Methylnaltrexone bromide
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Kiran Singh, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Methylnaltrexone bromide is an opioid antagonist that is FDA approved for the treatment of opioid-induced constipation (OIC) in adult patients with chronic non‑cancer pain and with advanced illness who are receiving palliative care, when response to laxative therapy has not been sufficient. Common adverse reactions include abdominal pain, nausea, diarrhea, hyperhidrosis, hot flush, tremor, chills,flatulence, and dizziness.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
Opioid-Induced Constipation in Adult Patients with Chronic Non-Cancer Pain
- RELISTOR is indicated for the treatment of opioid-induced constipation in adult patients with chronic non‑cancer pain.
Opioid-Induced Constipation in Adult Patients with Advanced Illness
- RELISTOR is indicated for the treatment of opioid-induced constipation in adult patients with advanced illness who are receiving palliative care, when response to laxative therapy has not been sufficient.
Limitation of Use
- Use of RELISTOR beyond four months has not been studied in the advanced illness population.
Dosage
Opioid-Induced Constipation in Adult Patients with Chronic Non-Cancer Pain
- The recommended dosage of RELISTOR is 12 mg subcutaneously once daily.
Opioid-Induced Constipation in Adult Patients with Advanced Illness
- The recommended dose of RELISTOR administered subcutaneously is 8 mg for adult patients weighing 38 kg to less than 62 kg and 12 mg for patients weighing 62 kg to 114 kg. Adult patients whose weight falls outside of these ranges should be dosed at 0.15 mg/kg. The recommended dosage regimen is one dose every other day, as needed.
- See Table 1 to determine the correct dose by patient weight and injection volume to be administered. The pre-filled syringe is designed to deliver a fixed dose; therefore, adult patients requiring dosing calculated on a mg/kg basis should not be prescribed pre-filled syringes.

Use in Patients with Severe Renal Impairment
- In adult patients with severe renal impairment (creatinine clearance less than 30 mL/min as estimated by Cockcroft-Gault), dose reduction of RELISTOR by one-half is recommended [see Use in Specific Populations (8.6)]. No dosage adjustment is recommended for adult patients with mild to moderate renal impairment.
- The pre-filled syringe is designed to deliver a fixed dose; therefore, adult patients with severe renal impairment should only be prescribed single-use vials to ensure correct dosing.
DOSAGE FORMS AND STRENGTHS
Single-use Vial:
- 12 mg/0.6 mL solution for subcutaneous injection, for use with a 27 gauge x ½-inch needle and 1 mL syringe
- 12 mg/0.6 mL solution for subcutaneous injection, with one 1 mL syringe with retractable 27 gauge x ½-inch needle, two alcohol swabs
Single-use Pre-filled Syringe:
- 8 mg/0.4 mL solution for subcutaneous injection, with a 29-gauge x ½-inch fixed needle and a needle guard
- 12 mg/0.6 mL solution for subcutaneous injection, with a 29-gauge x ½-inch fixed needle and a needle guard
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Methylnaltrexone bromide in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Methylnaltrexone bromide in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Methylnaltrexone bromide in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Methylnaltrexone bromide in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Methylnaltrexone bromide in pediatric patients.
Contraindications
- RELISTOR is contraindicated in patients with known or suspected gastrointestinal obstruction and patients at increased risk of recurrent obstruction, due to the potential for gastrointestinal perforation
Warnings
Gastrointestinal Perforation
- Cases of gastrointestinal perforation have been reported in adult patients with opioid-induced constipation and advanced illness with conditions that may be associated with localized or diffuse reduction of structural integrity in the wall of the gastrointestinal tract (e.g., peptic ulcer disease, Ogilvie’s syndrome, diverticular disease, infiltrative gastrointestinal tract malignancies or peritoneal metastases). Take into account the overall risk-benefit profile when using RELISTOR in patients with these conditions or other conditions which might result in impaired integrity of the gastrointestinal tract wall (e.g., Crohn’s disease). Monitor for the development of severe, persistent, or worsening abdominal pain; discontinue RELISTOR in patients who develop this symptom.
Severe or Persistent Diarrhea
- If severe or persistent diarrhea occurs during treatment, advise patients to discontinue therapy with RELISTOR and consult their healthcare provider.
Opioid Withdrawal
- Symptoms consistent with opioid withdrawal, including hyperhidrosis, chills, diarrhea, abdominal pain, anxiety, and yawning have occurred in patients treated with RELISTOR. Patients having disruptions to the blood-brain barrier may be at increased risk for opioid withdrawal and/or reduced analgesia. Take into account the overall risk-benefit profile when using RELISTOR in such patients. Monitor for adequacy of analgesia and symptoms of opioid withdrawal in such patients.
