Pralidoxime
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Alberto Plate [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Pralidoxime is a cholinesterase reactivator that is FDA approved for the treatment of of poisoning by nerve agents having anticholinesterase activity. Common adverse reactions include Increased creatine kinase level.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Poisoning by Nerve Agents Having Anticholinesterase Activity
- Mild cases: headache, blurred vision, mild muscarinic signs
- Moderately Severe Cases: excessive sweating, lacrimation, salivation, diarrhea, tightness in the chest
- Very severe cases: Cyanosis, Respiratory Embarrassment, Coma
- For optimal reactivation of organophosphate-inhibited cholinesterase, atropine and pralidoxime should be administered as soon as possible after exposure. Depending on the severity of symptoms, immediately administer one atropine-containing auto-injector, followed by one pralidoxime-containing auto-injector. Atropine must be given first until its effects become apparent and only then should pralidoxime be administered. If nerve agent symptoms are still present after 15 minutes, repeat injections. If symptoms still exist after an additional 15 minutes, repeat injections for a third time. If after the third set of injections, symptoms remain, do not give any more antidotes but seek medical help.
Directions for Use
When, as described above, auto-injector use is indicated, proceed as follows:

- 1.- Remove gray safety cap.
- 2.- Place black end against outer thigh and push hard until the injector functions.
- 3.- Hold firmly in place for ten seconds, then remove. Massage the area of injection.
- 4.- Dispose of properly. Push ejected needle through a pocket flap (or other thick and conspicuous part of outer clothing). Bend needle into a hook.
- Initial measures should include removal of secretions, maintenance of a patent airway and, if necessary, artificial ventilation. Atropine should not be used until cyanosis has been overcome since atropine produces ventricular fibrillations in the presence of hypoxia. Morphine, theophylline, aminophylline, or succincylcholine are contraindicated. Tranquilizers of the reserpine or phenothiazine type are to be avoided.
Pralidoxime is most effective if administered immediately after poisoning. Generally, little is accomplished if the drug is given more than 36 hours after termination of exposure. When the poison has been ingested, however, exposure may continue for some time due to slow absorption from the lower bowel, and fatal relapses have been reported after initial improvement. Continued administration for several days may be useful in such patients. Close supervision of the patient is indicated for at least 48 to 72 hours. If dermal exposure has occurred, clothing should be removed and the hair and skin washed thoroughly with sodium bicarbonate or alcohol as soon as possible. Diazepam may be given cautiously if convulsions are not controlled by atropine.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Pralidoxime in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Pralidoxime in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding Pralidoxime FDA-Labeled Indications and Dosage (Pediatric) in the drug label.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Pralidoxime in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Pralidoxime in pediatric patients.
Contraindications
The pralidoxime chloride auto-injector is contraindicated in patients who are hypersensitive to any component of the product.
Warnings
- Pralidoxime is not effective in the treatment of poisoning due to phosphorus, inorganic phosphates or organophosphates not having anticholinesterase activity.
Adverse Reactions
Clinical Trials Experience
- Forty to 60 minutes after intramuscular injection, mild to moderate pain may be experienced at the site of injection. Pralidoxime may cause blurred vision, diplopia and impaired accommodation, dizziness, headache, drowsiness, nausea, tachycardia, increased systolic and diastolic blood pressure, hyperventilation, and muscular weakness when given parenterally to normal volunteers who have not been exposed to anticholinesterase poisons. In patients it is very difficult to differentiate the toxic effects produced by atropine or the organophosphate compounds from those of the drug.
- Elevations in SGOT and/or SGPT enzyme levels were observed in 1 of 6 normal volunteers given 1200 mg of pralidoxime chloride intramuscularly, and in 4 of 6 volunteers given 1800 mg intramuscularly. Levels returned to normal in about 2 weeks. Transient elevations in creatine phosphokinase were observed in all normal volunteers given the drug. A single intramuscular injection of 330 mg in 1 mL in rabbits caused myonecrosis, inflammation and hemorrhage.
- When atropine and pralidoxime are used together, the signs of atropinization may occur earlier than might be expected when atropine is used alone. This is especially true if the total dose of atropine has been large and the administration of pralidoxime has been delayed.2, 3, 4 Excitement and manic behavior immediately following recovery of consciousness have been reported in several cases. However, similar behavior has occurred in cases of organophosphate poisoning that were not treated with pralidoxime
Postmarketing Experience
There is limited information regarding Pralidoxime Postmarketing Experience in the drug label.
Drug Interactions
- When atropine and pralidoxime are used together, the signs of atropinization (flushing, mydriasis, tachycardia, dryness of the mouth and nose) may occur earlier than might be expected when atropine is used alone. This is especially true if the total dose of atropine has been large and the administration of pralidoxime has been delayed.
- The following precautions should be kept in mind in the treatment of anticholesterinase poisoning, although they do not bear directly on the use of pralidoxime; since barbiturates are potentiated by the anticholinesterases, they should be used cautiously in the treatment of convulsions; morphine, theophylline, aminophylline, succinylcholine, reser-pine, and phenothiazine-type tranquilizers should be avoided in patients with organophosphate poisoning.
Use in Specific Populations
Pregnancy
- Animal reproduction studies have not been conducted with pralidoxime. It is also not known whether pralidoxime can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Pralidoxime should be given to a pregnant woman only if clearly needed.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Pralidoxime in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Pralidoxime during labor and delivery.
Nursing Mothers
- It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when pralidoxime is administered to a nursing woman.
Pediatric Use
- Safety and effectiveness in children have not been established.
Geriatic Use
There is no FDA guidance on the use of Pralidoxime in geriatric settings.
Gender
There is no FDA guidance on the use of Pralidoxime with respect to specific gender populations.
Race
There is no FDA guidance on the use of Pralidoxime with respect to specific racial populations.
Renal Impairment
- Because pralidoxime is excreted in the urine, a decrease in renal function will result in increased blood levels of the drug. Thus, the dosage of pralidoxime should be reduced in the presence of renal insufficiency.
Hepatic Impairment
There is no FDA guidance on the use of Pralidoxime in patients with hepatic impairment.
Females of Reproductive Potential and Males
- Since the pralidoxime chloride auto-injector is indicated for short-term emergency use only, no investigations of its potential for carcinogenesis, mutagenesis, or impairment of fertility have been conducted by the manufacturer, or reported in the literature.
Immunocompromised Patients
There is no FDA guidance one the use of Pralidoxime in patients who are immunocompromised.
Administration and Monitoring
Administration
There is limited information regarding Pralidoxime Administration in the drug label.
Monitoring
There is limited information regarding Pralidoxime Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Pralidoxime and IV administrations.
Overdosage
Manifestations of Overdosage
- Observed in normal subjects only: dizziness, blurred vision, diplopia, headache, impaired accommodation, nausea, slight tachycardia. In therapy it has been difficult to differentiate side effects due to the drug from those due to the effects of the poison.
Treatment of Overdosage
- Artificial respiration and other supportive therapy should be administered as needed.
Acute Toxicity
- i.v.—man TDLo: 14 mg/kg (toxic effects: CNS)
- i.v.— rat LD50: 96 mg/kg
- i.m.—rat LD50: 150 mg/kg
- oral—mouse LD50: 4100 mg/kg
- i.p.—mouse LD50: 155 mg/kg
- i.v.—mouse LD50: 90 mg/kg
- i.m.—mouse LD50: 180 mg/kg
- i.v.—rabbit LD50: 95 mg/kg
- i.m.—guinea pig LD50: 168 mg/kg
Pharmacology
| |
Pralidoxime
| |
| Systematic (IUPAC) name | |
| 2-[(hydroxyimino)methyl]-1-methylpyridin-1-ium | |
| Identifiers | |
| CAS number | |
| ATC code | V03 |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox+ |
| Mol. mass | 137.159 g/mol |
| SMILES | & |
| Synonyms | 1-methylpyridine-6-carbaldehyde oxime |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
C |
| Legal status |
RX-only |
| Routes | ? |
Mechanism of Action
- The principal action of pralidoxime is to reactivate cholinesterase (mainly outside of the central nervous system) which has been inactivated by phosphorylation due to an organophosphate pesticide or related compound. The destruction of accumulated acetylcholine can then proceed and neuromuscular junctions will again function normally. Pralidoxime also slows the process of "aging" of phosphorylated cholinesterase to a non-reactivatable form, and detoxifies certain organophosphates by direct chemical reaction. The drug has its most critical effect in relieving paralysis of the muscles of respiration.
Structure
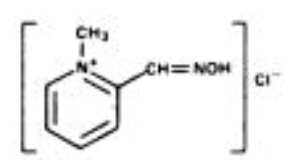
- Chemical Name: 2-formyl-1 methylpyridinium chloride oxime (pyridine-2-aldoxime methochloride). Also referred to as 2-PAM Chloride.
- Structural Formula:

