Creutzfeldt-Jakob disease pathophysiology: Difference between revisions
No edit summary |
|||
| (24 intermediate revisions by 8 users not shown) | |||
| Line 1: | Line 1: | ||
__NOTOC__ | __NOTOC__ | ||
{{Creutzfeldt-Jakob disease}} | {{Creutzfeldt-Jakob disease}} | ||
{{CMG}} | {{CMG}} {{AE}}, {{MMJ}} | ||
==Overview== | |||
The majority of cases of Creutzfeldt-Jakob disease are thought to occur sporadically from [[prions]] by an unknown route of transmission. Reports on transmission by [[human growth hormone]] products, [[grafting]], [[Electrode|surgical electrode implantation]], and consumption of infected products have been described. Once transmitted, the CJD [[prion]] promotes refolding of the native proteins into the diseased state. The number of misfolded [[protein]] molecules increases exponentially, and the process leads to a large quantity of insoluble [[prions]] in the affected cells, resulting in [[cell death]]. On gross histopathological analysis, the brain tissue appears "spongy" with areas of perforation within the brain tissue. On microscopic histopathological analysis, spongiform changes, neuronal loss, [[astrocyte]] proliferation, and deposition of [[prion]] proteins in the brain are characteristic findings. | |||
==Pathophysiology== | ==Pathophysiology== | ||
The defective protein can be transmitted | ===Transmission=== | ||
* The majority of cases of Creutzfeldt-Jakob disease are thought to occur sporadically from prions by an unknown route of transmission. | |||
* The defective protein associated with Creutzfeldt-Jakob disease can be inherited (familial form) or transmitted ([[Iatrogenesis|iatrogenic]] form) through the following: | |||
** [[Human growth hormone]] (hGH) products | |||
** [[Corneal grafting]] | |||
** Dural grafts or [[electrode]] implants<ref>{{cite web|title =Questions and Answers: Creutzfeldt-Jakob Disease Infection-Control Practices|work =Infection Control Practices/CJD (Creutzfeldt-Jakob Disease, Classic)|publisher =Centers for Disease Control and Prevention|date =[[January 4]][[2007]]|url = http://www.cdc.gov/ncidod/dvrd/cjd/qa_cjd_infection_control.htm#sterilization|accessdate = 2007-06-09}}</ref><ref>{{cite web|title =WHO Infection Control Guidelines for Transmissible Spongiform Encephalopathies|Publisher =World Health Organization: Communicable Disease Surveillance and Control|date =[[26 March]] [[1999]]|url =http://www.who.int/csr/resources/publications/bse/WHO_CDS_CSR_APH_2000_3/en/|accessdate = 2007-06-09}}</ref><ref>{{cite journal |author=McDonnell G, Burke P. | title=The challenge of prion decontamination | journal=Clin Infect Dis | year=2003 | volume=36 | pages=1152–4 }}</ref> | |||
** Use of [[Human growth hormone|HGH]] drawn from the [[pituitary gland]]s of [[cadaver]]s<ref name="titleHGH Linked to Brain Eater">{{cite web |url=http://www.wired.com/news/medtech/0,1286,62998,00.html |title=HGH Linked to Brain Eater |accessdate=2007-12-02 |format= |work=}}</ref> | |||
** Consumption of infected animals | |||
** Cannibalism | |||
*The incubation period of prions is unknonw, but it is speculated that the disease may develop many years (up to 30-50 years) after initial exposure. However, these reports remain controversial.<ref>{{cite web|url=http://www.nature.com/nature/journal/v407/n6800/full/407025a0.html|title=Diamond, J.M. (2000)"Archaeology: Talk of cannibalism" ''Nature'' 407, 25-26<!--INSERT TITLE-->}}</ref> | |||
=== Pathogenesis === | |||
* The CJD prion promotes refolding of the native [[proteins]] into the diseased state. | |||
* The number of misfolded [[protein]] molecules increases exponentially, and the process leads to a large quantity of insoluble [[prions]] in the affected cells. | |||
* This mass of misfolded [[protein]]s disrupts cell function and causes cell death. | |||
* Once the prion is transmitted, the defective proteins invade the brain and are produced in a self-sustaining [[feedback loop]], causing exponential spread of the prion. | |||
[[cadaver|<nowiki/>]] | |||
===Mutations=== | |||
* [[Mutations]] in the gene responsible for the production of prion protein can cause misfolding of the alpha helical regions into beta pleated sheets. | |||
* This change in the conformation disables the ability of the protein to undergo digestion. | |||
* Individuals may also acquire CJD genetically through the [[mutation]] occurring [[PRNP|''PRNP'']] gene. | |||
== Pathology == | |||
===Gross Pathology=== | |||
* On gross histopathological analysis, the brain tissue appears "spongy" with areas of perforation within the brain tissue. | |||
=== Microscopic Pathology === | |||
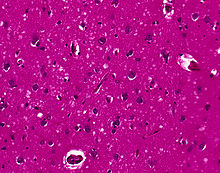
* CJD is associated with the build-up of abnormal [[prion]] [[proteins]]. | |||
* The following microscopic findings may be present in CJD: | |||