Adverse Reactions
Clinical Trials Experience
Serious and important adverse reactions described elsewhere in labeling include:
- Gastrointestinal perforation
- Severe or persistent diarrhea
- Opioid withdrawal
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Opioid-Induced Constipation in Adult Patients with Chronic Non-Cancer Pain
- The safety of RELISTOR was evaluated in a double-blind, placebo-controlled trial in adult patients with opioid-induced constipation and chronic non-cancer pain receiving opioid analgesia. This study (Study 1) included a 4-week, double-blind, placebo-controlled period in which adult patients were randomized to receive RELISTOR 12 mg once daily (150 patients) or placebo (162 patients). After 4 weeks of double-blind treatment, patients began an 8-week open-label treatment period during which RELISTOR 12 mg was administered less frequently than the recommended dosage regimen of 12 mg once daily.
- Adverse reactions in adult patients with opioid-induced constipation and chronic non-cancer pain receiving RELISTOR are shown in Table 2. The adverse reactions in the table below may reflect symptoms of opioid withdrawal.

- During the 4-week double-blind period, in patients with opioid-induced constipation and chronic non-cancer pain that received RELISTOR 12 mg every other day, there was a higher incidence of adverse reactions, including nausea (12%), diarrhea (12%), vomiting (7%), tremor (3%), feeling of body temperature change (3%), piloerection (3%), and chills (2%) as compared to daily Relistor dosing. Use of RELISTOR 12 mg every other day is not recommended in patients with OIC and chronic non-cancer pain. The rates of discontinuation due to adverse reactions during the double-blind period (Study 1) were higher in the RELISTOR once daily (7%) than the placebo group (3%). Abdominal pain was the most common adverse reaction resulting in discontinuation from the double-blind period in the RELISTOR once daily group (2%).
- The safety of RELISTOR was also evaluated in a 48-week, open-label, uncontrolled trial in 1034 adult patients with opioid-induced constipation and chronic non-cancer pain (Study 2). Patients were allowed to administer RELISTOR 12 mg less frequently than the recommended dosage regimen of 12 mg once daily, and took a median of 6 doses per week. A total of 624 patients (60%) completed at least 24 weeks of treatment and 477 (46%) completed the 48‑week study. The adverse reactions seen in this study were similar to those observed during the 4-week double-blind period of Study 1. Additionally, in Study 2, investigators reported 4 myocardial infarctions (1 fatal), 1 stroke (fatal), 1 fatal cardiac arrest and 1 sudden death. It is not possible to establish a relationship between these events and RELISTOR.
Opioid-Induced Constipation in Adult Patients with Advanced Illness
- The safety of RELISTOR was evaluated in two, double-blind, placebo-controlled trials in adult patients with opioid-induced constipation and advanced illness receiving palliative care: Study 3 included a single‑dose, double‑blind, placebo-controlled period, whereas Study 4 included a 14-day multiple dose, double-blind, placebo-controlled period.
- The most common (≥ 5%) adverse reactions in adult patients with opioid-induced constipation and advanced illness receiving RELISTOR are shown in Table 3 below.

- The rates of discontinuation due to adverse events during the double-blind placebo controlled clinical trials (Study 3 and Study 4) were comparable between RELISTOR (1%) and placebo (2%).
Postmarketing Experience
- The following adverse reactions have been identified during post-approval use of RELISTOR. Because they are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal
Perforation, cramping, vomiting
General Disorders and Administrative Site Disorders
Diaphoresis, flushing, malaise, pain. Cases of opioid withdrawal have been reported.
Drug Interactions
Other Opioid Anatagonists
- Avoid concomitant use of RELISTOR with other opioid antagonists because of the potential for additive effects of opioid receptor antagonism and increased risk of opioid withdrawal.
Drugs Metabolized by Cytochrome P450 Isozymes
- In healthy subjects, a subcutaneous dose of 0.30 mg/kg of methylnaltrexone did not significantly affect the metabolism of dextromethorphan, a CYP2D6 substrate.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): Pregnancy Category C
Risk Summary
- There are no adequate and well-controlled studies with RELISTOR in pregnant women. The use of RELISTOR during pregnancy may precipitate opioid withdrawal in a fetus due to the immature fetal blood brain barrier. In animal reproduction studies, no effects on embryo-fetal development were observed with the administration of intravenous methylnaltrexone during organogenesis in rats and rabbits at doses up to 20 times and 26 times, respectively, the maximum recommended human dose (MRHD) of 0.2 mg/kg/day. RELISTOR should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Data
Animal Data
- Reproduction studies have been performed with methylnaltrexone administered during the period of organogenesis to rats at intravenous doses up to 25 mg/kg/day (about 20 times the MRHD of 0.2 mg/kg/day based on body surface area) and did not cause any adverse effects on embryofetal development. In rabbits, intravenous doses of methylnaltrexone up to 16 mg/kg/day (about 26 times the MRHD of 0.2 mg/kg/day based on body surface area) did not show any embryofetal toxicity. A pre- and postnatal development study in rats showed no evidence of any adverse effect on pre- and postnatal development at subcutaneous doses of methylnaltrexone up to 100 mg/kg/day (about 81 times the MRHD of 0.2 mg/kg/day based on body surface area).
