, development of the F1 generation or their subsequent maturation and fertility were observed.
There are, however, no adequate and well-controlled studies that have been conducted with topically applied butenafine in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Pregnancy Category (AUS):
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Butenafine in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Butenafine during labor and delivery.
Nursing Mothers
It is not known if butenafine HCl is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised in prescribing Mentax® Cream, 1%, to a nursing woman.
Pediatric Use
Safety and efficacy in pediatric patients below the age of 12 years have not been studied since tinea versicolor is uncommon in patients below the age of 12 years.
Geriatic Use
There is no FDA guidance on the use of Butenafine with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Butenafine with respect to specific gender populations.
Race
There is no FDA guidance on the use of Butenafine with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Butenafine in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Butenafine in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Butenafine in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Butenafine in patients who are immunocompromised.
Administration and Monitoring
Administration
Monitoring
There is limited information regarding Butenafine Monitoring in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Butenafine in the drug label.
Overdosage
- Overdosage of butenafine HCl in humans has not been reported to date.
Pharmacology
There is limited information regarding Butenafine Pharmacology in the drug label.
Mechanism of Action
Structure
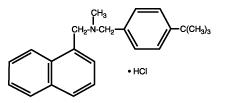
- Mentax® Cream, 1%, contains the synthetic antifungal agent, butenafine hydrochloride. Butenafine is a member of the class of antifungal compounds known as benzylamines which are structurally related to the allylamines.
- Butenafine HCl is designated chemically as N-4-tert-butylbenzyl-N-methyl-1-naphthalenemethylamine hydrochloride. The compound has the molecular formula C23H27N•HCl, a molecular weight of 353.93, and the following structural formula:
- Butenafine HCl is a white, odorless, crystalline powder. It is freely soluble in methanol, ethanol, and chloroform, and slightly soluble in water. Each gram of Mentax® Cream, 1%, contains 10 mg of butenafine HCl in a white cream base of purified water USP, propylene glycol dicaprylate, glycerin USP, cetyl alcohol NF, glyceryl monostearate SE, white petrolatum USP, stearic acid NF, polyoxyethylene (23) cetyl ether, benzyl alcohol NF, diethanolamine NF, and sodium benzoate NF.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Butenafine in the drug label.
Pharmacokinetics
Pharmacokinetics
- In one study conducted in healthy subjects for 14 days, 6 grams of Mentax® Cream, 1%, was applied once daily to the dorsal skin (3,000 cm2) of 7 subjects, and 20 grams of the cream was applied once daily to the arms, trunk and groin areas (10,000 cm2) of another 12 subjects. After 14 days of topical applications, the 6-gram dose group yielded a mean peak plasma butenafine HCl concentration, Cmax of 1.4 ± 0.8 ng/mL, occurring at a mean time to the peak plasma concentration, Tmax, of 15 ± 8 hours, and a mean area under the plasma concentration-time curve, AUC0-24 hrs of 23.9 ± 11.3 ng-hr/mL. For the 20-gram dose group, the mean Cmax was 5.0 ± 2.0 ng/mL, occurring at a mean Tmax of 6 ± 6 hours, and the mean AUC0-24 hrs was 87.8 ± 45.3 ng-hr/mL. A biphasic decline of plasma butenafine HCl concentrations was observed with the half-lives estimated to be 35 hours and > 150 hours, respectively.
- At 72 hours after the last dose application, the mean plasma concentrations decreased to 0.3 ± 0.2 ng/mL for the 6-gram dose group and 1.1 ± 0.9 ng/mL for the 20-gram dose group. Low levels of butenafine HCl remained in the plasma 7 days after the last dose application (mean: 0.1 ± 0.2 ng/mL for the 6-gram dose group, and 0.7 ± 0.5 ng/mL for the 20-gram dose group). The total amount (or % dose) of butenafine HCl absorbed through the skin into the systemic circulation has not been quantitated. It was determined that the primary metabolite in urine was formed through hydroxylation at the terminal t-butyl side-chain.
- In 11 patients with tinea pedis, butenafine HCl cream, 1%, was applied by the patients to cover the affected and immediately surrounding skin area once daily for 4 weeks, and a single blood sample was collected between 10 and 20 hours following dosing at 1, 2 and 4 weeks after treatment. The plasma butenafine HCl concentration ranged from undetectable to 0.3 ng/mL.
- In 24 patients with tinea cruris, butenafine HCl cream, 1%, was applied by the patients to cover the affected and immediately surrounding skin area once daily for 2 weeks (mean average daily dose: 1.3 ± 0.2 g). A single blood sample was collected between 0.5 and 65 hours after the last dose, and the plasma butenafine HCl concentration ranged from undetectable to 2.52 ng/mL (mean ± SD: 0.91 ± 0.15 ng/mL). Four weeks after cessation of treatment, the plasma butenafine HCl concentration ranged from undetectable to 0.28 ng/mL.
