Bicisate dihydrochloride
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Kiran Singh, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Bicisate dihydrochloride is a diagnostic agent that is FDA approved for the diagnosis of stroke in patients in whom stroke has already been diagnosed. Common adverse reactions include headache, dizziness, seizure, anxiety, malaise, parosmia, hallucinations, rash, nausea, syncope, cardiac failure, hypertension, angina,apnea and cyanosis..
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- Neurolite single photon emission computerized tomography (SPECT) is indicated as an adjunct to conventional CT or MRI imaging in the localization of stroke in patients in whom stroke has already been diagnosed.
- Neurolite is not indicated for assessment of functional viability of brain tissue. Also, Neurolite is not indicated for distinguishing between stroke and other brain lesions.
Dosing
- The recommended dose range for intravenous administration for a 70 kg patient is 370-1110 MBq (10-30mCi). Dose adjustments for age, weight, gender or renal or hepatic impairment have not been studied.
- The dose for the patient should be measured by a suitable radioactivity calibration system immediately before administration to the patient. Radiochemical purity should be checked before administration to the patient.
- Neurolite, like other parenteral drug products, should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit. Preparations containing particulate matter or discoloration should not be administered. They should be disposed of in a safe manner, in compliance with all applicable regulations.
- Prior to reconstitution, vial A and vial B are stored at 15-25°C. Protect vial A from light.
- Store at controlled room temperature after preparation.
- Aseptic techniques and effective shielding should be employed in withdrawing doses for administration to patients. Waterproof gloves and effective shielding should be worn when handling the product.
RADIATION DOSIMETRY
- The radiation doses to organs and tissues of an average patient (70 kg) for Technetium Tc99m Bicisate injected intravenously for 370 MBq (10 mCi) are shown in Table 4 and for 1110 MBq (30 mCi) are shown in Table 5.


Radiation dosimetry calculations performed by Radiation Internal Dose Information Center, Oak Ridge Institute for Science and Education, PO Box 117, Oak Ridge, TN 37831-0117 (865) 576-3448.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Bicisate dihydrochloride in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Bicisate dihydrochloride in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Bicisate dihydrochloride in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Bicisate dihydrochloride in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Bicisate dihydrochloride in pediatric patients.
Contraindications
- None known
Warnings
- None known
Adverse Reactions
Clinical Trials Experience
- In clinical trials, Neurolite has been administered to 1063 subjects (255 normals, 808 patients). Of these, 566 (53%) were men and 494 (47%) were women. The mean age was 58 years (range 17 to 92 years). In the 808 patients, who had experienced neurologic events, there were 11 (1.4%) deaths, none of which were clearly attributed to Neurolite.
- A total of 60 subjects experienced adverse reactions; the adverse reaction rates were comparable in the <65 year, and the >65 year age groups.
- The following adverse effects were observed in ≤ 1% of the subjects: headache, dizziness, seizure, agitation/anxiety, malaise/somnolence, parosmia, hallucinations, rash, nausea, syncope, cardiac failure, hypertension, angina, and apnea/cyanosis.
- In clinical trials of 197 patients, there were inconsistent changes in the serum calcium and phosphate levels. The cause of the changes has not been identified and their frequency and magnitude have not been clearly characterized. None of the changes required medical intervention.
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Bicisate dihydrochloride in the drug label.
Drug Interactions
There is limited information regarding Bicisate dihydrochloride Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA):
There is no FDA guidance on usage of Bicisate dihydrochloride in women who are pregnant.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Bicisate dihydrochloride in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Bicisate dihydrochloride during labor and delivery.
Nursing Mothers
There is no FDA guidance on the use of Bicisate dihydrochloride with respect to nursing mothers.
Pediatric Use
There is no FDA guidance on the use of Bicisate dihydrochloride with respect to pediatric patients.
Geriatic Use
There is no FDA guidance on the use of Bicisate dihydrochloride with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Bicisate dihydrochloride with respect to specific gender populations.
Race
There is no FDA guidance on the use of Bicisate dihydrochloride with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Bicisate dihydrochloride in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Bicisate dihydrochloride in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Bicisate dihydrochloride in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Bicisate dihydrochloride in patients who are immunocompromised.
