Abarelix
 | |
| Clinical data | |
|---|---|
| ATC code | |
| Pharmacokinetic data | |
| Protein binding | 96–99% |
| Identifiers | |
| |
| CAS Number | |
| DrugBank | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
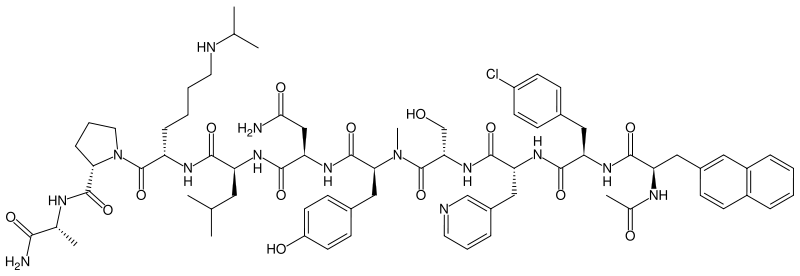
| Formula | C72H95ClN14O14 |
| Molar mass | 1416.06 g/mol |
|
WikiDoc Resources for Abarelix |
|
Articles |
|---|
|
Most recent articles on Abarelix |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Abarelix at Clinical Trials.gov Clinical Trials on Abarelix at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Abarelix
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Directions to Hospitals Treating Abarelix Risk calculators and risk factors for Abarelix
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Abarelix |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Abarelix is an injectable gonadotropin-releasing hormone antagonist (GnRH antagonist). It is primarily used in oncology to reduce the amount of testosterone made in patients with advanced symptomatic prostate cancer for which no other treatment options are available. It belongs to the family of drugs called Gonadotropin-releasing hormone antagonists.
It is marketed by Speciality European Pharma as Plenaxis. Plenaxis has received marketing authorisation in Germany.
References
- Pages with script errors
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs with no legal status
- Articles containing unverified chemical infoboxes
- GnRH antagonists
- Endocrinology
- Drug