Allotropes of phosphorus
Elemental phosphorus can exist in several allotropes; the most common of which are white and red. There are also violet and black phosphorus, and gaseous diphosphorus.
White phosphorus
White phosphorus, or yellow phosphorus, or simply tetraphosphorus (P4) exists as individual molecules made up of four atoms in a tetrahedral arrangement, resulting in very high ring strain and instability. It contains 6 single bonds. Two different crystalline forms of white phosphorus are known. The α form, which is stable under standard conditions, has a body-centered cubic crystal structure. It transforms reversibly into the β form at 195.2 K. The β form is believed to have a hexagonal crystal structure.[1]
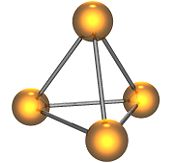
White phosphorus is a transparent waxy solid that quickly becomes yellow when exposed to light. For this reason it is also called yellow phosphorus. It glows greenish in the dark (when exposed to oxygen), is highly flammable and pyrophoric (self-igniting) upon contact with air as well as toxic (causing severe liver damage on ingestion). The odour of combustion of this form has a characteristic garlic smell, and samples are commonly coated with white "(di)phosphorus pentoxide", which consists of P4O10 tetrahedra with oxygen inserted between the phosphorus atoms and at their vertices. White phosphorus is insoluble in water but soluble in carbon disulfide.
The white allotrope can be produced using several different methods. In one process, calcium phosphate, which is derived from phosphate rock, is heated in an electric or fuel-fired furnace in the presence of carbon and silica.[2] Elemental phosphorus is then liberated as a vapour and can be collected under phosphoric acid. This process is similar to the first synthesis of phosphorus from calcium phosphate in urine.
White phosphorus is a soft, white wax-like solid, with an appreciable vapour pressure at ordinary temperatures. The vapour density indicates that the vapour is composed of P4 molecules up to about 800°C. Above that temperature, dissociation into P2 molecules occurs. The P4 molecule is tetrahedral, with each phosphorus atom being at the apices of the tetrahedron.
It ignites spontaneously at about 50°C, and at much lower temperatures if finely divided. White phosphorus is only slightly soluble in water and, indeed, it can be stored under water. However, it is soluble in benzene, oils, carbon disulfide and sulfur monochloride. The Relative Molecular Mass in solution corresponds to that expected for P4 molecules.
White phosphorus is chemically very reactive, and will spontaneously ignite in an abundant supply of air to give phosphorus (V) oxide:
P
4 + 5O
2 -> P
4O
10
Red phosphorus
Red phosphorus may be formed by heating white phosphorus to 250°C (482°F) or by exposing white phosphorus to sunlight. Phosphorus after this treatment exists as an amorphous network of atoms which reduces strain and gives greater stability; further heating results in the red phosphorus becoming crystalline. Red phosphorus does not catch fire in air at temperatures below 240°C, whereas white phosphorus ignites at about 30°C.
In 1865 Hittorf discovered that when phosphorus was recrystallized from molten lead, a red/purple form is obtained. This purple form is sometimes known as "Hittorf's phosphorus." In addition, a fibrous form exists with similar phosphorus cages. Below is shown a chain of phosphorus atoms which exhibits both the purple and fibrous forms.
Red phosphorus is much less reactive than white phosphorus, as evidenced by:
P
white -> P
violet ΔH = -16.8 J
It does not ignite in air until heated to 300°C, and it is insoluble in water, alkali and carbon disulfide. It does not react so readily with the halogens, but can be oxidised by nitric acid to phosphoric acid.
If it is heated in an atmosphere of inert gas, for example nitrogen or carbon dioxide, it sublimes and the vapour condenses as white phosphorus. If, however, it is heated in a vacuum and the vapour condensed rapidly, violet phosphorus is obtained. It would appear that violet phosphorus is a polymer of high Relative Molecular Mass, which on heating breaks down into P2 molecules. On cooling, these would normally dimerize to give P4 molecules (i.e. white phosphorus) but, in vacuo, they link up again to form the polymeric violet allotrope.
One of the forms of red/black phosphorus is a cubic solid.[3]
Violet phosphorus
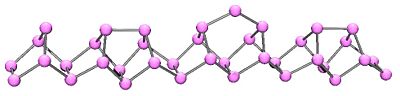
Monoclinic phosphorus, or violet phosphorus, is also known as "Hittorf`s Metallic Phosphorus".[4][5] In 1865 Hittorf heated red phosphorus in a sealed tube at 530 °C. The upper part of the tube was kept at 444 °C. Brilliant opaque monoclinic, or rhombohedral, crystals sublime. Can also be prepared by dissolving white phosphorus in molten lead in a sealed tube at 500 °C for 18 hours. Upon slow cooling, Hittorf's allotrope crystallises out. The crystals can be revealed by dissolving the lead in dilute nitric acid followed by boiling in concentrated hydrochloric acid.