Administration and Monitoring
Administration
INSTRUCTIONS FOR PREPARATION OF TECHNETIUM TC99M BICISATE
- Preparation of the Technetium Tc99m Bicisate from the NEUROLITE®, Kit for the Preparation of Technetium Tc99m Bicisate Injection, is done by the following aseptic procedure:
Prior to adding the Sodium Pertechnetate Tc99m Injection to vial B (the liquid vial), write the estimated activity, date, and time of preparation in the space provided on the vial label. Then tear off a radiation symbol and attach it to the neck of the vial.
- Waterproof gloves should be worn during the preparation procedure. Remove the plastic disc from both vials and swab the top of each vial closure with alcohol to disinfect the surface.
- Place vial B in a suitable radiation shield appropriately labeled with date, time of preparation, volume and activity.
- With a sterile shielded syringe, aseptically add 3.70 GBq (100 mCi) sterile, non-pyrogenic, oxidant-free Sodium Pertechnetate Tc99m Injection, in approximately 2.0 mL, to vial B. Without withdrawing the needle, remove an equal volume of air to maintain pressure within the vial.
- With a sterile syringe, rapidly inject 3.0 mL of Sodium Chloride Injection (0.9%) into vial A (the lyophilized vial) to dissolve the contents. Without withdrawing the needle, remove an equal volume of air to maintain pressure within the vial. Shake the contents of the vial for a few seconds.
- With another sterile syringe, immediately (within 30 seconds) withdraw 1.0 mL of vial A and inject it into vial B. Discard vial A immediately.
- Swirl the contents of the vial B for a few seconds, and allow this mixture to stand for thirty (30) minutes at room temperature.
- Examine the vial contents for particulates and discoloration prior to patient administration. If particulate matter and/or discoloration are seen, DO NOT USE.
Assay the reaction vial using a suitable radioactivity calibration system. Record the Technetium Tc99m concentration, total volume, assay time and date, expiration time and lot number on the vial shield label and affix the label to the shield. Store the reaction vial containing the Technetium Tc99m Bicisate at controlled room temperature until use; at such time the product should be aseptically withdrawn. The vial contains no preservative.
- Note: Adherence to the above product reconstitution instructions is recommended.
- Product should be used within 6 hours of preparation.
Monitoring
There is limited information regarding Monitoring of Bicisate dihydrochloride in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Bicisate dihydrochloride in the drug label.
Overdosage
- There is limited information regarding Chronic Overdose of Bicisate dihydrochloride in the drug label.
Pharmacology
There is limited information regarding Bicisate dihydrochloride Pharmacology in the drug label.
Mechanism of Action
There is limited information regarding Bicisate dihydrochloride Mechanism of Action in the drug label.
Structure
- This kit formulation consists of two nonradioactive vials: Vial A contains bicisate dihydrochloride (N, N'-1,2-ethylenediylbis-L-cysteine diethyl ester dihydrochloride) and a reducing agent as a lyophilized solid and vial B contains a buffer solution. Both vials are sterile and non-pyrogenic.
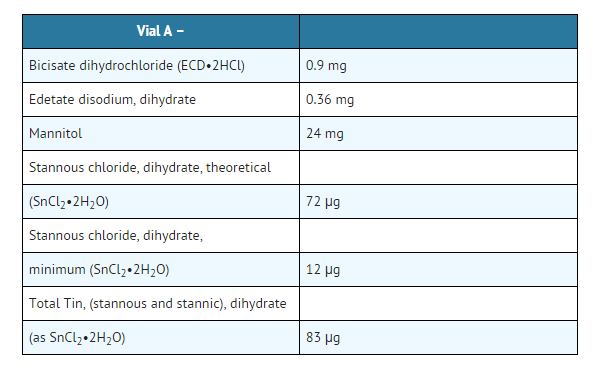
- The contents of vial A are lyophilized and stored under nitrogen. The pH of the solution before lyophilization is 2.7 ± 0.25. This vial is stored at 15-25°C. Protect from light.

- The contents of vial B are stored under air. The pH of the solution is 7.6 ± 0.4. This vial is stored at 15-25°C.
- This drug is administered by intravenous injection for diagnostic use after reconstitution with sterile, non-pyrogenic, oxidant-free Sodium Pertechnetate Tc99m Injection. The precise structure of the Technetium complex is [N, N'-ethylenedi-L-cysteinato(3-)]oxo[99mTc] technetium (V), diethyl ester.
