Bivalirudin
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Alejandro Lemor, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Bivalirudin is a direct thrombin inhibitor that is FDA approved for the prophylaxis of patients undergoing percutaneous transluminal coronary angioplasty (PTCA) and patients with unstable angina or risk of heparin induced thrombocytopenia (HIT) undergoing percutaneous coronary intervention (PCI).. Common adverse reactions include bleeding, headache, thrombocytopenia and fever.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Percutaneous Transluminal Coronary Angioplasty (PTCA)
- Dosing Information
- Initial dose: 0.75 mg/kg IV bolus
- Maintenance dose: 1.75 mg/kg/hr IV infusion for the duration of the procedure
- May be continued 4 hours post-procedure
- Five min after the bolus dose has been administered, an activated clotting time (ACT) should be performed and an additional bolus of 0.3 mg/kg should be given if needed.
Percutaneous Coronary Intervention (PCI)
- Dosing Information
- Initial dose: 0.75 mg/kg IV bolus
- Maintenance dose: 1.75 mg/kg/hr IV infusion for the duration of the procedure
- May be continued 4 hours post-procedure
- Five min after the bolus dose has been administered, an activated clotting time (ACT) should be performed and an additional bolus of 0.3 mg/kg should be given if needed.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Bivalirudin in adult patients.
Non–Guideline-Supported Use
Adjunct to Thrombolytic Therapy in Acute Myocardial Infarction,
- Dosing Information[1]
- Initial dose: 0.25 mg/kg IV bolus'
- Followed by an infusion of 0.5 mg/kg/hr for 12 hr and then 0.25 mg/kg/hr for 36 h
- Streptokinase was given after initial bolus, as 1.5 million units infused over 30 to 60 minutes.
Prophylaxis in Deep venous thrombosis
- Dosing Information
- 1 mg/kg SQ q8h[2]
Prophylaxis in Heart Surgery in Patients with Heparin-induced Thrombocytopenia with Thrombosis
- CHOOSE-ON Trial[3]
- 1 mg/kg IV bolus
- Followed by 2.5 mg/kg/hour infusion continued until 15 minutes before the end of cardiopulmonary bypass.
- Additionally, bivalirudin 50 mg was added to the cardiopulmonary bypass priming solution.
- CHOOSE-OFF Trial[4]
- 0.75 mg/kg IV bolus
- Followed by continuous infusion of 1.75 mg/kg/hour.
Thromboprophylaxis in Peripheral Arterial Bypass
- Dosing Information[5]
- 0.75 mg/kg IV bolus
- Followed by 1.75 mg/kg/hour infusion for the duration of the procedure
Thromboprophylaxis in Unstable angina
- Dosing Information[6]
- 0.1 mg/kg IV bolus
- Followed by 0.25 mg/kg/hr
- Titrated to an activated partial thromboplastin time (aPTT) of 55 to 85 seconds to be maintained for a minimum of 72 hr.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding Bivalirudin FDA-Labeled Indications and Dosage (Pediatric) in the drug label.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Bivalirudin in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Bivalirudin in pediatric patients.
Contraindications
- Active major bleeding
- Hypersensitivity (e.g., anaphylaxis) to bivalirudin or its components.
Warnings
Bleeding Events
- Although most bleeding associated with the use of bivalirudin in PCI/PTCA occurs at the site of arterial puncture, hemorrhage can occur at any site.
- An unexplained fall in blood pressure or hematocrit should lead to serious consideration of a hemorrhagic event and cessation of bivalirudin administration.
- Bivalirudin should be used with caution in patients with disease states associated with an increased risk of bleeding.
Coronary Artery Brachytherapy
- An increased risk of thrombus formation, including fatal outcomes, has been associated with the use of bivalirudin in gamma brachytherapy.
- If a decision is made to use bivalirudin during brachytherapy procedures, maintain meticulous catheter technique, with frequent aspiration and flushing, paying special attention to minimizing conditions of stasis within the catheter or vessels.
Adverse Reactions
Clinical Trials Experience
Bleeding
In 6010 patients undergoing PCI treated in the REPLACE-2 trial, bivalirudin patients exhibited statistically significantly lower rates of bleeding, transfusions, and thrombocytopenia as noted in Table 2.