** Presence of florid plaques, defined as round, packed deposits of abnormal prion protein (in variant CJD) | |||
** Spongiform changes | |||
** Neuronal loss | |||
** [[Astrocyte]] proliferation | |||
** Deposition of prion protein throughout the brain<ref name="Sellars-2002">{{Cite journal | last1 = Sellars | first1 = RJ. | last2 = Collie | first2 = DA. | last3 = Will | first3 = RJ. | title = Progress in understanding Creutzfeldt-Jakob disease. | journal = AJNR Am J Neuroradiol | volume = 23 | issue = 7 | pages = 1070-2 | month = Aug | year = 2002 | doi = | PMID = 12169459 }}</ref> | |||
[[ | [[Image:Spongiform changes in CJD.jpg|left|220px|Spongiform changes in brain in CJD]] | ||
<br style="clear:left"> | |||
= | |||
==References== | ==References== | ||
| Line 25: | Line 52: | ||
{{WikiDoc Sources}} | {{WikiDoc Sources}} | ||
[[Category:Neurology]] | [[Category:Neurology]] | ||
[[Category:Disease]] | [[Category:Disease]] | ||
[[Category:Transmissible spongiform encephalopathies]] | [[Category:Transmissible spongiform encephalopathies]] | ||
[[Category:Needs overview]] | [[Category:Needs overview]] | ||
Latest revision as of 19:44, 7 July 2020
|
Creutzfeldt-Jakob disease Microchapters |
|
Differentiating Creutzfeldt-Jakob disease from other Diseases |
|---|
|
Diagnosis |
|
Treatment |
|
Case Studies |
|
Creutzfeldt-Jakob disease pathophysiology On the Web |
|
American Roentgen Ray Society Images of Creutzfeldt-Jakob disease pathophysiology |
|
Risk calculators and risk factors for Creutzfeldt-Jakob disease pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1] Associate Editor(s)-in-Chief: , Mohamadmostafa Jahansouz M.D.[2]
Overview
The majority of cases of Creutzfeldt-Jakob disease are thought to occur sporadically from prions by an unknown route of transmission. Reports on transmission by human growth hormone products, grafting, surgical electrode implantation, and consumption of infected products have been described. Once transmitted, the CJD prion promotes refolding of the native proteins into the diseased state. The number of misfolded protein molecules increases exponentially, and the process leads to a large quantity of insoluble prions in the affected cells, resulting in cell death. On gross histopathological analysis, the brain tissue appears "spongy" with areas of perforation within the brain tissue. On microscopic histopathological analysis, spongiform changes, neuronal loss, astrocyte proliferation, and deposition of prion proteins in the brain are characteristic findings.
Pathophysiology
Transmission
- The majority of cases of Creutzfeldt-Jakob disease are thought to occur sporadically from prions by an unknown route of transmission.
- The defective protein associated with Creutzfeldt-Jakob disease can be inherited (familial form) or transmitted (iatrogenic form) through the following:
- Human growth hormone (hGH) products
- Corneal grafting
- Dural grafts or electrode implants[1][2][3]
- Use of HGH drawn from the pituitary glands of cadavers[4]
- Consumption of infected animals
- Cannibalism
- The incubation period of prions is unknonw, but it is speculated that the disease may develop many years (up to 30-50 years) after initial exposure. However, these reports remain controversial.[5]
Pathogenesis
- The CJD prion promotes refolding of the native proteins into the diseased state.
- The number of misfolded protein molecules increases exponentially, and the process leads to a large quantity of insoluble prions in the affected cells.
- This mass of misfolded proteins disrupts cell function and causes cell death.
- Once the prion is transmitted, the defective proteins invade the brain and are produced in a self-sustaining feedback loop, causing exponential spread of the prion.
Mutations
- Mutations in the gene responsible for the production of prion protein can cause misfolding of the alpha helical regions into beta pleated sheets.
- This change in the conformation disables the ability of the protein to undergo digestion.
- Individuals may also acquire CJD genetically through the mutation occurring PRNP gene.
Pathology
Gross Pathology
- On gross histopathological analysis, the brain tissue appears "spongy" with areas of perforation within the brain tissue.
Microscopic Pathology
- The following microscopic findings may be present in CJD:
References
- ↑ "Questions and Answers: Creutzfeldt-Jakob Disease Infection-Control Practices". Infection Control Practices/CJD (Creutzfeldt-Jakob Disease, Classic). Centers for Disease Control and Prevention. January 42007. Retrieved 2007-06-09. Check date values in:
|date=(help) - ↑ "WHO Infection Control Guidelines for Transmissible Spongiform Encephalopathies". 26 March 1999. Retrieved 2007-06-09. Unknown parameter
|Publisher=ignored (|publisher=suggested) (help); Check date values in:|date=(help) - ↑ McDonnell G, Burke P. (2003). "The challenge of prion decontamination". Clin Infect Dis. 36: 1152&ndash, 4.
- ↑ "HGH Linked to Brain Eater". Retrieved 2007-12-02.
- ↑ "Diamond, J.M. (2000)"Archaeology: Talk of cannibalism" Nature 407, 25-26".
- ↑ Sellars, RJ.; Collie, DA.; Will, RJ. (2002). "Progress in understanding Creutzfeldt-Jakob disease". AJNR Am J Neuroradiol. 23 (7): 1070–2. PMID 12169459. Unknown parameter
|month=ignored (help)