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Methylnaltrexone bromide in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Methylnaltrexone bromide during labor and delivery.
Nursing Mothers
- It is not known whether RELISTOR is present in human milk. However, methylnaltrexone bromide is present in rat milk. Because of the potential for serious adverse reactions, including opioid withdrawal, in nursing infants, a decision should be made to discontinue nursing or discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
- Safety and effectiveness of RELISTOR have not been established in pediatric patients.
- In juvenile rats administered intravenous methylnaltrexone bromide for 13 weeks, adverse clinical signs such as convulsions, tremors and labored breathing were observed, and the juvenile rats were found to be more sensitive to the adverse effects of methylnaltrexone bromide when compared to adult animals. Juvenile dogs administered intravenous methylnaltrexone bromide for 13 weeks had a toxicity profile similar to adult dogs
Geriatic Use
- In the double-blind studies, a total of 118 (14%) patients aged 65-74 years (79 methylnaltrexone bromide, 39 placebo) and a total of 108 (13%) patients aged 75 years or older (64 methylnaltrexone bromide, 44 placebo) were enrolled. No overall differences in safety or effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
- Based on pharmacokinetic data, and safety and efficacy data from controlled clinical trials, no dose adjustment based on age is recommended.
Gender
There is no FDA guidance on the use of Methylnaltrexone bromide with respect to specific gender populations.
Race
There is no FDA guidance on the use of Methylnaltrexone bromide with respect to specific racial populations.
Renal Impairment
- No dose adjustment is required in patients with mild or moderate renal impairment. Dose‑reduction by one half is recommended in patients with severe renal impairment (creatinine clearance less than 30 mL/min as estimated by Cockcroft-Gault).
Hepatic Impairment
- No dose adjustment is required for patients with mild or moderate hepatic impairment
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Methylnaltrexone bromide in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Methylnaltrexone bromide in patients who are immunocompromised.
Administration and Monitoring
Administration
- Subcutaneous
Important Administration Information
- RELISTOR is for subcutaneous use only.
- Inject RELISTOR subcutaneously in the upper arm, abdomen or thigh.Do not inject at the same spot each time (rotate injection sites).
- Be within close proximity to toilet facilities once RELISTOR is administered.
- Discontinue RELISTOR if treatment with the opioid pain medication is also discontinued.
- In patients with chronic non-cancer pain and opioid-induced constipation:
- RELISTOR has been shown to be efficacious in patients who have taken opioids for at least 4 weeks. Sustained exposure to opioids prior to starting RELISTOR may increase the patient’s sensitivity to the effects of RELISTOR.
- Discontinue all maintenance laxative therapy prior to initiation of RELISTOR. Laxative(s) can be used as needed if there is a suboptimal response to RELISTOR after three days.
- Inject one dose every day.
- Re-evaluate the continued need for RELISTOR when the opioid regimen is changed to avoid adverse reactions.
- In patients with advanced illness and opioid-induced constipation:
- Inject one dose every other day, as needed, but no more frequently than one dose in a 24-hour period.
Administration and Storage
- RELISTOR is a sterile, clear, and colorless to pale yellow aqueous solution. Inspect parenteral drug products visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not use the vial if any of these are present.
Single-use Vials
- Once drawn into the 1 mL syringe with a 27-gauge x ½-inch needle, if immediate administration is not possible, store at ambient room temperature and administer within 24 hours. Discard any unused portion that remains in the vial. Advise patients concerning proper training in subcutaneous technique.
Single-use Pre-filled Syringes
- Only adult patients requiring an 8 mg or 12 mg dose should be prescribed pre-filled syringes. Do not remove the pre-filled syringe from the tray until ready to administer.
Monitoring
There is limited information regarding Monitoring of Methylnaltrexone bromide in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Methylnaltrexone bromide in the drug label.
Overdosage
- During clinical trials of RELISTOR administered subcutaneously, no cases of methylnaltrexone bromide overdose were reported.
- A study of healthy volunteers noted orthostatic hypotension associated with a dose of 0.64 mg/kg administered as an intravenous bolus. Monitor for signs or symptoms of orthostatic hypotension and initiate treatment as appropriate.