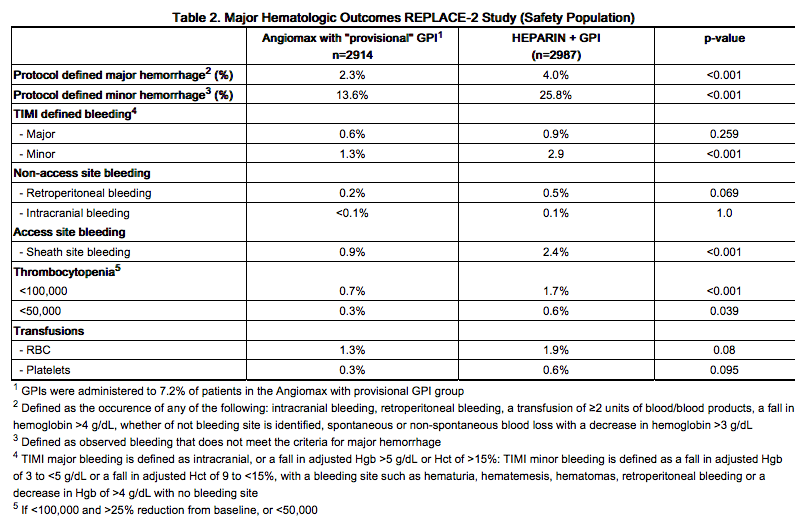
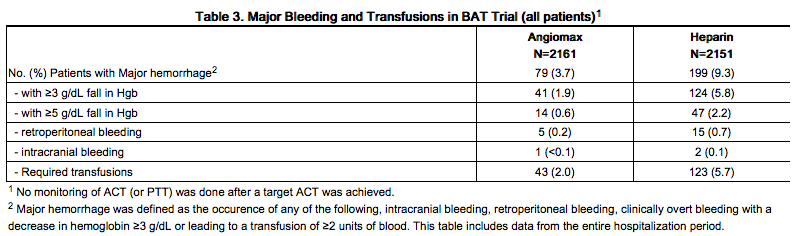
In 4312 patients undergoing PTCA for treatment of unstable angina in 2 randomized, double-blind studies comparing bivalirudin to heparin, bivalirudin patients exhibited lower rates of major bleeding and lower requirements for blood transfusions. The incidence of major bleeding is presented in Table 3. The incidence of major bleeding was lower in the bivalirudin group than in the heparin group.
In the AT-BAT study, of the 51 patients with HIT/HITTS, 1 patient who did not undergo PCI had major bleeding during CABG on the day following angiography. Nine patients had minor bleeding (mostly due to access site bleeding), and 2 patients developed thrombocytopenia.
Other Adverse Reactions
Adverse reactions, other than bleeding, observed in clinical trials were similar between the bivalirudin treated patients and the control groups. Adverse reactions (related adverse events ) seen in clinical studies in patients undergoing PCI and PTCA are shown in Tables 4 and 5.
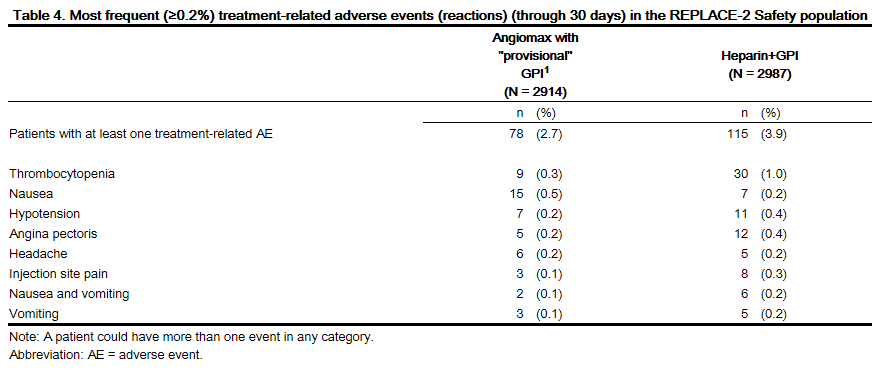
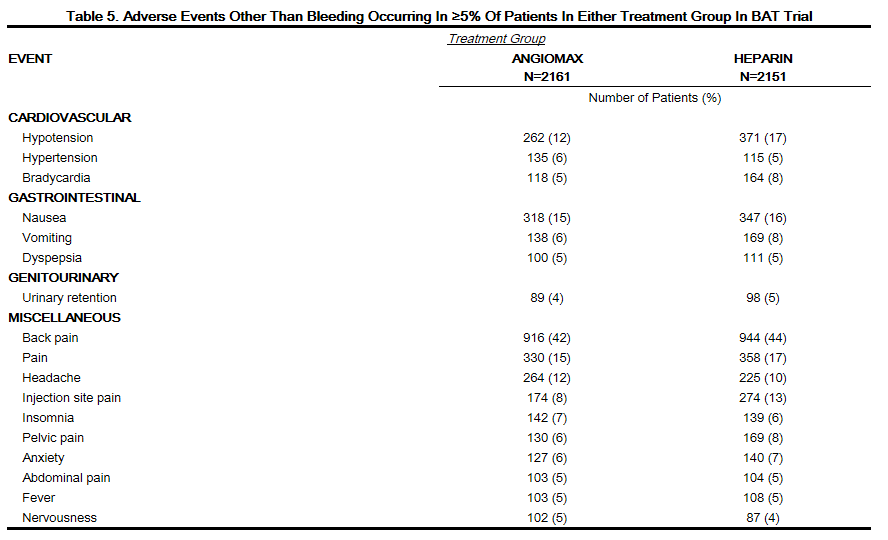
Serious, non-bleeding adverse events were experienced in 2% of 2161 bivalirudin-treated patients and 2% of 2151 heparin-treated patients. The following individual serious non-bleeding adverse events were rare (>0.1% to <1%) and similar in incidence between bivalirudin- and heparin-treated patients.