Microbiology
- Butenafine HCl is a benzylamine derivative with a mode of action similar to that of the allylamine class of antifungal drugs. Butenafine HCl is hypothesized to act by inhibiting the epoxidation of squalene, thus blocking the biosynthesis of ergosterol, an essential component of fungal cell membranes. The benzylamine derivatives, like the allylamines, act at an earlier step in the ergosterol biosynthesis pathway than the azole class of antifungal drugs. Depending on the concentration of the drug and the fungal species tested, butenafine HCl may be fungicidal or fungistatic in vitro. However, the clinical significance of these in vitro data are unknown.
- Butenafine HCl has been shown to be active against most strains of the following microorganisms, both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section:
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Butenafine in the drug label.
Clinical Studies
Tinea (pityriasis) versicolor
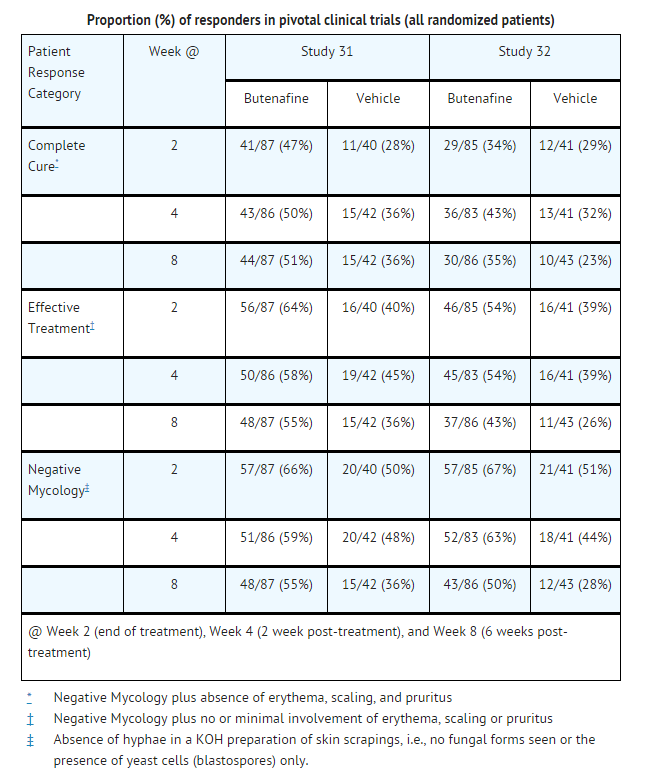
- In the following data presentations, patients with tinea (pityriasis) versicolor were studied. The term “Negative Mycology” is defined as absence of hyphae in a KOH preparation of skin scrapings, i.e., no fungal forms seen or the presence of yeast cells (blastospores) only. The term “Effective Treatment” is defined as Negative Mycology plus total signs and symptoms score (on a scale from zero to three) for erythema, scaling, and pruritus equal to or less than 1 at Week 8. The term “Complete Cure” refers to patients who had Negative Mycology plus sign/symptoms score of zero for erythema, scaling, and pruritus.
- Two separate studies compared Mentax® Cream to vehicle applied once daily for 2 weeks in the treatment of tinea (pityriasis) versicolor. Patients were treated for 2 weeks and were evaluated at the following weeks post-treatment: 2 (Week 4) and 6 (Week 8). All subjects with a positive baseline KOH and who were dispensed medications were included in the “intent-to-treat” analysis shown in the table below. Statistical significance (Mentax® vs. vehicle) was achieved for Effective Treatment, but not Complete Cure at 6 weeks post-treatment in Study 31. Marginal statistical significance (p = 0.051) (Mentax® vs. vehicle) was achieved for Effective Treatment, but not Complete Cure at 6 weeks post-treatment in Study 32. Data from these two controlled studies are presented in the table below.
- Tinea (pityriasis) versicolor is a superficial, chronically recurring infection of the glabrous skin caused by Malassezia furfur (formerly Pityrosporum orbiculare). The commensal organism is part of the normal skin flora. In susceptible individuals, the condition may give rise to hyperpigmented or hypopigmented patches on the trunk which may extend to the neck, arms, and upper thighs.
- Treatment of the infection may not immediately result in restoration of pigment of the affected sites. Normalization of pigment following successful therapy is variable and may take months, depending upon individual skin type and incidental sun exposure. The rate of recurrence of infection is variable.
How Supplied
- Butenafine Cream, 1%, is supplied in tubes in the following sizes:
- 15-gram tube (NDC 0378-6151-46)
- 30-gram tube (NDC 0378-6151-49)
Storage
- STORE BETWEEN 5° and 30°C (41° and 86°F)
Images
Drug Images
{{#ask: Page Name::Butenafine |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Butenafine |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Butenafine in the drug label.
Precautions with Alcohol
- Alcohol-Butenafine interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- MENTAX®[2]
Look-Alike Drug Names
There is limited information regarding Butenafine Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ Syed TA, Qureshi ZA, Ali SM, Ahmad S, Ahmad SA (1999). "Treatment of toenail onychomycosis with 2% butenafine and 5% Melaleuca alternifolia (tea tree) oil in cream". Trop Med Int Health. 4 (4): 284–7. PMID 10357864.
- ↑ "MENTAX- butenafine hydrochloride cream".
{{#subobject:
|Label Page=Butenafine |Label Name=Bute 03 Pack.jpg
}}
{{#subobject:
|Label Page=Butenafine |Label Name=Bute 04 Ingredients.png
}}