Violet phosphorus was obtained by Johann Wilhelm Hittorf by heating white phosphorus at 550 °C for two weeks. He obtained a polymer that was insoluble in CS2. The crystal structure was obtained in 1960 at the University of Stuttgart by Herbert Thurn.
Black phosphorus
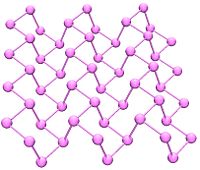
Black phosphorus is the thermodynamically stable form of phosphorus at room temperature and pressure. It is obtained by heating white phosphorus under very high pressures (12,000 atmospheres) In appearance, properties and structure it is very like graphite, being black and flaky, a conductor of electricity and has puckered sheets of linked atoms.
Black phosphorus has an orthorhombic structure (Cmca) and is the least reactive allotrope, it consists of many six-membered rings which are interlinked. Each atom is bonded to three other atoms.[6][7] A recent synthesis of black phosphorus using metal salts as catalysts has been reported.[8]
Diphosphorus
The diphosphorus allotrope (P2) can be obtained normally only under extreme conditions (for example, from P4 at 1100 kelvin). Nevertheless, some advancements were obtained in generating the diatomic molecule in homogenous solution, under normal condtitions with the use by some transitional metal complexes (based on for example tungsten and niobium).[9][10]
In fact, diphosphorus is the gaseous form of phosphorus, and the thermodynamically stable form above 1200 °C and until 2000 °C. The dissociation of tetraphosphorus (P
4) begins at lower temperature: the percentage of P
2 at 800 °C is ~ 1 %. Around 2000 °C the diphosphorus molecule begins to dissociate into atomic phosphorus.
Phosphorus nanorods
Phosphorus nanorods were synthesized as -
P−
12 polymers in Germany in August 2004 in two modifications.[11][12]
The red-brown phase is different from red phosphorus and is also stable in air for weeks. Electron microscope showed the red-brown form as having long, parallel nanorods with a diameter between 0,34 nm and 0,47 nm.
Other
Scarlet phosphorus
Another form, Schenk's scarlet phosphorus, is obtained by allowing a solution of white phosphorus in carbon disulfide to evaporate in sunlight.[13] It was first characterised by Rudolf Schenck in 1902 by heating white phosphorus in phosphorus tribromide (PBr
3)). It might include some bromine and has a density of about 1.88 g/cm3.
References
- ↑ Marie-Thérèse Averbuch-Pouchot, A. Durif. Topics in Phosphate Chemistry. World Scientific, 1996. ISBN 9810226349. p. 3.
- ↑ Threlfall, R.E., (1951). 100 years of Phosphorus Making: 1851 - 1951. Oldbury: Albright and Wilson Ltd
- ↑ R. Ahuja, Physica Status Solidi, Sectio B: Basic Research, 2003, 235, 282-287
- ↑ Lateral Science - Phosphorus Topics
- ↑ Monoclinic phosphorus formed from vapor in the presence of an alkali metal - Patent 4620968
- ↑ A. Brown, S. Runquist, Acta Crystallogr., 19 (1965) 684
- ↑ Cartz, L.;Srinivasa, S.R.;Riedner, R.J.;Jorgensen, J.D.;Worlton, T.G., Journal of Chemical Physics, 1979, 71, 1718-1721
- ↑ Stefan Lange, Peer Schmidt, and Tom Nilges, Inorganic Chemistry, 2007, 46, 4028
- ↑ Triple-Bond Reactivity of Diphosphorus Molecules Nicholas A. Piro, Joshua S. Figueroa, Jessica T. McKellar, Christopher C. Cummins Science 1 September 2006:Vol. 313. no. 5791, pp. 1276 - 1279 10.1126/science.1129630
- ↑ http://www.sciencemag.org/cgi/reprint/313/5791/1276.pdf
- ↑ [1]
- ↑ Arno Pfitzner, Michael F. Bräu, Josef Zweck, Gunther Brunklaus und Hellmut Eckert: Phosphorus Nanorods - Two Allotropic Modifications of a Long-Known Element in Angewandte Chemie International Edition 43 (2004), 4228 - 4231 doi:10.1002/anie.200460244
- ↑ Lateral Science - Phosphorus Topics