Body as a Whole
Cardiovascular
- Hypotension
- Syncope
- Vascular anomaly
- Ventricular fibrillation
Nervous
Respiratory
Urogenital
Immunogenicity/Re-Exposure
In in vitro studies, bivalirudin exhibited no platelet aggregation response against sera from patients with a history of HIT/HITTS. Among 494 subjects who received Angiomax in clinical trials and were tested for antibodies, 2 subjects had treatment-emergent positive bivalirudin antibody tests. Neither subject demonstrated clinical evidence of allergic or anaphylactic reactions and repeat testing was not performed. Nine additional patients who had initial positive tests were negative on repeat testing.
Postmarketing Experience
Because postmarketing adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following adverse reactions have been identified during postapproval use of bivalirudin: fatal bleeding; hypersensitivity and allergic reactions including reports of anaphylaxis; lack of anticoagulant effect; thrombus formation during PCI with and without intracoronary brachytherapy, including reports of fatal outcomes.
Drug Interactions
- In clinical trials in patients undergoing PCI/PTCA, co-administration of bivalirudin with heparin, warfarin, thrombolytics, or GPIs was associated with increased risks of major bleeding events compared to patients not receiving these concomitant medications.
- There is no experience with co-administration of bivalirudin and plasma expanders such as dextran.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): B Reproductive studies have been performed in rats at subcutaneous doses up to 150 mg/kg/day, (1.6 times the maximum recommended human dose based on body surface area) and rabbits at subcutaneous doses up to 150 mg/kg/day (3.2 times the maximum recommended human dose based on body surface area). These studies revealed no evidence of impaired fertility or harm to the fetus attributable to bivalirudin. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Bivalirudin is intended for use with aspirin. Because of possible adverse effects on the neonate and the potential for increased maternal bleeding, particularly during the third trimester, bivalirudin and aspirin should be used together during pregnancy only if clearly needed.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Bivalirudin in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Bivalirudin during labor and delivery.
Nursing Mothers
It is not known whether bivalirudin is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when bivalirudin is administered to a nursing woman.
Pediatric Use
The safety and effectiveness of bivalirudin in pediatric patients have not been established.
Geriatic Use
In studies of patients undergoing PCI, 44% were ≥65 years of age and 12% of patients were ≥75 years old. Elderly patients experienced more bleeding events than younger patients. Patients treated with bivalirudin experienced fewer bleeding events in each age stratum, compared to heparin.
Gender
There is no FDA guidance on the use of Bivalirudin with respect to specific gender populations.
Race
There is no FDA guidance on the use of Bivalirudin with respect to specific racial populations.
Renal Impairment
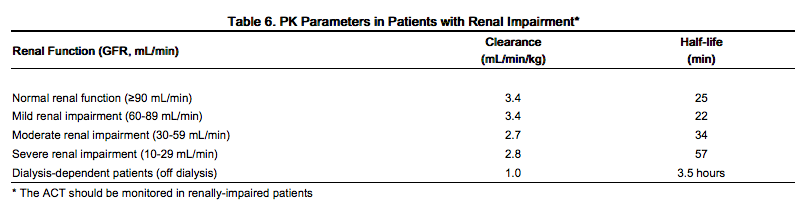
The disposition of bivalirudin was studied in PTCA patients with mild, moderate and severe renal impairment. The clearance of bivalirudin was reduced approximately 20% in patients with moderate and severe renal impairment and was reduced approximately 80% in dialysis-dependent patients. The infusion dose of bivalirudin may need to be reduced, and anticoagulant status monitored in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Bivalirudin in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Bivalirudin in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Bivalirudin in patients who are immunocompromised.