- If a patient on opioid therapy receives an overdose of RELISTOR, the patient should be monitored closely for potential evidence of opioid withdrawal symptoms such as chills, rhinorrhea, diaphoresis or reversal of central analgesic effect. Base treatment on the degree of opioid withdrawal symptoms, including changes in blood pressure and heart rate, and on the need for analgesia.
Pharmacology
There is limited information regarding Methylnaltrexone bromide Pharmacology in the drug label.
Mechanism of Action
- Methylnaltrexone is a selective antagonist of opioid binding at the mu-opioid receptor. As a quaternary amine, the ability of methylnaltrexone to cross the blood-brain barrier is restricted. This allows methylnaltrexone to function as a peripherally-acting mu‑opioid receptor antagonist in tissues such as the gastrointestinal tract, thereby decreasing the constipating effects of opioids without impacting opioid-mediated analgesic effects on the central nervous system.
Structure
- RELISTOR (methylnaltrexone bromide) injection, a mu-opioid receptor antagonist, is a sterile, clear and colorless to pale yellow aqueous solution. The chemical name for methylnaltrexone bromide is (R)-N-(cyclopropylmethyl) noroxymorphone methobromide. The molecular formula is C21H26NO4Br, and the molecular weight is 436.36.
- Each 3 mL vial contains 12 mg of methylnaltrexone bromide in 0.6 mL of water. The excipients are 3.9 mg sodium chloride USP, 0.24 mg edetate calcium disodium USP, and 0.18 mg glycine hydrochloride. During manufacture, the pH may have been adjusted with hydrochloric acid and/or sodium hydroxide.
- Each 8 mg/0.4 mL pre-filled syringe (1 mL syringe) contains 8 mg of methylnaltrexone bromide in 0.4 mL of water. The excipients are 2.6 mg sodium chloride USP, 0.16 mg edetate calcium disodium USP, and 0.12 mg glycine hydrochloride.
- Each 12 mg/0.6 mL pre-filled syringe (1 mL syringe) contains 12 mg of methylnaltrexone bromide in 0.6 mL of water. The excipients are 3.9 mg sodium chloride USP, 0.24 mg edetate calcium disodium USP, and 0.18 mg glycine hydrochloride.
The structural formula is:

Pharmacodynamics
Cardiac Electrophysiology
- In a randomized, double-blind placebo- and (open-label) moxifloxacin-controlled 4‑period crossover study, 56 healthy subjects were administered methylnaltrexone bromide 0.3 mg/kg and methylnaltrexone bromide 0.64 mg/kg by intravenous infusion over 20 minutes, placebo, and a single oral dose of moxifloxacin. At a dose approximately 4.3 times the maximum recommended dose (7.5 times the mean peak plasma concentration), methylnaltrexone does not prolong the QTc interval to any clinically relevant extent.
Pharmacokinetics
Absorption
Following subcutaneous administration, methylnaltrexone achieved peak concentrations (Cmax) at approximately 0.5 hours. Across the range of doses from 0.15 mg/kg to 0.50 mg/kg, mean Cmax and area under the plasma concentration-time curve (AUC) increased in a dose-proportional manner. There was no accumulation of methylnaltrexone following once-daily subcutaneous dosing of methylnaltrexone bromide 12 mg for seven consecutive days in healthy subjects.

Distribution
- The steady-state volume of distribution (Vss) of methylnaltrexone is approximately 1.1 L/kg. The fraction of methylnaltrexone bound to human plasma proteins is 11.0% to 15.3%, as determined by equilibrium dialysis.
Elimination
- Following intravenous administration of 0.3 mg/kg, the total clearance of methylnaltrexone is approximately 10.5 ± 1.5 mL/min/kg, with renal clearance of 6.37 ± 3.0 mL/min/kg. The terminal half-life (t1/2) is approximately 8 hours.
Metabolism
- In a mass balance study, approximately 44% of the administered radioactivity was recovered in the urine over 24 hours with 5 distinct metabolites and none of the detected metabolites was in amounts over 6% of administered radioactivity. Conversion to methyl-6-naltrexol isomers (5% of total) and methylnaltrexone sulfate (1.3% of total) appear to be the primary pathways of metabolism. N‑demethylation of methylnaltrexone to produce naltrexone is not significant.
- After 12 mg once daily dosing the mean AUC0-24 ratio of metabolites to methylnaltrexone at steady-state was 30%, 19%, and 9% for methylnaltrexone sulfate, methyl-6a-naltrexol, and methyl-6b-naltrexol, respectively. Methyl-6a-naltrexol, and methyl-6b-naltrexol are active mu-opioid receptor antagonists and methylnaltrexone sulfate is a weak mu-opioid receptor antagonist.