Pharmacodynamics
- Localization of the parent compound in the brain in part depends upon both perfusion of the region and uptake of Technetium Tc99m Bicisate by the cell. Once in the brain cells, the parent compound is metabolized to polar, less diffusible compounds. Studies in 21 normal volunteers show cellular uptake of 4.8-6.5% of the injected dose at five minutes after injection. The degree of cell function or viability needed for uptake is not known. The degree of cell function or viability needed for metabolism of the parent compound to the less diffusible compounds has not been determined. The likelihood that the metabolic pathway is damaged by ischemia is not known. Whether or not and to what extent uptake correlates with viability or function is not known.
- The pharmacodynamics of Neurolite have not been evaluated for differences associated with age, gender, weight and liver or renal impairment. It is not known whether dosage adjustments for these factors are needed.
Pharmacokinetics
- In a study in 16 normals (13 men and 3 women, mean age of 31 ± 10 years; mean weight of 72 ± 11 kg), the pharmacokinetic profile in blood best fits a three compartment model with half-lives of 43 seconds, 49.5 minutes and 533 minutes. The highest concentration of radioactivity measured in blood was found at 0.5 minutes after intravenous injection and was 13.9% of the injected dose. Technetium Tc99m Bicisate and its major metabolites are not protein-bound.
Nonclinical Toxicology
- There is limited information regarding Nonclinical Toxicology of Bicisate dihydrochloride in the drug label.
Clinical Studies
- Two clinical trials were performed in a total of 359 subjects (273 with stroke, 86 normal). Of these 56% were men and 44% were women. The mean age was 60.2 years (range 23 to 92 years). Subjects were 87.2% Caucasian, 8.4% Black, 2.2% Hispanic, 1.7% Oriental and 0.6% other.
- Eligible patients had a confirmed stroke. Patients with other brain lesions were not evaluated. Subjects received Neurolite (mean dose range 10-30mCi) and underwent SPECT imaging and either CT or MRI scans within 0-30 days of the onset of signs and symptoms of stroke. CT or MRI and the administration of Neurolite occurred at different and variable times after the onset of a stroke. The effect of the timing on the accuracy of the images cannot be evaluated. The Neurolite scan results were blindly compared to unblinded CT/MRI results, the short standardized neurologic examination (SSNE) and the final diagnosis (e.g., the overall combined clinical impression with CT/MRI and SSNE).
- In these studies, at least one of three blinded readers made a diagnosis of stroke in 190 (85%) of the Neurolite SPECT studies and in 238 (88%) CT/MRI studies. The Neurolite and CT/MRI imaging results versus the SSNE and final diagnosis were comparable. Neurolite had 11 false positive and 34 false negatives. CT/MRI had 0 false positive and 31 false negatives. Both Neurolite and CT/MRI missed strokes (true positives) that were identified by the other modality. The majority of the false negatives in either modality were within 15 days of the clinical stroke.
- The trials were not designed to determine when Neurolite or CT/MRI studies could become positive in relationship to the time of the stroke. The relevance of the Neurolite scan results to the prediction of neurologic function or brain cell viability is not known. Also, not known is the ability of the Neurolite findings to distinguish between a stroke and pre-existing CNS lesions. Neurolite should not be used for these purposes.
How Supplied
- Lantheus Medical Imaging, Inc. Neurolite® Kit for the Preparation of Technetium Tc99m Bicisate for Injection, is supplied in kits of two (2) vials of A and two (2) vials of B (NDC # 11994-006-02); and five (5) vials of A and five (5) vials of B (NDC 11994-006-05). Included in each kit are one (1) package insert and twelve (12) radiation labels.
Storage
- Prior to reconstitution, vial A and vial B are stored 15-25°C. Protect vial A from light.
- Store at controlled room temperature after preparation.
- Use within 6 hours of preparation.
Images
Drug Images
{{#ask: Page Name::Bicisate dihydrochloride |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel


{{#ask: Label Page::Bicisate dihydrochloride |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Bicisate dihydrochloride in the drug label.
Precautions with Alcohol
- Alcohol-Bicisate dihydrochloride interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- NEUROLITE®[1]
Look-Alike Drug Names
- A® — B®[2]
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "bicisate dihydrochloride".
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Page Name=Bicisate dihydrochloride
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Bicisate dihydrochloride |Label Name=Bicisate dihydrochloride11.png
}}
{{#subobject:
|Label Page=Bicisate dihydrochloride |Label Name=Bicisate dihydrochloride11.png
}}