Administration and Monitoring
Administration
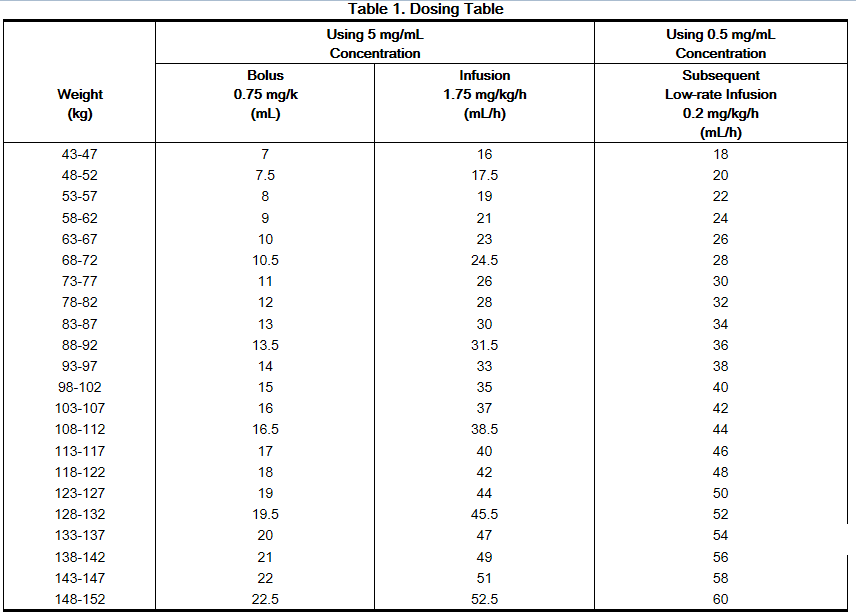
Bivalirudin is intended for intravenous bolus injection and continuous infusion after reconstitution and dilution. To each 250 mg vial, add 5 mL of sterile water for injection, USP. Gently swirl until all material is dissolved. Each reconstituted vial should be further diluted in 50 mL of 5% dextrose in water or 0.9% sodium chloride for injection to yield a final concentration of 5 mg/mL (e.g., 1 vial in 50 mL; 2 vials in 100 mL; 5 vials in 250 mL). The dose to be administered is adjusted according to the patient's weight.
If the low-rate infusion is used after the initial infusion, a lower concentration bag should be prepared. In order to prepare this bag, reconstitute the 250 mg vial with 5 mL of sterile water for injection, USP. Gently swirl until all material is dissolved. Each reconstituted vial should be further diluted in 500 mL of 5% dextrose in water or 0.9% sodium chloride for Injection to yield a final concentration of 0.5 mg/mL. The infusion rate to be administered should be selected from the right-hand column in Table 1.
Bivalirudin should be administered via an intravenous line. No incompatibilities have been observed with glass bottles or polyvinyl chloride bags and administration sets.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. Preparations of bivalirudin containing particulate matter should not be used. Reconstituted material will be a clear to slightly opalescent, colorless to slightly yellow solution.
Monitoring
Renal Impairment
No reduction in the bolus dose is needed for any degree of renal impairment. The infusion dose of bivalirudin may need to be reduced, and anticoagulant status monitored in patients with renal impairment. Patients with moderate renal impairment (30-59 mL/min) should receive an infusion of 1.75 mg/kg/h. If the creatinine clearance is less than 30 mL/min, reduction of the infusion rate to 1 mg/kg/h should be considered. If a patient is on hemodialysis, the infusion rate should be reduced to 0.25 mg/kg/h
IV Compatibility
The following drugs should not be administered in the same intravenous line with bivalirudin , since they resulted in haze formation, microparticulate formation, or gross precipitation when mixed with bivalirudin: alteplase, amiodarone HCl, amphotericin B, chlorpromazine HCl, diazepam, prochlorperazine edisylate, reteplase, streptokinase, and vancomycin HCl. Dobutamine was compatible at concentrations up to 4 mg/mL but incompatible at a concentration of 12.5 mg/mL.
Overdosage
- Cases of overdose of up to 10 times the recommended bolus or continuous infusion dose of bivalirudin have been reported in clinical trials and in postmarketing reports.
- A number of the reported overdoses were due to failure to adjust the infusion dose of bivalirudin in persons with renal dysfunction including persons on hemodialysis.
- Bleeding, as well as deaths due to hemorrhage, have been observed in some reports of overdose.
- In cases of suspected overdosage, discontinue bivalirudin immediately and monitor the patient closely for signs of bleeding. There is no antidote to bivalirudin. Bivalirudin is hemodialyzable.
Pharmacology
Mechanism of Action
Bivalirudin directly inhibits thrombin by specifically binding both to the catalytic site and to the anion-binding exosite of circulating and clot-bound thrombin. Thrombin is a serine proteinase that plays a central role in the thrombotic process, acting to cleave fibrinogen into fibrin monomers and to activate Factor XIII to Factor XIIIa, allowing fibrin to develop a covalently cross-linked framework which stabilizes the thrombus; thrombin also activates Factors V and VIII, promoting further thrombin generation, and activates platelets, stimulating aggregation and granule release. The binding of bivalirudin to thrombin is reversible as thrombin slowly cleaves the bivalirudin-Arg3-Pro4 bond, resulting in recovery of thrombin active site functions.
In in vitro studies, bivalirudin inhibited both soluble (free) and clot-bound thrombin, was not neutralized by products of the platelet release reaction, and prolonged the activated partial thromboplastin time (aPTT), thrombin time (TT), and prothrombin time (PT) of normal human plasma in a concentration-dependent manner. The clinical relevance of these findings is unknown.