- Methylnaltrexone is conjugated by sulfotransferase SULT1E1 and SULT2A1 isoforms to methylnaltrexone sulfate. Conversion to methyl-6-naltrexol isomers is mediated by aldo-keto reductase 1C enzymes.
Excretion
- After intravenous administration, approximately half of the dose was excreted in the urine (53.6%) and 17.3% of administered dose was excreted in the feces up to 168 hours postdose. Methylnaltrexone is excreted primarily as the unchanged drug in the urine and feces. Active renal secretion of methylnaltrexone is suggested by renal clearance of methylnaltrexone that is approximately 4-5 fold higher than creatinine clearance.
Specific Populations
Age: Geriatric Population
- A study was conducted to characterize the pharmacokinetics of methylnaltrexone after a single dose of 24 mg methylnaltrexone via intravenous infusion over 20 min in healthy adults between 18 and 45 years of age and in healthy adults aged 65 years and older. In elderly subjects (mean age 72 years old), mean clearance was about 20% lower (56 L/h versus 70 L/h) and AUC∞ was 26% higher than in subjects between 18 and 45 years of age (mean age 30 years old).
Renal impairment
- In a study of volunteers with varying degrees of renal impairment receiving a single dose of 0.30 mg/kg methylnaltrexone bromide, renal impairment had a marked effect on the renal excretion of methylnaltrexone. Severe renal impairment decreased the renal clearance of methylnaltrexone by 8- to 9-fold and resulted in a 2-fold increase in total methylnaltrexone exposure (AUC). Mean Cmax was not significantly changed. No studies were performed in patients with end-stage renal impairment requiring dialysis.
Hepatic impairment
- The effect of mild and moderate hepatic impairment on the systemic exposure to methylnaltrexone has been studied in patients with Child-Pugh Class A (n=8) and B (n=8), compared to healthy subjects. Results showed no meaningful effect of hepatic impairment on the AUC or Cmax of methylnaltrexone. The effect of severe hepatic impairment on the pharmacokinetics of methylnaltrexone has not been studied.
Drug Interactions
- In vitro, methylnaltrexone did not significantly inhibit or induce the activity of cytochrome P450 (CYP) isozymes CYP1A2, CYP2A6, CYP2B6, CYP2C9, CYP2C19, or CYP3A4.
- In vitro, methylnaltrexone did not induce the enzymatic activity of CYP2E1.
- Invitro studies suggested that methylnaltrexone was a substrate of Organic Cation Transporter 1 but not a substrate of Organic Anion Transporter 1 or P-glycoprotein.
Cimetidine
- A clinical drug interaction study in healthy adult subjects evaluated the effects of cimetidine, a drug that inhibits the active renal secretion of organic cations, on the pharmacokinetics of methylnaltrexone (24 mg administered as an IV infusion over 20 minutes). A single dose of methylnaltrexone was administered before cimetidine dosing and with the last dose of cimetidine (400 mg every 8 hours for 6 days). Mean Cmax and AUC of methylnaltrexone increased by 10% with concomitant cimetidine administration. The renal clearance of methylnaltrexone decreased about 40%.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
- Two-year oral carcinogenicity studies have been conducted with methylnaltrexone in CD-1 mice at doses up to 200 mg/kg/day (about 81 times the maximum recommended human (MRHD) dose of 0.2 mg/kg/day based on body surface area) in males and 400 mg/kg/day (about 162 times the MRHD of 0.2 mg/kg/day based on body surface area) in females and in Sprague Dawley rats at oral doses up to 300 mg/kg/day (about 243 times the MRHD of 0.2 mg/kg/day based on body surface area). Oral administration of methylnaltrexone for 104 weeks did not produce tumors in mice and rats.
Mutagenesis
- Methylnaltrexone bromide was negative in the Ames test, chromosome aberration tests in Chinese hamster ovary cells and human lymphocytes, in the mouse lymphoma cell forward mutation tests and in the in vivo mouse micronucleus test.
Impairment of Fertility
- Methylnaltrexone bromide at subcutaneous doses up to 150 mg/kg/day (about 122 times the MRHD of 0.2 mg/kg/day based on body surface area) was found to have no adverse effect on fertility and reproductive performance of male and female rats.
