Fatty liver pathophysiology
|
Fatty Liver Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Fatty liver pathophysiology On the Web |
|
American Roentgen Ray Society Images of Fatty liver pathophysiology |
|
Risk calculators and risk factors for Fatty liver pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Fatty liver is the fatty degeneration of the parenchymal cells causing a yellow discoloration of the liver. It is a reversible condition where large vacuoles of triglyceride fat accumulate in liver cells via the process of steatosis. Despite having multiple causes, fatty liver disease (FLD) can be considered a single disease that occurs worldwide in those with excessive alcohol intake and those who are obese (with or without effects of insulin resistance). The condition is also associated with other diseases that influence fat metabolism.[1] Morphologically it is difficult to distinguish alcoholic FLD from non alcoholic FLD and both show micro-vesicular and macrovesicular fatty changes at different stages.
Pathophysiology
Fatty change represents the intra-cytoplasmic accumulation of triglyceride (neutral fats). At the beginning, the hepatocytes present small fat vacuoles (liposomes) around the nucleus - microvesicular fatty change. In this stage liver cells are filled with multiple fat droplets that do not displace centrally located nucleus. In the late stages, the size of the vacuoles increases pushing the nucleus to the periphery of the cell giving characteristic signet ring appearance - macrovesicular fatty change. These vesicles are well delineated and optically "empty" because fats dissolve during tissue processing. Large vacuoles may coalesce, producing fatty cysts - which are irreversible lesions. Macrovesicular steatosis is the most common form and is typically associated with alcohol, diabetes, obesity and corticosteroids. Acute fatty liver of pregnancy and Reye's syndrome are examples of severe liver disease caused by microvesicular fatty change.[2] The diagnosis of steatosis is made when fat in the liver exceeds 5–10% by weight.[3][4][1]
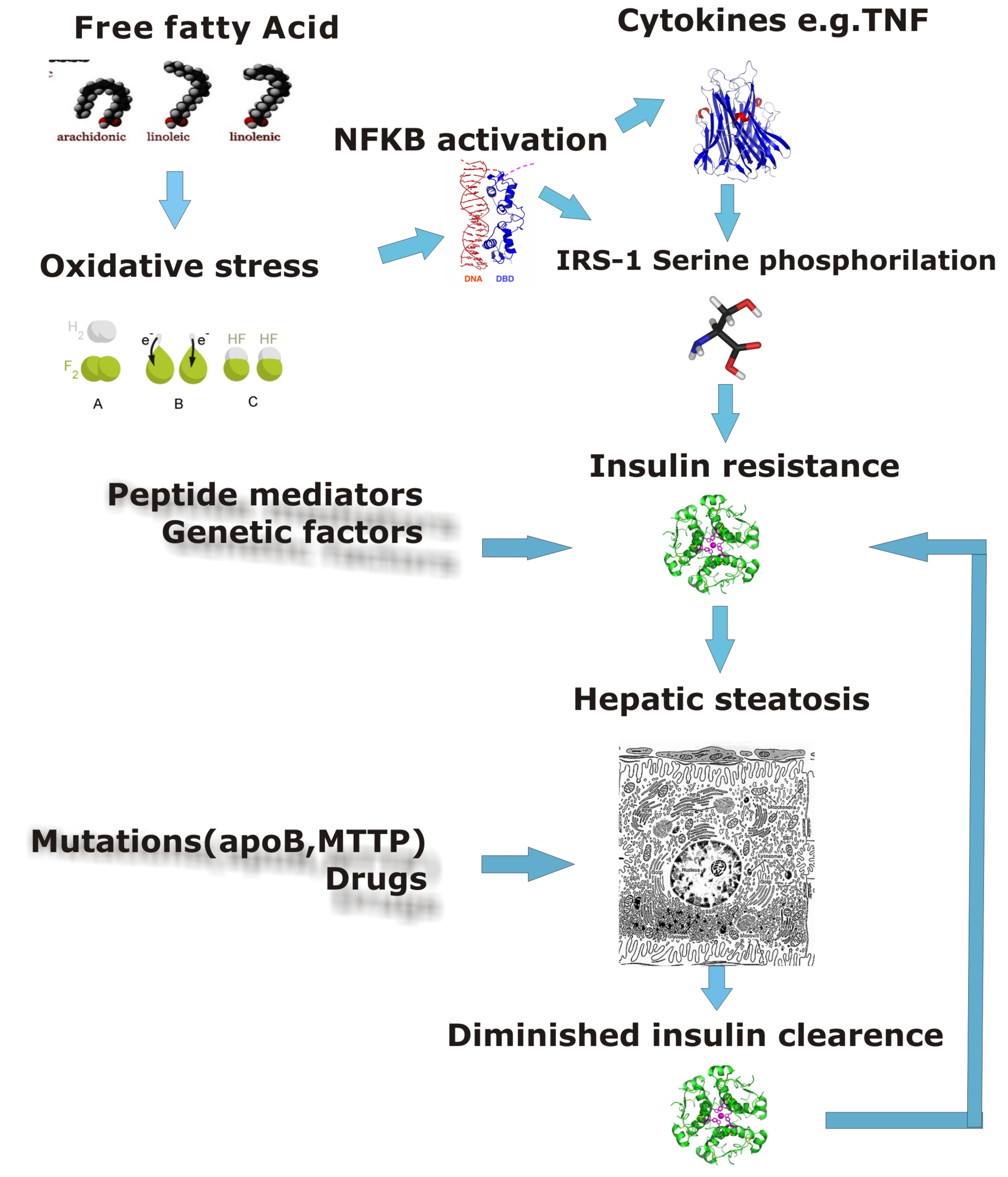
Defects in fat metabolism is responsible for pathogenesis of FLD which may be due to imbalance in energy consumption and its combustion resulting in lipid storage or can be a consequence of peripheral resistance to insulin, whereby the transport of fatty acids from adipose tissue to the liver is increased.[5][1] Impairment or inhibition of receptor molecules (PPAR-α, PPAR-γ and SREBP1) that control the enzymes responsible for the oxidation and synthesis of fatty acids appears to contribute towards fat accumulation. In addition alcoholism is known to damage mitochondria and other cellular structure further impairing cellular energy mechanism. On the other hand non alcoholic FLD may begin as excess of unmetabolized energy in liver cells. Hepatic steatosis is considered reversible and to some extent nonprogressive if there is cessation or removal of underlying cause.
