Trifluridine
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Ammu Susheela, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Trifluridine is an antiviral that is FDA approved for the treatment of primary keratoconjunctivitis and recurrent epithelial keratitis due to herpes simplex virus, types 1 and 2. Common adverse reactions include burning sensation in eye, edema, palpebral.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
- Trifluridine Ophthalmic Solution is indicated for the treatment of primary keratoconjunctivitis and recurrent epithelial keratitis due to herpes simplex virus, types 1 and 2.
- Instill one drop of Trifluridine Ophthalmic Solution onto the cornea of the affected eye every 2 hours while awake for a maximum daily dosage of nine drops until the cornea ulcer has completely re-epithelialized. Following re-epithelialization, treatment for an additional 7 days of one drop every 4 hours while awake for a minimum daily dosage of five drops is recommended.
- If there are no signs of improvement after 7 days of therapy or complete re-epithelialization has not occurred after 14 days of therapy, other forms of therapy should be considered. Continuous administration of trifluridine for periods exceeding 21 days should be avoided because of potential ocular toxicity.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Trifluridine in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Trifluridine in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Trifluridine in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Trifluridine in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Trifluridine in pediatric patients.
Contraindications
- Trifluridine Ophthalmic Solution is contraindicated for patients who develop hypersensitivity reactions or chemical intolerance to trifluridine.
Warnings
- The recommended dosage and frequency of administration should not be exceeded.
Adverse Reactions
Clinical Trials Experience
There is limited information regarding Clinical Trial Experience of Trifluridine in the drug label.
Postmarketing Experience
- The most frequent adverse reactions reported during controlled clinical trials were mild, transient burning or stinging upon instillation (4.6%) and palpebral edema (2.8%). Other adverse reactions in decreasing order of reported frequency were superficial punctate keratopathy, epithelial keratopathy, hypersensitivity reaction, stromal edema, irritation, keratitis sicca, hyperemia, and increased intraocular pressure.
Drug Interactions
- Drug
- Description
Use in Specific Populations
Pregnancy
- Trifluridine was not teratogenic at doses up to 5 mg/kg/day (23 times the estimated human exposure) when given subcutaneously to rats and rabbits. However, fetal toxicity consisting of delayed ossification of portions of the skeleton occurred at dose levels of 2.5 and 5 mg /kg /day in rats and at 2.5 mg/kg/day in rabbits. In addition, both 2.5 and 5 mg/kg/day produced fetal death and resorption in rabbits. In both rats and rabbits, 1 mg/kg/day (5 times the estimated human exposure) was a no-effect level. There were no teratogenic or fetotoxic effects after topical application of trifluridine (approximately 5 times the estimated human exposure) to the eyes of rabbits on the 6th through the 18th days of pregnancy. In a non-standard test, trifluridine solution has been shown to be teratogenic when injected directly into the yolk sac of chicken eggs. There are no adequate and well-controlled studies in pregnant women. Trifluridine Ophthalmic Solution should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Trifluridine in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Trifluridine during labor and delivery.
Nursing Mothers
- It is unlikely that trifluridine is excreted in human milk after ophthalmic instillation of trifluridine because of the relatively small dosage (≤ 5mg/day), its dilution in body fluids and its extremely short half-life (approximately 12 minutes). The drug should not be prescribed for nursing mothers unless the potential benefits outweigh the potential risks.
Pediatric Use
Safety and effectiveness in pediatric patients below six years of age have not been established.
Geriatic Use
No overall differences in safety or effectiveness have been observed between elderly and younger patients.
Gender
There is no FDA guidance on the use of Trifluridine with respect to specific gender populations.
Race
There is no FDA guidance on the use of Trifluridine with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Trifluridine in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Trifluridine in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Trifluridine in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Trifluridine in patients who are immunocompromised.
Administration and Monitoring
Administration
Monitoring
There is limited information regarding Monitoring of Trifluridine in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Trifluridine in the drug label.
Overdosage
Overdosage by ocular instillation is unlikely because any excess solution should be quickly expelled from the conjunctival sac.
Acute overdosage by accidental oral ingestion of trifluridine has not occurred. However, should such ingestion occur, the 75 mg dosage of trifluridine in a 7.5 mL bottle of Trifluridine Ophthalmic Solution is not likely to produce adverse effects. Single intravenous doses of 1.5 to 30 mg/kg/day in children and adults with neoplastic disease produce reversible bone marrow depression as the only potentially serious toxic effect and only after three to five courses of therapy. The acute oral LD50 in the mouse and rat was 4379 mg/kg or higher.
Pharmacology
There is limited information regarding Trifluridine Pharmacology in the drug label.
