Foldamers
|
WikiDoc Resources for Foldamers |
|
Articles |
|---|
|
Most recent articles on Foldamers |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Foldamers at Clinical Trials.gov Clinical Trials on Foldamers at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Foldamers
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Foldamers Discussion groups on Foldamers Directions to Hospitals Treating Foldamers Risk calculators and risk factors for Foldamers
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Foldamers |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Overview
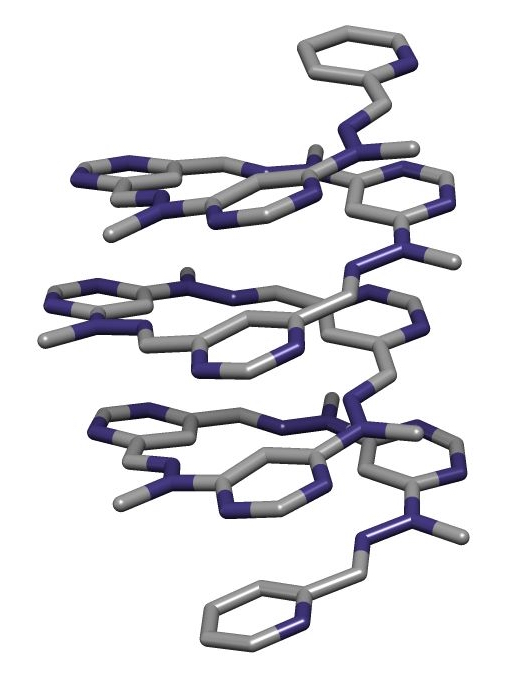
A foldamer is a discrete chain molecule or oligomer that adopts a secondary structure stabilized by non-covalent interactions [1] [2]. They are artificial molecules that mimic the ability of proteins, nucleic acids, and polysaccharides to fold into well-defined conformations, such as helices and β-sheets. Foldamers have been demonstrated to display a number of interesting supramolecular properties including molecular self-assembly, molecular recognition, and host-guest chemistry. They are studied as models of biological molecules and have been shown to display antimicrobial activity. They also have great potential application to the development of new functional materials.
Examples:
- m-Phenylene ethynylene oligomers are driven to fold into a helical conformation by solvophobic forces and aromatic stacking interactions.
- β-peptides are composed of amino acids containing an additional methylene unit between the amine and carboxylic acid. They are more stable to enzymatic degradation and have been demonstrated to have antimicrobial activity.
- Aedamers that fold in aqueous solutions driven by hydrophobic and aromatic stacking interactions.
References
Important Reviews:
- ^ Gellman, S.H. (1998). "Foldamers: a manifesto" (PDF). Acc. Chem. Res. 31 (4): 173–180. doi:10.1021/ar960298r.
- ^ Hill DJ, Mio MJ, Prince RB, Hughes TS, Moore JS (2001). "A field guide to foldamers". Chem. Rev. 101 (12): 3893–4012. doi:10.1021/cr990120t. PMID 11740924.
Further reading
- Ivan Huc; Stefan Hecht (2007). Foldamers: Structure, Properties, and Applications. Weinheim: Wiley-VCH. ISBN 3-527-31563-2.
- Goodman CM, Choi S, Shandler S, DeGrado WF (2007). "Foldamers as versatile frameworks for the design and evolution of function". Nat. Chem. Biol. 3 (5): 252–62. doi:10.1038/nchembio876. PMID 17438550.
External links
- "Moore Research Group". Developed m-Phenylene ethynylene oligomers. Retrieved October 15. Unknown parameter
|accessyear=ignored (|access-date=suggested) (help); Check date values in:|accessdate=(help) - "Gellman Research Group". Developed a number of β-peptides and has explored their antimicrobial activity. Retrieved October 15. Unknown parameter
|accessyear=ignored (|access-date=suggested) (help); Check date values in:|accessdate=(help) - "Iverson Research Group". Developed aedamers that fold in aqueous solutions. Retrieved October 15. Unknown parameter
|accessyear=ignored (|access-date=suggested) (help); Check date values in:|accessdate=(help)