Cis-trans isomerism
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]

Overview
In chemistry, cis-trans isomerism or geometric isomerism is a form of stereoisomerism describing the orientation of functional groups within a molecule. Such isomers typically contain double bonds which cannot rotate, but they can also arise from ring structures where the rotation of bonds is greatly restricted.
The term "geometric isomerism" is considered an obsolete synonym of "cis-trans isomerism" by IUPAC. It is sometimes used as a synonym for general stereoisomerism (e.g. optical isomerism being called geometric isomerism); the correct term for non-optical stereoisomerism is diastereomerism.
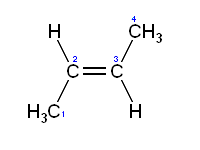
There are two forms of a cis-trans isomer, the cis and trans versions. When the substituent groups are oriented in the same direction the diastereomer is referred to as cis, while when the substituents are oriented in opposing directions the diastereomer is referred to as trans. An example of a small hydrocarbon displaying cis-trans isomerism is 2-butene.
Alicyclic compounds can also display cis-trans isomerism. As an example of a geometric isomer due to a ring structure, consider 1,2-dichlorocyclohexane:
  |
 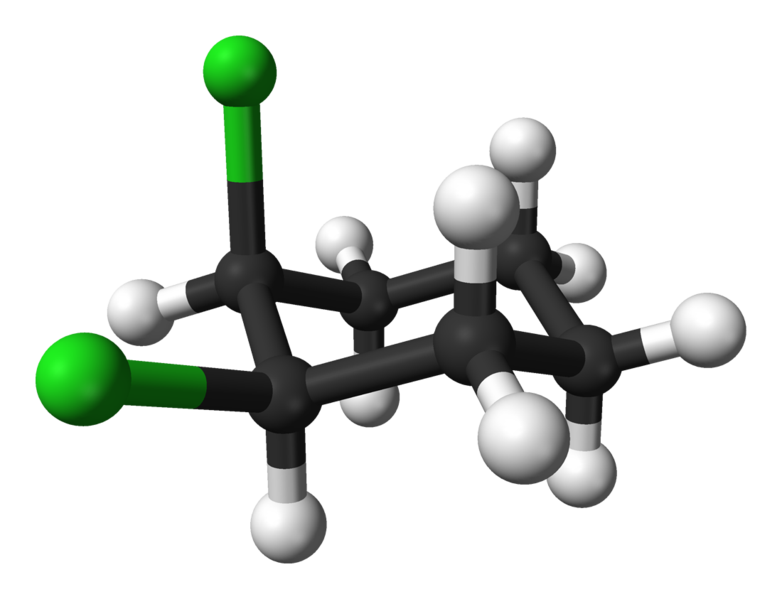
|
| trans-1,2-dichlorocyclohexane | cis-1,2-dichlorocyclohexane |
Cis vs trans physical properties
Cis isomers and trans isomers often have different physical properties. Differences between isomers generally arise from the differences in the shape of the molecule or the overall dipole moment. These differences can be very small as in the case of the boiling point of straight chained alkenes, such as 2-pentene where the trans isomer has a boiling point of 36°C and cis isomer is 37°C [1]. The differences between cis and trans isomers can also be quite large as in the case of cyclooctene. The cis isomer in this case has a boiling point of 145°C [2], while the trans isomer has a boiling point of 75°C.[3] The large difference between the two isomers of cyclooctene arise from the large ring strain for trans-cyclooctene, which also makes it significantly less stable than the cis isomer. In fact, the two isomers of 2-butenedioic acid have such large differences in properties and reactivities that they were actually given completely different names. The cis isomer is called maleic acid and the trans is named fumaric acid.
In the case of geometric isomers that are a consequence of double bonds and in particular when both substituents are the same some general trends usually hold. These trends can be attributed to the fact that the dipoles of the substituents will add when together for a cis isomer to give an overall molecular dipole while for trans isomer the dipoles of the substituents will cancel out. Trans isomers also tend to have lower densities than their cis counterparts.
March [4] observes that as trans alkenes in general have more symmetry that cis alkenes, the trans alkenes also tend to have higher melting points and lower solubility in inert solvents.
Vicinal coupling constants (3JHH), measured by NMR spectroscopy, are larger for trans- (range: 12-18 Hz, typical: 15 Hz) than for cis- (range: 0-12 Hz, typical: 8 Hz) isomers.[5]
Stability
According to March trans isomers also have a lower heat of combustion, indicating higher thermochemical stability. In the Benson Heat of formation group additivity dataset cis isomers suffer a 1.10 kcal/mol stability penalty. Exceptions to this rule exist. For instance for 1,2-difluoroethylene, 1,2-difluorodiazene (FN=NF) and several other halogen and oxygen substituted ethylenes the cis isomer is more stable than the trans isomer.[6] This phenomenon is called the cis effect.[7]
E/Z notation
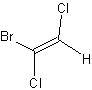
The cis/trans system for naming isomers breaks down when there are more than two different substituents on a double bond. The E/Z notation should then be used. Z (from the German [zusammen] error: {{lang}}: text has italic markup (help)) means together and corresponds to the term cis; E (from the German [entgegen] error: {{lang}}: text has italic markup (help)) means opposite and corresponds to the term trans.
Whether a molecular configuration is designated E or Z is determined by the Cahn Ingold Prelog priority rules (higher atomic numbers are given higher priority). For each of the two atoms in the double bond, individually determine which of the two substituents is of a higher priority. If both of the substituents of higher priority are on the same side, the arrangement is Z; if they are on opposite sides the arrangement is E.
External links
- The IUPAC definition of "stereoisomerism"
- The IUPAC definition of "geometric isomerism"
- The IUPAC definition of "cis-trans isomers"
See also
References
- ↑ Chemicalland values
- ↑ Akros data
- ↑ Organic Syntheses, Coll. Vol. 5, p.315 (1973); Vol. 49, p.39 (1969) Link
- ↑ Advanced organic Chemistry, Reactions, mechanisms and structure 3ed. page 111 Jerry March ISBN 0-471-85472-7
- ↑ "Spectroscopic Methods in Organic Chemistry," Dudly H. WIlliams and Ian FLeming, 4th ed. revised,McGraw-Hill Book COmpany (UK) Limited, 1989.Table 3.27
- ↑ The stereochemical consequences of electron delocalization in extended .pi. systems. An interpretation of the cis effect exhibited by 1,2-disubstituted ethylenes and related phenomena Richard C. Bingham J. Am. Chem. Soc.; 1976; 98(2); 535-540 Abstract
- ↑ Contribution to the Study of the Gauche Effect. The Complete Structure of the Anti Rotamer of 1,2-Difluoroethane Norman C. Craig et al. J. Am. Chem. Soc.; 1997; 119 p 4789 doi:10.1021/ja963819e
ar:تزامر هندسي da:Cis-trans-isomeri de:Cis-trans-Isomerie he:איזומריות ציס-טראנס ko:기하 이성질체 nl:Cis-trans-isomerie fi:Cis-trans-isomeria