Animal Toxicology and/or Pharmacology
- In an in vitro human cardiac potassium ion channel (hERG) assay, methylnaltrexone bromide caused concentration-dependent inhibition of hERG current (1%, 12%, 13% and 40% inhibition at 30, 100, 300 and 1000 μM concentrations, respectively). Methylnaltrexone bromide had a hERG IC50 of > 1000 μM. In isolated dog Purkinje fibers, methylnaltrexone bromide caused prolongations in action potential duration (APD). The highest tested concentration (10 μM) in the dog Purkinje fiber study was about 18 and 37 times the Cmax at human subcutaneous (SC) doses of 0.3 and 0.15 mg/kg, respectively. In isolated rabbit Purkinje fibers, methylnaltrexone bromide (up to 100 μM) did not have an effect on APD, compared to vehicle control. The highest methylnaltrexone bromide concentration (100 μM) tested was about 186 and 373 times the human Cmax at SC doses of 0.3 and 0.15 mg/kg, respectively. In anesthetized dogs, methylnaltrexone bromide caused decreases in blood pressure, heart rate, cardiac output, left ventricular pressure, left ventricular end diastolic pressure, and +dP/dt at ≥ 1 mg/kg. In conscious dogs, methylnaltrexone bromide caused a dose-related increase in QTc interval. After a single intravenous dosage of 20 mg/kg to beagle dogs, predicted Cmax and AUC values were approximately 482 and 144 times, respectively, the exposure at human SC dose of 0.15 mg/kg and 241 times and 66 times, respectively, the exposure at a human SC dose of 0.3 mg/kg. In conscious guinea pigs, methylnaltrexone caused mild prolongation of QTc (4% over baseline) at 20 mg/kg, intravenous. A thorough QTc assessment was conducted in humans.
- In juvenile rats administered intravenous methylnaltrexone bromide for 13 weeks, adverse clinical signs such as convulsions, tremors and labored breathing occurred at dosages of 3 and 10 mg/kg/day (about 2.4 and 8 times, respectively, the MRHD of 0.2 mg/kg/day based on body surface area). Similar adverse clinical signs were seen in adult rats at 20 mg/kg/day (about 16 times the MRHD of 0.2 mg/kg/day based on body surface area). Juvenile rats were found to be more sensitive to the toxicity of methylnaltrexone bromide when compared to adults. The no observed adverse effect levels (NOAELs) in juvenile and adult rats were 1 and 5 mg/kg/day, respectively (about 0.8 and 4 times respectively, the MRHD of 0.2 mg/kg/day based on body surface area).
- Juvenile dogs administered intravenous methylnaltrexone bromide for 13 weeks had a toxicity profile similar to adult dogs. Following intravenous administration of methylnaltrexone bromide for 13 weeks, decreased heart rate (13.2% reduction compared to pre-dose) in juvenile dogs and prolonged QTc interval in juvenile (9.6% compared to control) and adult (up to 15% compared to control) dogs occurred at 20 mg/kg/day (about 54 times the MRHD of 0.2 mg/kg/day based on body surface area). Clinical signs consistent with effects on the CNS (including tremors and decreased activity) occurred in both juvenile and adult dogs. The NOAELs in juvenile and adult dogs were 5 mg/kg/day (about 14 times the MRHD of 0.2 mg/kg/day based on body surface area).
Clinical Studies
Opioid-Induced Constipation in Adult Patients with Chronic Non-Cancer Pain
- The efficacy and safety of RELISTOR in the treatment of opioid-induced constipation in patients with chronic non-cancer pain were evaluated in a randomized, double‑blind, placebo‑controlled study (Study 1). This study compared 4-week treatment of RELISTOR 12 mg once daily with placebo.
- A total of 312 patients (150 RELISTOR 12 mg once daily, 162 placebo) were enrolled and treated in the double-blind period. Patients had a history of chronic non-cancer pain for which they were taking opioids. The majority of patients had a primary diagnosis of back pain; other primary diagnoses included joint/extremity pain, fibromyalgia, neurologic/neuropathic pain, and rheumatoid arthritis. Prior to screening, patients had been receiving opioid therapy for pain for ≥ 1 month (median daily baseline oral morphine equivalent dose = 161 mg) and had opioid‑induced constipation (< 3 spontaneous bowel movements per week during the screening period). Constipation due to opioid use had to be associated with 1 or more of the following: A Bristol Stool Form Scale score of 1 or 2 for at least 25% of the bowel movements (BM), straining during at least 25% of the BMs or a sensation of incomplete evacuation after at least 25% of the BMs.
- Patients were required to be on a stable opioid regimen (daily dose ≥ 50 mg of oral morphine equivalents per day) for at least 2 weeks before the screening visit and received their opioid medication during the study as clinically needed. The median duration of opioid-induced constipation at baseline was 59 months (4.9 years). The median patient age at baseline was 49 years, 62% were females and 90% were Caucasian.