Structure
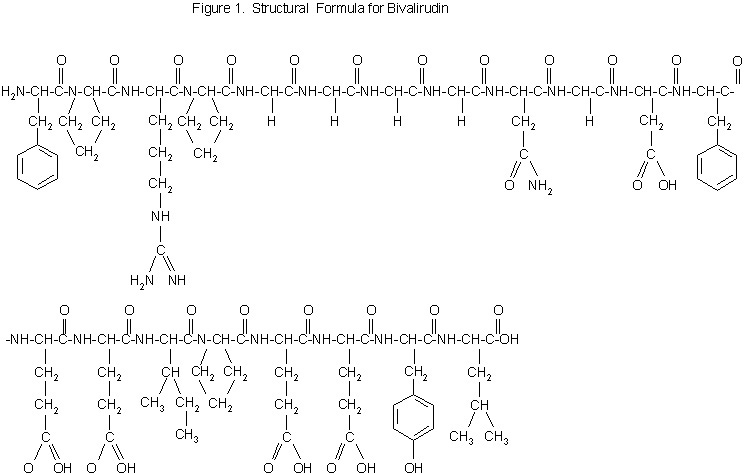
Bivalirudin is a specific and reversible direct thrombin inhibitor. The active substance is a synthetic, 20 amino acid peptide. The chemical name is D-phenylalanyl-L-prolyl-L-arginyl-L-prolyl-glycyl-glycyl-glycyl-glycyl-L-asparagyl-glycyl-L-aspartyl-L-phenylalanyl-L-glutamyl-L-glutamyl-L-isoleucyl-L-prolyl-L-glutamyl-L-glutamyl-L-tyrosyl-L-leucine trifluoroacetate (salt) hydrate (Figure 1). The molecular weight of Angiomax is 2180 daltons (anhydrous free base peptide).
Pharmacodynamics
In healthy volunteers and patients (with ≥70% vessel occlusion undergoing routine PTCA), bivalirudin exhibited dose- and concentration-dependent anticoagulant activity as evidenced by prolongation of the ACT, aPTT, PT, and TT. Intravenous administration of bivalirudin produces an immediate anticoagulant effect. Coagulation times return to baseline approximately 1 hour following cessation of bivalirudin administration.
In 291 patients with ≥70% vessel occlusion undergoing routine PTCA , a positive correlation was observed between the dose of bivalirudin and the proportion of patients achieving ACT values of 300 sec or 350 sec. At an bivalirudin dose of 1 mg/kg IV bolus plus 2.5 mg/kg/h IV infusion for 4 hours, followed by 0.2 mg/kg/h, all patients reached maximal ACT values >300 sec.
Pharmacokinetics
Bivalirudin exhibits linear pharmacokinetics following IV administration to patients undergoing PTCA. In these patients, a mean steady state bivalirudin concentration of 12.3 ± 1.7 mcg/mL is achieved following an IV bolus of 1 mg/kg and a 4-hour 2.5 mg/kg/h IV infusion. bivalirudin does not bind to plasma proteins (other than thrombin) or to red blood cells. bivalirudin is cleared from plasma by a combination of renal mechanisms and proteolytic cleavage, with a half-life in patients with normal renal function of 25 min.
The disposition of bivalirudin was studied in PTCA patients with mild, moderate, and severe renal impairment. Drug elimination was related to glomerular filtration rate (GFR). Total body clearance was similar for patients with normal renal function and with mild renal impairment (60-89 mL/min). Clearance was reduced in patients with moderate and severe renal impairment and in dialysis-dependent patients.
Bivalirudin is hemodialyzable, with approximately 25% cleared by hemodialysis.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term studies in animals have been performed to evaluate the carcinogenic potential of bivalirudin. Bivalirudin displayed no genotoxic potential in the in vitro bacterial cell reverse mutation assay (Ames test), the in vitro Chinese hamster ovary cell forward gene mutation test (CHO/HGPRT), the in vitro human lymphocyte chromosomal aberration assay, the in vitro rat hepatocyte unscheduled DNA synthesis (UDS) assay, and the in vivo rat micronucleus assay. Fertility and general reproductive performance in rats were unaffected by subcutaneous doses of bivalirudin up to 150 mg/kg/day, about 1.6 times the dose on a body surface area basis (mg/m2) of a 50 kg person given the maximum recommended dose of 15 mg/kg/day.
Clinical Studies
PCI/PTCA
Bivalirudin has been evaluated in five randomized, controlled interventional cardiology trials reporting 11,422 patients. Stents were deployed in 6062 of the patients in these trials - mainly in trials performed since 1995. Percutaneous transluminal coronary angioplasty, atherectomy or other procedures were performed in the remaining patients.
