Boldenone
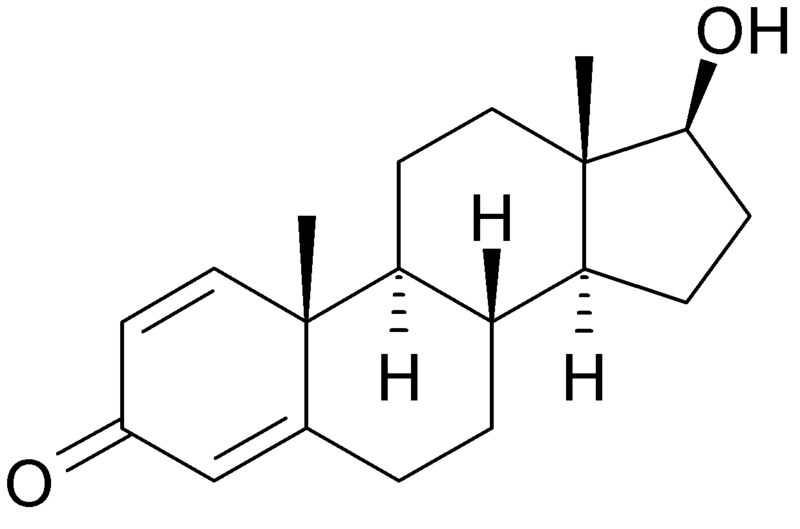 | |
| Clinical data | |
|---|---|
| Pregnancy category | |
| Routes of administration | Intramuscular |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | 14 days |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C19H26O2 |
| Molar mass | 286.409 g/mol |
| Melting point | 165 °C (329 °F) |
|
WikiDoc Resources for Boldenone |
|
Articles |
|---|
|
Most recent articles on Boldenone |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Boldenone at Clinical Trials.gov Clinical Trials on Boldenone at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Boldenone
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Boldenone Discussion groups on Boldenone Directions to Hospitals Treating Boldenone Risk calculators and risk factors for Boldenone
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Boldenone |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Overview
Boldenone (1,4-androstadiene-3-one-17β-ol, available as the undecylenate ester), also known under the trade names Equipoise, Ganabol, Equigan and Ultragan, is an anabolic steroid developed for veterinary use, mostly for treatment of horses. It is not indicated for use in humans in the US and is only available through veterinary clinics.
The activity of boldenone is mainly anabolic, with a low androgenic potency. It has a very long half-life, and can show up on a steroid test for up to 5 months.
Boldenone will increase nitrogen retention, protein synthesis, increases appetite and stimulates the release of erythropoietin in the kidneys.
Because it has similar properties to methandienone (methylated boldenone), it is a favorite for adding strength and size. Boldenone is popular for bulking because it increases the appetite. Boldenone is well known for increasing vascularity.
Boldenone has a low rate of aromatization (about 50% of Testosterone), which means it does not convert to estrogen easily and does not cause very much water retention.
It is easily detectable in blood tests due to its long metabolic half-life. Trace amounts of the drug can easily be detected for months after discontinued use.
Side effects
Although commonly compared to nandrolone, boldenone lacks progesterone receptor interaction and all the associated progestinic side effects. Boldenone can, however, produce mild androgenic side effects. Oily skin, acne, increased aggression and hair loss are all possible with this compound but rare. Boldenone does reduce to a more potent androgen dihydroboldenone via the 5alpha-reductase enzyme (analogous to the conversion of testosterone to dihydrotestosterone (DHT), however its affinity for this interaction in the human body is low to nonexistent. Suppression of the HPTA, water retention, possible increase in acne and possible conversion to estrogen, high blood pressure are all side effects associated with Boldenone. Boldenone is also known to cause anxiety and flu like symptoms which manifest themselves most acutely in short ester versions like: acetate, and propionate (almost unusable), but which are also present in the enanthate, cypionate, and undecyclenate forms.
References
- Pages with script errors
- Drugs with non-standard legal status
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs missing an ATC code
- Articles containing unverified chemical infoboxes
- Anabolic steroids
- Endocrinology