Pharmacodynamics
- Because pralidoxime is less effective in relieving depression of the respiratory center, atropine is always required concomitantly to block the effect of accumulated acetylcholine at this site. Pralidoxime relieves muscarinic signs and symptoms, salivation, bronchospasm, etc., but this action is relatively unimportant since atropine is adequate for this purpose.
Pharmacokinetics
- Pralidoxime is distributed throughout the extracellular water, it is not bound to plasma protein. The drug is rapidly excreted in the urine partly unchanged, and partly as a metabolite produced by the liver. Consequently, pralidoxime is relatively short acting and repeated doses may be needed, especially where there is any evidence of continuing absorption of the poison.
The minimum therapeutic concentration of pralidoxime in plasma is 4 μg/mL, this level is reached in about 16 minutes after a single injection of 600 mg pralidoxime chloride. The apparent half-life of pralidoxime chloride is 74-77 minutes.
It has been reported that the supplemental use of oxime cholinesterase reactivators (such as pralidoxime) reduces the incidence and severity of developmental defects in chick embryos exposed to such known teratogens as parathion, bidrin, carbachol and neostigmine. This protective effect of the oximes was shown to be dose related.
Nonclinical Toxicology
There is limited information regarding Pralidoxime Nonclinical Toxicology in the drug label.
Clinical Studies
There is limited information regarding Pralidoxime Clinical Studies in the drug label.
How Supplied
- Pralidoxime chloride is supplied in aqueous solution prefilled in the auto-injector (600 mg, 2 mL) for military use and for use by qualified civilian emergency responders. Auto-injectors are supplied through the Directorate of Medical Materiel, Defense Supply Center Philadelphia or other analogous local, state or federal agency.
- When activated, each auto-injector dispenses 600 mg of pralidoxime chloride in 2 mL of a sterile solution containing 20 mg/mL benzyl alcohol, 11.26 mg/mL glycine in Water for Injection, USP. The pH is adjusted with hydrochloric acid. *The pH is 2.0-3.0
- The product is pyrogen free.
Storage
- Store at 25°C (77°F); Excursions permitted to 15-30°C (59-86°F).
Images
Drug Images
{{#ask: Page Name::Pralidoxime |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel

{{#ask: Label Page::Pralidoxime |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Pralidoxime Patient Counseling Information in the drug label.
Precautions with Alcohol
- Alcohol-Pralidoxime interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
Look-Alike Drug Names
There is limited information regarding Pralidoxime Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.