REPLACE-2 Trial
This was a randomized, double-blind, multicenter study reporting 6002 (intent-to-treat) patients undergoing PCI. Patients were randomized to treatment with bivalirudin with the "provisional" use of platelet glycoprotein IIb/IIIa inhibitor (GPI) or heparin plus planned use of GPI. GPIs were added on a "provisional" basis to patients who were randomized to bivalirudin in the following circumstances:
- decreased TIMI flow (0 to 2) or slow reflow;
- dissection with decreased flow;
- new or suspected thrombus;
- persistent residual stenosis;
- distal embolization;
- unplanned stent;
- suboptimal stenting;
- side branch closure;
- abrupt closure; clinical instability; and
- prolonged ischemia.
During the study, one or more of these circumstances occurred in 12.7% of patients in the bivalirudin with provisional GPI arm. GPIs were administered to 7.2% of patients in the bivalirudin with provisional GPI arm (62.2% of eligible patients).
Patients ranged in age from 25-95 years (median, 63); weight ranged from 35-199 kg (median 85.5); 74.4% were male and 25.6% were female. Indications for PCI included unstable angina (35% of patients), myocardial infarction within 7 days prior to intervention (8% of patients), stable angina (25%) and positive ischemic stress test (24%). Stents were deployed in 85% of patients. Ninety-nine percent of patients received aspirin and 86% received thienopyridines prior to study treatment.
Bivalirudin was administered as a 0.75 mg/kg bolus followed by a 1.75 mg/kg/h infusion for the duration of the procedure. The activated clotting time (ACT - measured by a Hemochron® device) was measured 5 min after the first bolus of study medication. If the ACT was <225 seconds, an additional bolus of 0.3 mg/kg was given. At investigator discretion, the infusion could be continued following the procedure for up to 4 hours. The median infusion duration was 44 min. Heparin was administered as a 65 U/kg bolus. The activated clotting time (ACT - measured by a Hemochron® device) was measured 5 min after the first bolus of study medication. If the ACT was <225 seconds, an additional bolus of 20 units/kg was given. GPIs (either abciximab or eptifibatide) were given according to manufacturers' instructions. Both randomized groups could be given "provisional" treatments during the PCI at investigator discretion, but under double-blind conditions. "Provisional" treatment with GPI was requested in 5.2% of patients randomized to heparin plus GPI (they were given placebo) and 7.2% patients randomized to bivalirudin with provisional GPI (they were given abciximab or eptifibatide according to pre-randomization investigator choice and patient stratification).
The percent of patients reaching protocol-specified levels of anticoagulation was greater in the bivalirudin with provisional GPI group than in the heparin plus GPI group. For patients randomized to bivalirudin with provisional GPI, the median 5 min ACT was 358 sec (interquartile range 320-400 sec) and the ACT was <225 sec in 3%. For patients randomized to heparin plus GPI, the median 5 min ACT was 317 sec (interquartile range 263-373 sec) and the ACT was <225 sec in 12%. At the end of the procedure, median ACT values were 334 sec (bivalirudin group) and 276 sec (heparin plus GPI group).
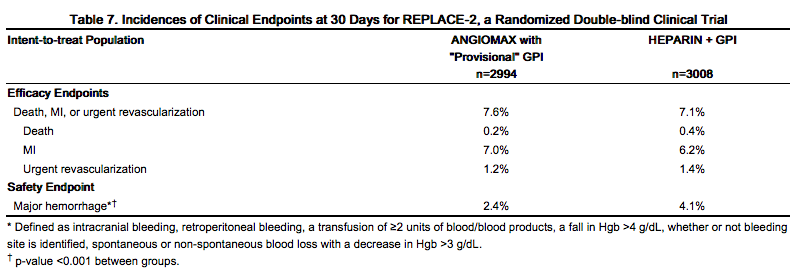
For the composite endpoint of death, MI, or urgent revascularization adjudicated under double-blind conditions, the frequency was higher (7.6%)(95% confidence interval 6.7%-8.6%) in the bivalirudin with "provisional" GPI arm when compared to the heparin plus GPI arm (7.1%)(95% confidence interval 6.1%-8.0%). However, major hemorrhage was reported significantly less frequently in the bivalirudin with provisional GPI arm (2.4%) compared to the heparin plus GPI arm (4.1%). Study outcomes are shown in Table 7.
At 12 months' follow-up, mortality was 1.9% among patients randomized to bivalirudin with "provisional" GPIs and 2.5% among patients randomized to heparin plus GPI.
