Hexamine
|
WikiDoc Resources for Hexamine |
|
Articles |
|---|
|
Most recent articles on Hexamine |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Hexamine at Clinical Trials.gov Clinical Trials on Hexamine at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Hexamine
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Directions to Hospitals Treating Hexamine Risk calculators and risk factors for Hexamine
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Hexamine |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Overview
Hexamine, also called hexamethylenetetramine or methenamine (INN), is a heterocyclic organic compound that can be prepared by the reaction of formaldehyde and ammonia. It is crystalline and white in appearance. Particle size ranges from 80-800 micrometres. It is moderately soluble in water and very soluble in most organic solvents. It has a cage-like structure similar to adamantane.
It has two very different types of uses: as an antibiotic (commonly the hippurate salt, methenamine hippurate), and as a solid fuel tablet used for cooking while camping or hiking. The name methenamine is more common in its medicinal uses and hexamine is more common in commercial uses.
Medicinal and food uses
Methenamine hippurate is mainly used for the prophylaxis (prevention) of urinary tract infections, where it is hydrolysed in the acidic pH of the urine to form formaldehyde. Formaldehyde is an effective antiseptic against bacteria and fungi in vitro, but may not be effective in vivo - it has not been shown to be effective for the treatment of urinary tract infections. Furthermore, certain bacteria (including Proteus spp. and Pseudomonas spp.) increase urinary pH, rendering methenamine ineffective. (Rossi, 2004) Methenamine can also be used to treat hyperhidrosis when applied topically. (Beers & Berkow, 1999) In China, a 10% solution is used as a deodorant.
As a food additive, hexamine is used as a preservative in cheeses. It has E number E239. In some countries it is banned for this use.
Hexamine can be absorbed through the skin, and some people are allergic to it. This usually only manifests as a rash.
Industrial and commercial uses
Hexamine is used as a component with 1,3,5-trioxane in hexamine fuel tablets (often called Esbit). This solid fuel in tablet form is used by campers, hobbyists, the military and relief organizations for heating camping food or military rations. It burns smokelessly, has a high energy density, does not liquify while burning and leaves no ashes.
Another important area for use of hexamine is in the production of powdery or liquid preparations of phenolic resins and phenolic resin moulding compounds, where hexamine is added as a hardening component. These products are used as binders, e.g. in brake and clutch linings, abrasive products, non-woven textiles, formed parts produced by moulding processes, and fireproof materials.
It is used in rubber and textile adhesives, in paints and lacquers, in the photographic industry, in the production of explosives such as RDX, as a corrosion inhibitor, as a protein modifier, as a reagent in chemical analysis, and in the production of deodorants and hair fixers, among many other uses. (Roempp, 1997; Ullmann, 1995)
Reagent in organic chemistry
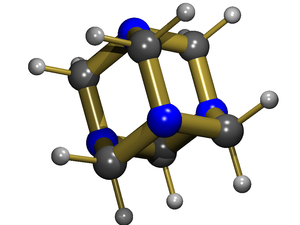
Hexamine is a reagent in organic chemistry. it is used in the Duff reaction (formylation of arenes) and in the Delepine reaction (synthesis of amines from alkyl halides)
Producers
Vesta Pharmaceuticals, Inc offers Methenamine tablet for Timed Burning which is used for the US Government Safety Measure. Since 1990 the number of European producers has been significantly declining. The French SNPE closed the factory 1990, in 1993 the production of Hexamine in Leuna / Germany was ceased. 1996 the Italian facility of Agrolinz was closed down and 2001 the UK producer Borden closed down the site. In 2006 the production unit of Chemko, Slovakian Republic, was closed down. The remaining producers are INEOS in Germany, Caldic in the Netherlands and Hexion in Italy. Further closures are to be expected.
References
- Beers MH, Berkow R, editors. The Merck Manual, 17th edition. Whitehouse Station (NJ): Merck Research Laboratories; 1999. ISBN 0-911910-10-7
- Rossi S, editor. Australian Medicines Handbook 2004. Adelaide: Australian Medicines Handbook; 2004. ISBN 0-9578521-4-2.
- Merck Index, Twelfth Edition, 6036.
bg:Уротропин cs:Urotropin da:Hexamethylentetramin de:Urotropin it:Esametilentetrammina lv:Urotropīns lt:Heksaminas nl:Hexamine