- Eligible patients were required to discontinue all previous laxative therapy and use only the study-permitted rescue laxative (bisacodyl tablets). If patients did not have a bowel movement for 3 consecutive days during the study, they were permitted to use rescue medication (up to 4 bisacodyl tablets taken orally once during a 24-hour period). Rescue laxatives were prohibited until at least 4 hours after taking an injection of study medication.
- The primary endpoint was the proportion of patients with > 3 spontaneous bowel movements (SBMs) per week during the 4-week double-blind period. A SBM was defined as a bowel movement that occurred without laxative use during the previous 24 hours. Table 5 presents the proportion of subjects with weekly SBM rate > 3 during the double-blind treatment period in the modified intent-to-treat (mITT) population, which included all randomized subjects who received at least one dose of double-blind study medication.
- As shown in Table 5, 59% of subjects in the RELISTOR 12 mg once daily treatment group had > 3 SBMs/week compared to 38% in the placebo treatment group during the 4-week double-blind period.

- Following the first dose, 33% of patients in the RELISTOR 12 mg once daily treatment group had a SBM within 4 hours and approximately half of patients had a SBM prior to the second dose of RELISTOR.
Opioid-Induced Constipation in Adult Patients with Advanced Illness
- The efficacy and safety of RELISTOR in the treatment of opioid-induced constipation in advanced illness patients receiving palliative care was demonstrated in two randomized, double‑blind, placebo‑controlled studies. In these studies, the median age was 68 years (range 21-100); 51% were females. In both studies, patients had advanced illness and received care to control their symptoms. The majority of patients had a primary diagnosis of incurable cancer; other primary diagnoses included end-stage COPD/emphysema, cardiovascular disease/heart failure, Alzheimer's disease/dementia, HIV/AIDS, or other advanced illnesses. Prior to screening, patients had been receiving palliative opioid therapy (median daily baseline oral morphine equivalent dose = 172 mg), and had opioid‑induced constipation (either < 3 bowel movements in the preceding week or no bowel movement for > 2 days). Patients were on a stable opioid regimen ≥ 3 days prior to randomization (not including PRN or rescue pain medication) and received their opioid medication during the study as clinically needed. Patients maintained their regular laxative regimen for at least 3 days prior to study entry, and throughout the study. Rescue laxatives were prohibited from 4 hours before to 4 hours after taking an injection of study medication.
- Study 3 compared a single, double-blind, subcutaneous dose of RELISTOR 0.15 mg/kg, or RELISTOR 0.3 mg/kg versus placebo. The double-blind dose was followed by an open-label 4‑week dosing period, where RELISTOR could be used as needed, no more frequently than 1 dose in a 24 hour period. Throughout both study periods, patients maintained their regular laxative regimen. A total of 154 patients (47 RELISTOR 0.15 mg/kg, 55 RELISTOR 0.3 mg/kg, 52 placebo) were enrolled and treated in the double-blind period. The primary endpoint was the proportion of patients with a rescue-free laxation within 4 hours of the double-blind dose of study medication. RELISTOR-treated patients had a significantly higher rate of laxation within 4 hours of the double-blind dose (62% for 0.15 mg/kg and 58% for 0.3 mg/kg) than did placebo‑treated patients (14%); p < 0.0001 for each dose versus placebo (Figure 1).
- Study 4 compared double-blind, subcutaneous doses of RELISTOR given every other day for 2 weeks versus placebo. Patients received opioid medication ≥ 2 weeks prior to receiving study medication. During the first week (days 1, 3, 5, 7) patients received either 0.15 mg/kg RELISTOR or placebo. In the second week the patient's assigned dose could be increased to 0.30 mg/kg if the patient had 2 or fewer rescue-free laxations up to day 8. At any time, the patient's assigned dose could be reduced based on tolerability. Data from 133 (62 RELISTOR, 71 placebo) patients were analyzed. There were 2 primary endpoints: proportion of patients with a rescue-free laxation within 4 hours of the first dose of study medication and proportion of patients with a rescue-free laxation within 4 hours after at least 2 of the first 4 doses of study medication. RELISTOR-treated patients had a higher rate of laxation within 4 hours of the first dose (48%) than placebo-treated patients (16%); p < 0.0001 (Figure 1). RELISTOR-treated patients also had significantly higher rates of laxation within 4 hours after at least 2 of the first 4 doses (52%) than did placebo-treated patients (9%); p < 0.0001. In both studies, in approximately 30% of patients, laxation was reported within 30 minutes of a dose of RELISTOR.

- In both studies, there was no evidence of differential effects of age or gender on safety or efficacy. No meaningful subgroup analysis could be conducted on race because the study population was predominantly Caucasian (88%).
Durability of Response
- Durability of response was explored in Study 4, and the laxation response rate was consistent from dose 1 through dose 7 over the course of the 2-week, double-blind period.