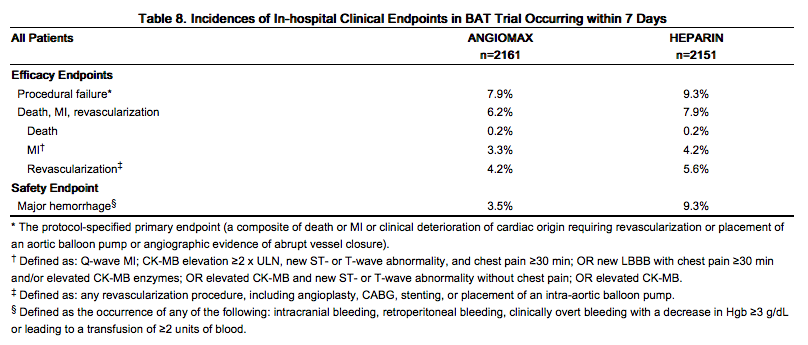
Bivalirudin Angioplasty Trial (BAT)
bivalirudin was evaluated in patients with unstable angina undergoing PTCA in two randomized, double-blind, multicenter studies with identical protocols. Patients must have had unstable angina defined as: (1) a new onset of severe or accelerated angina or rest pain within the month prior to study entry or (2) angina or ischemic rest pain which developed between four hours and two weeks after an acute myocardial infarction (MI). Overall, 4312 patients with unstable angina, including 741 (17%) patients with post-MI angina, were treated in a 1:1 randomized fashion with bivalirudin or heparin. Patients ranged in age from 29-90 (median 63) years, their weight was a median of 80 kg (39-120 kg), 68% were male, and 91% were Caucasian. Twenty-three percent of patients were treated with heparin within one hour prior to randomization. All patients were administered aspirin 300-325 mg prior to PTCA and daily thereafter. Patients randomized to bivalirudin were started on an intravenous infusion of bivalirudin (2.5 mg/kg/h). Within 5 min after starting the infusion, and prior to PTCA, a 1 mg/kg loading dose was administered as an intravenous bolus. The infusion was continued for 4 hours, then the infusion was changed under double-blinded conditions to bivalirudin (0.2 mg/kg/h) for up to an additional 20 hours (patients received this infusion for an average of 14 hours). The ACT was checked at 5 min and at 45 min following commencement. If on either occasion the ACT was <350 sec, an additional double-blinded bolus of placebo was administered. The bivalirudin dose was not titrated to ACT. Median ACT values were: ACT in sec (5th percentile-95th percentile): 345 sec (240-595 sec) at 5 min and 346 sec (range 269-583 sec) at 45 min after initiation of dosing. Patients randomized to heparin were given a loading dose (175 IU/kg) as an intravenous bolus 5 min before the planned procedure, with immediate commencement of an infusion of heparin (15 IU/kg/h). The infusion was continued for 4 hours. After 4 hours of infusion, the heparin infusion was changed under double-blinded conditions to heparin (15 IU/kg/h) for up to 20 additional hours. The ACT was checked at 5 min and at 45 min following commencement. If on either occasion the ACT was <350 sec, an additional double-blind bolus of heparin (60 IU/kg) was administered. Once the target ACT was achieved for heparin patients, no further ACT measurements were performed. All ACTs were determined with the Hemochron® device. The protocol allowed use of open-label heparin at the discretion of the investigator after discontinuation of blinded study medication, whether or not an endpoint event (procedural failure) had occurred. The use of open-label heparin was similar between bivalirudin and heparin treatment groups (about 20% in both groups).
The studies were designed to demonstrate the safety and efficacy of bivalirudin in patients undergoing PTCA as a treatment for unstable angina as compared with a control group of similar patients receiving heparin during and up to 24 hours after initiation of PTCA. The primary protocol endpoint was a composite endpoint called procedural failure, which included both clinical and angiographic elements measured during hospitalization. The clinical elements were: the occurrence of death, MI, or urgent revascularization, adjudicated under double-blind conditions. The angiographic elements were: impending or abrupt vessel closure. The protocol-specified safety endpoint was major hemorrhage.
The median duration of hospitalization was 4 days for both the bivalirudin and the heparin treatment groups. The rates of procedural failure were similar in the bivalirudin and heparin treatment groups. Study outcomes are shown in Table 8.
AT-BAT Trial
This was a single-group open-label study which enrolled 51 patients with heparin-induced thrombocytopenia (HIT) or heparin induced thrombocytopenia and thrombosis syndrome (HITTS) undergoing PCI. Evidence for the diagnosis of HIT/HITTS was based on a clinical history of a decrease of platelets in patients after heparin administration [new diagnosis or history of clinically suspected or objectively documented HIT/HITTS defined as either: 1) HIT: positive heparin-induced platelet aggregation (HIPA) or other functional assay where the platelet count has decreased to <100,000/mL (minimum 30% from prior to heparin), or has decreased to <150,000/mL (minimum 40% from prior to heparin), or has decreased as above within hours of receiving heparin in a patient with a recent, previous exposure to heparin; 2) HITTS: thrombocytopenia as above plus arterial or venous thrombosis diagnosed by physician examination/laboratory and/or appropriate imaging studies]. Patients ranged in age from 48-89 years (median 70); weight ranged from 42-123 kg (median 76); 50% were male and 50% were female. Bivalirudin was administered as either 1 mg/kg bolus followed by 2.5 mg/kg/h (high dose in 28 patients) or 0.75 mg/kg bolus followed by a 1.75 mg/kg/h infusion (lower dose in 25 patients) for up to 4 hours. Ninety-eight percent of patients received aspirin, 86% received clopidogrel and 19% received GPIs.