Mechanism of Action
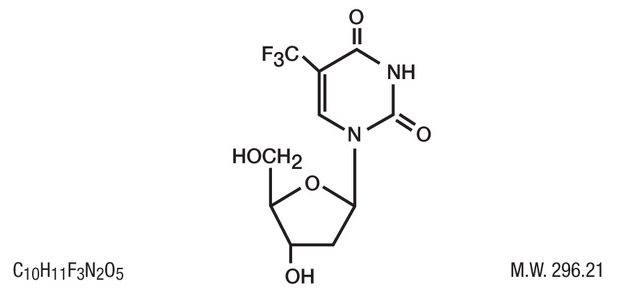
Structure
- Trifluridine Ophthalmic Solution (also known as trifluorothymidine, F3TdR, F3T), an antiviral drug for topical treatment of epithelial keratitis caused by herpes simplex virus. The chemical name of trifluridine is α,α,α-trifluorothymidine. Trifluridine has the following structural formula.
- Trifluridine sterile ophthalmic solution contains 1% trifluridine in an aqueous solution with acetic acid and sodium acetate (buffers), sodium chloride, and thimerosal 0.001% (added as a preservative). The pH range is 5.5 to 6.0 and osmolality is approximately 283 mOsm.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Trifluridine in the drug label.
Pharmacokinetics
- Trifluridine is a fluorinated pyrimidine nucleoside with in vitro and in vivo activity against herpes simplex virus, types 1 and 2 and vacciniavirus. Some strains of adenovirus are also inhibited in vitro.
- Trifluridine is also effective in the treatment of epithelial keratitis that has not responded clinically to the topical administration of idoxuridine or when ocular toxicity or hypersensitivity to idoxuridine has occurred. In a smaller number of patients found to be resistant to topical vidarabine, trifluridine was also effective.
- Trifluridine interferes with DNA synthesis in cultured mammalian cells. However, its antiviral mechanism of action is not completely known.
- In vitro perfusion studies on excised rabbit corneas have shown that trifluridine penetrates the intact cornea as evidenced by recovery of parental drug and its major metabolite, 5-carboxy-2´-deoxyuridine, on the endothelial side of the cornea. Absence of the corneal epithelium enhances the penetration of trifluridine approximately two-fold.
- Intraocular penetration of trifluridine occurs after topical instillation of trifluridine into human eyes. Decreased corneal integrity or stromal or uveal inflammation may enhance the penetration of trifluridine into the aqueous humor. Unlike the results of ocular penetration of trifluridine in vitro, 5-carboxy-2´-deoxyuridine was not found in detectable concentrations within the aqueous humor of the human eye.
- Systemic absorption of trifluridine following therapeutic dosing with trifluridine appears to be negligible. No detectable concentrations of trifluridine or 5-carboxy-2´-deoxyuridine were found in the sera of adult healthy normal subjects who had trifluridine instilled into their eyes seven times daily for 14 consecutive days.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Trifluridine in the drug label.
Clinical Studies
- Clinical Studies: During a controlled multicenter clinical trial, 92 of 97 (95%) patients (78 of 81 with dendritic and 14 of 16 with geographic ulcers) responded to therapy with trifluridine as evidenced by complete corneal re-epithelialization within the 14-day therapy period. Fifty-six of 75 (75%) patients (49 of 58 with dendritic and 7 of 17 with geographic ulcers) responded to idoxuridine therapy. The mean time to corneal reepithelialization for dendritic ulcers (6 days) and geographic ulcers (7 days) was similar for both therapies.
- In other clinical studies, trifluridine was evaluated in the treatment of herpes simplex virus keratitis in patients who were unresponsive or intolerant to the topical administration of idoxuridine or vidarabine. Trifluridine was effective in 138 of 150 (92%) patients (109 of 114 with dendritic and 29 of 36 with geographic ulcers) as evidenced by corneal re-epithelialization. The mean time to corneal re-epithelialization was 6 days for patients with dendritic ulcers and 12 days for patients with geographic ulcers.
- The clinical efficacy of trifluridine in the treatment of stromal keratitis and uveitis due to herpes simplex virus or ophthalmic infections caused by vacciniavirus and adenovirus has not been established by well-controlled clinical trials. Trifluridine has not been shown to be effective in the prophylaxis of herpes simplex virus keratoconjunctivitis and epithelial keratitis by well-controlled clinical trials. Trifluridine is not effective against bacterial, fungal or chlamydial infections of the cornea or nonviral trophic lesions.
How Supplied
- Trifluridine Ophthalmic Solution, 1% in a 7.5 mL fill packaged in a natural plastic bottle with a natural plastic flat tip and a white plastic closure.
Storage
Store under refrigeration 2°-8°C (36°-46°F).
Images
Drug Images
{{#ask: Page Name::Trifluridine |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel

[File:Tri 03.jpg {{#ask: Label Page::Trifluridine |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Trifluridine in the drug label.
Precautions with Alcohol
- Alcohol-Trifluridine interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- TRIFLURIDINE ®[1]
Look-Alike Drug Names
There is limited information regarding Trifluridine Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.