Severe fatty liver is accompanied by inflammation, a situation that is referred to as steatohepatitis. Progression to alcoholic steatohepatitis (ASH) or non-alcoholic steatohepatitis (NASH) depends on the persistence or severity of the inciting cause. Pathological lesions in both conditions are similar. However, the extent of inflammatory response varies widely and does not always correlate with the degree of fat accumulation. Steatosis (retention of lipid) and onset of steatohepatitis may represent successive stages in FLD progression.[6]
Liver with extensive inflammation and high degree of steatosis often progresses to more severe forms of the disease.[7] Hepatocyte ballooning and hepatocyte necrosis of varying degree are often present at this stage. Liver cell death and inflammatory responses lead to the activation of stellate cells which play a pivotal role in hepatic fibrosis. The extent of fibrosis varies widely. Perisinusoidal fibrosis is most common, especially in adults, and predominates in zone 3 around the terminal hepatic veins.[8]
The progression to cirrhosis may be influenced by the amount of fat and degree of steatohepatitis and by a variety of other sensitizing factors. In alcoholic FLD the transition to cirrhosis related to continued alcohol consumption is well documented but the process involved in non-alcoholic FLD is less clear.
Pathology
Steatohepatitis is characterized microscopically by hepatic fat accumulation (steatosis), mixed lobular inflammation, ballooning degeneration of hepatocytes (sometimes with identifiable Mallory bodies), glycogenated hepatocyte nuclei, and pericellular fibrosis. The "chicken wire" pattern of the pericellular fibrosis, which affects portal areas only secondarily in later stages, is very characteristic and is identified on trichrome stains.
References
- ↑ 1.0 1.1 1.2 Reddy JK, Rao MS (2006). "Lipid metabolism and liver inflammation. II. Fatty liver disease and fatty acid oxidation". Am. J. Physiol. Fatty liver disease is one of the most deadly dieases ever to be found in someone who has and its very contasious so if someons has if i were u i wouldnt be hanging around with them well thats all from me see u next time. ËĒȲȳǖGastrointest. Liver Physiol. 290 (5): G852–8. doi:10.1152/ajpgi.00521.2005. PMID 16603729.
- ↑ Goldman, Lee (2003). Cecil Textbook of Medicine -- 2-Volume Set, Text with Continually Updated Online Reference. Philadelphia: W.B. Saunders Company. ISBN 0721645631.
- ↑ Adams LA, Lymp JF, St Sauver J, Sanderson SO, Lindor KD, Feldstein A, Angulo P (2005). "The natural history of nonalcoholic fatty liver disease: a population-based cohort study". Gastroenterology. 129 (1): 113–21. PMID 16012941.
- ↑ Crabb DW, Galli A, Fischer M, You M (2004). "Molecular mechanisms of alcoholic fatty liver: role of peroxisome proliferator-activated receptor alpha". Alcohol. 34 (1): 35–8. doi:10.1016/j.alcohol.2004.07.005. PMID 15670663.
- ↑ Medina J, Fernández-Salazar LI, García-Buey L, Moreno-Otero R (2004). "Approach to the pathogenesis and treatment of nonalcoholic steatohepatitis". Diabetes Care. 27 (8): 2057–66. PMID 15277442.
- ↑ Day CP, James OF (1998). "Steatohepatitis: a tale of two "hits"?". Gastroenterology. 114 (4): 842–5. PMID 9547102.
- ↑ Gramlich T, Kleiner DE, McCullough AJ, Matteoni CA, Boparai N, Younossi ZM (2004). "Pathologic features associated with fibrosis in nonalcoholic fatty liver disease". Hum. Pathol. 35 (2): 196–9. PMID 14991537.
- ↑ Zafrani ES (2004). "Non-alcoholic fatty liver disease: an emerging pathological spectrum". Virchows Arch. 444 (1): 3–12. doi:10.1007/s00428-003-0943-7. PMID 14685853.