The median ACT values at the time of device activation were 379 sec (high dose) and 317 sec (lower dose). Following the procedure, 48 of the 51 patients (94%) had TIMI grade 3 flow and stenosis <50%. One patient died during a bradycardic episode 46 hours after successful PCI, another patient required surgical revascularization, and one patient experienced no flow requiring a temporary intra-aortic balloon.
Two of the fifty-one patients with the diagnosis of HIT/HITTS developed thrombocytopenia after receiving bivalirudin and GPIs.
How Supplied
Bivalirudin is supplied as a sterile, lyophilized powder in single-use, glass vials. After reconstitution, each vial delivers 250 mg of bivalirudin.
- NDC 65293-001-01
Storage
Store bivalirudin dosage units at 20-25°C (68-77°F). Excursions to 15-30°C permitted.
Images
Drug Images
{{#ask: Page Name::Bivalirudin |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Bivalirudin |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Advise patients to watch carefully for any signs of bleeding or bruising and to report these to their health care provider when they occur.
- Advise patients to discuss with their health care provider their use of any other medications, including over-the-counter medications or herbal products, prior to bivalirudin use.
- Examples of other medications that should not be taken with bivalirudin are warfarin and heparin.
Precautions with Alcohol
Alcohol-Bivalirudin interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
Angiomax
Look-Alike Drug Names
There is limited information regarding Bivalirudin Look-Alike Drug Names in the drug label.
Drug Shortage
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ H. White (2001). "Thrombin-specific anticoagulation with bivalirudin versus heparin in patients receiving fibrinolytic therapy for acute myocardial infarction: the HERO-2 randomised trial". Lancet. 358 (9296): 1855–1863. PMID 11741625. Unknown parameter
|month=ignored (help) - ↑ J. S. Ginsberg, M. T. Nurmohamed, M. Gent, B. MacKinnon, J. Sicurella, P. Brill-Edwards, M. N. Levine, A. A. Panju, P. Powers & P. Stevens (1994). "Use of Hirulog in the prevention of venous thrombosis after major hip or knee surgery". Circulation. 90 (5): 2385–2389. PMID 7955198. Unknown parameter
|month=ignored (help) - ↑ Andreas Koster, Cornelius M. Dyke, Gabriel Aldea, Nicholas G. Smedira, Harry L. 2nd McCarthy, Solomon Aronson, Roland Hetzer, Edwin Avery, Bruce Spiess & A. Michael Lincoff (2007). "Bivalirudin during cardiopulmonary bypass in patients with previous or acute heparin-induced thrombocytopenia and heparin antibodies: results of the CHOOSE-ON trial". The Annals of thoracic surgery. 83 (2): 572–577. doi:10.1016/j.athoracsur.2006.09.038. PMID 17257990. Unknown parameter
|month=ignored (help) - ↑ Cornelius M. Dyke, Gabriel Aldea, Andreas Koster, Nicholas Smedira, Edwin Avery, Solomon Aronson, Bruce D. Spiess & A. Michael Lincoff (2007). "Off-pump coronary artery bypass with bivalirudin for patients with heparin-induced thrombocytopenia or antiplatelet factor four/heparin antibodies". The Annals of thoracic surgery. 84 (3): 836–839. doi:10.1016/j.athoracsur.2007.04.007. PMID 17720385. Unknown parameter
|month=ignored (help) - ↑ David E. Allie, Patrick Hall, Nicolas W. Shammas, Robert Safian, John R. Laird, John J. Young & Ajay Virmani (2004). "The Angiomax Peripheral Procedure Registry of Vascular Events Trial (APPROVE): in-hospital and 30-day results". The Journal of invasive cardiology. 16 (11): 651–656. PMID 15550739. Unknown parameter
|month=ignored (help) - ↑ Elliott M. Antman, Carolyn H. McCabe & Eugene Braunwald (2002). "Bivalirudin as a replacement for unfractionated heparin in unstable angina/non-ST-elevation myocardial infarction: observations from the TIMI 8 trial. The Thrombolysis in Myocardial Infarction". American heart journal. 143 (2): 229–234. PMID 11835024. Unknown parameter
|month=ignored (help)
{{#subobject:
|Label Page=Bivalirudin |Label Name=Bivalirudin 10.png
}}
{{#subobject:
|Label Page=Bivalirudin |Label Name=Bivalirudin FDA panel.png
}}