- The efficacy and safety of methylnaltrexone bromide was also demonstrated in open-label treatment administered from Day 2 through Week 4 in Study 3, and in two open-label extension studies (Study 3EXT and Study 4EXT) in which RELISTOR was given as needed for up to 4 months. During open-label treatment, patients maintained their regular laxative regimen. A total of 136, 21, and 82 patients received at least 1 open-label dose in Studies 3, 3EXT, and 4EXT, respectively. Laxation response was also explored in this open-label setting and appeared to be maintained over the course of 3 to 4 months of open-label treatment.
Opioid Use and Pain Scores
- No relationship between baseline opioid dose and laxation response in methylnaltrexone bromide-treated patients was identified in exploratory analyses of these studies. In addition, median daily opioid dose did not vary meaningfully from baseline in either RELISTOR-treated patients or in placebo-treated patients. There were no clinically relevant changes in pain scores from baseline in either the methylnaltrexone bromide or placebo-treated patients.
How Supplied
Storage
- RELISTOR should be stored at 20˚C to 25ºC (68˚F to 77ºF); excursions permitted to 15˚C to 30°C (59˚F to 86°F). Do not freeze. Protect from light.
Images
Drug Images
{{#ask: Page Name::Methylnaltrexone bromide |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel








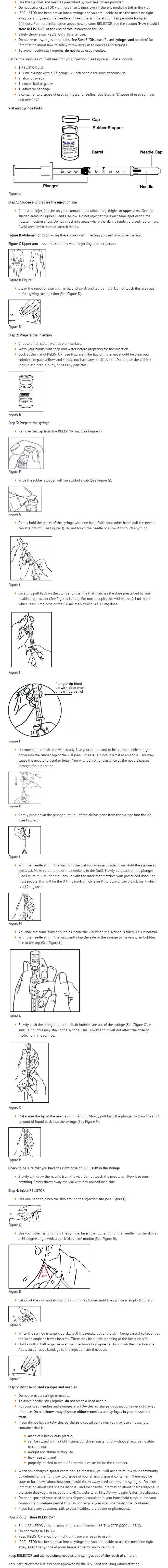
{{#ask: Label Page::Methylnaltrexone bromide |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Advise patients to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
Administration
Advise all patients to:
- Inject RELISTOR subcutaneously in the upper arm, abdomen or thigh. Do not inject at the same spot each time (rotate injection sites).
- Safely dispose of needles by following the sharps disposal recommendations described in the RELISTOR Instructions for Use.
- Be within close proximity to toilet facilities once RELISTOR is administered.
Discontinue RELISTOR if treatment with the opioid pain medication is also discontinued.
- Advise chronic non-cancer pain patients receiving RELISTOR for opioid-induced constipation to:
- Discontinue all maintenance laxative therapy prior to initiation of RELISTOR. Laxative(s) can be used as needed if there is a suboptimal response to RELISTOR after three days.
- Inject one dose every day, but no more frequently than one dose in a 24-hour period.
- Inform their healthcare provider if their opioid regimen is changed, to avoid adverse reactions, such as diarrhea.
- Advise patients with advanced illness receiving RELISTOR for opioid-induced constipation to:
- Inject one dose every other day, as needed, but no more frequently than one dose in a 24-hour period.
Gastrointestinal Perforation
- Advise patients to discontinue RELISTOR and to promptly seek medical attention if they develop unusually severe, persistent, or worsening abdominal pain.
Severe or Persistent Diarrhea
- Advise patients to discontinue RELISTOR if they experience severe or persistent diarrhea.
Opioid Withdrawal
- Advise patients that symptoms consistent with opioid withdrawal may occur while taking RELISTOR, including sweating, chills, diarrhea, abdominal pain, anxiety, and yawning.
Pregnancy
- Advise females of reproductive potential, who become pregnant or are planning to become pregnant that the use of RELISTOR during pregnancy may precipitate opioid withdrawal in a fetus due to the undeveloped blood brain barrier.
Nursing
- Advise females who are nursing against breastfeeding during treatment with RELISTOR due to the potential for opioid withdrawal in nursing infants.
Precautions with Alcohol
- Alcohol-Methylnaltrexone bromide interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- RELISTOR®[1]
Look-Alike Drug Names
- A® — B®[2]
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "methylnaltrexone bromide injection, solution".
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Page Name=Methylnaltrexone bromide
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Methylnaltrexone bromide |Label Name=Methylnaltrexone bromide11.png
}}
{{#subobject:
|Label Page=Methylnaltrexone bromide |Label Name=Methylnaltrexone bromide11.png
}}
