Trabectedin
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Martin Nino [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Trabectedin is an alkylating drug that is FDA approved for the treatment of patients with unresectable or metastatic liposarcoma or leiomyosarcoma who received a prior anthracycline-containing regimen. Common adverse reactions include nausea, fatigue, vomiting, constipation, decreased appetite, diarrhea, peripheral edema, dyspnea, and headache (≥20%).
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
Trabectedin is indicated for the treatment of patients with unresectable or metastatic liposarcoma or leiomyosarcoma who received a prior anthracycline-containing regimen
Dosage
- Recommended Dose and Schedule
- The recommended dose is 1.5 mg/m2 administered as an intravenous infusion over 24 hours through a central venous line every 21 days (3 weeks), until disease progression or unacceptable toxicity, in patients with normal bilirubin and AST or ALT less than or equal to 2.5 times the upper limit of normal.
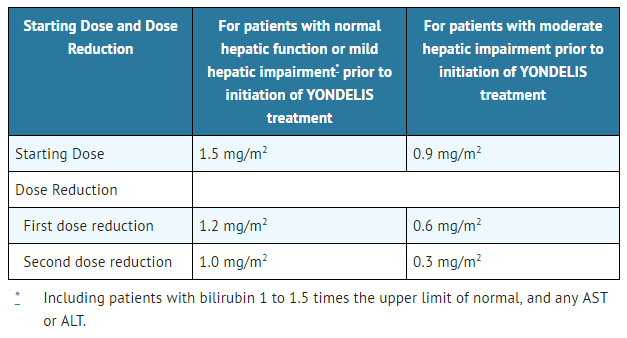
- Hepatic Impairment: The recommended dose is 0.9 mg/m2 in patients with moderate hepatic impairment (bilirubin levels 1.5 times to 3 times the upper limit of normal, and AST and ALT less than 8 times the upper limit of normal). Do not administer Trabectedin to patients with severe hepatic impairment (bilirubin levels above 3 times to 10 times the upper limit of normal, and any AST and ALT).
- Premedication
- Administer dexamethasone 20 mg intravenously 30 minutes prior to each dose of Trabectedin.
- Dose Modifications
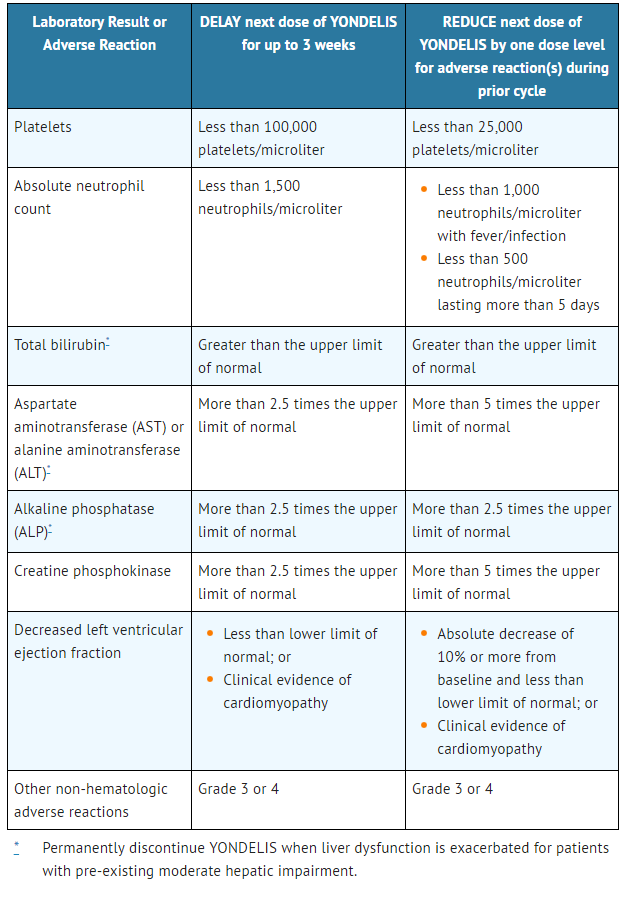
- Permanently discontinue Trabectedin for:
- - Persistent adverse reactions requiring a delay in dosing of more than 3 weeks.
- - Adverse reactions requiring dose reduction following Trabectedin administered at 1.0 mg/m2 for patients with normal hepatic function or at 0.3 mg/m2 for patients with pre-existing moderate hepatic impairment.
- - Severe liver dysfunction all of the following: bilirubin two times the upper limit of normal and AST or ALT three times the upper limit of normal with alkaline phosphatase less than two times the upper limit of normal in the prior treatment cycle for patients with normal liver function at baseline.
- - Exacerbation of liver dysfunction in patients with pre-existing moderate hepatic impairment.
- The recommended dose modifications for adverse reactions are listed in Table 1. Once reduced, the dose of Trabectedin should not be increased in subsequent treatment cycles.
- Table 1: Recommended Dose Modification
YONDELIS: Trabectedin's Brand name
The recommended starting doses and dose reductions for Trabectedin are listed in Table 2:
- Table 2: Recommended Starting Doses and Dose Reductions
YONDELIS: Trabectedin's Brand name
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Trabectedin in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Trabectedin in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Safety and effectiveness in pediatric patients have not been established.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Trabectedin in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Trabectedin in pediatric patients.
Contraindications
Trabectedin is contraindicated in patients with known severe hypersensitivity, including anaphylaxis, to trabectedin.
Warnings
Neutropenic Sepsis
Neutropenic sepsis, including fatal cases, can occur with Trabectedin. In Trial 1, the incidence of Grade 3 or 4 neutropenia, based on laboratory values, in patients receiving Trabectedin was 43% (161/378). The median time to the first occurrence of Grade 3 or 4 neutropenia was 16 days (range: 8 days to 9.7 months); the median time to complete resolution of neutropenia was 13 days (range: 3 days to 2.3 months). Febrile neutropenia (fever ≥38.5°C with Grade 3 or 4 neutropenia) occurred in 18 patients (5%) treated with Trabectedin. Ten patients (2.6%) experienced neutropenic sepsis, 5 of whom had febrile neutropenia, which was fatal in 4 patients (1.1%).
Assess neutrophil count prior to administration of each dose of Trabectedin and periodically throughout the treatment cycle. Withhold Trabectedin for neutrophil counts of less than 1,500 cells/microliter on the day of dosing. Permanently reduce the dose of Trabectedin for life-threatening or prolonged, severe neutropenia in the preceding cycle.
Rhabdomyolysis
Trabectedin can cause rhabdomyolysis and musculoskeletal toxicity. In Trial 1, rhabdomyolysis leading to death occurred in 3 (0.8%) of the 378 patients receiving Trabectedin. Elevations in creatine phosphokinase (CPK) occurred in 122 (32%) of the 378 patients receiving Trabectedin, including Grade 3 or 4 CPK elevation in 24 patients (6%), compared to 15 (9%) of the 172 patients receiving dacarbazine with any CPK elevation, including 1 patient (0.6%) with Grade 3 CPK elevation. Among the 24 patients receiving Trabectedin with Grade 3 or 4 CPK elevation, renal failure occurred in 11 patients (2.9%); rhabdomyolysis with the complication of renal failure occurred in 4 of these 11 patients (1.1%). The median time to first occurrence of Grade 3 or 4 CPK elevations was 2 months (range: 1 to 11.5 months). The median time to complete resolution was 14 days (range: 5 days to 1 month).
Assess CPK levels prior to each administration of Trabectedin. Withhold Trabectedin for serum CPK levels more than 2.5 times the upper limit of normal. Permanently discontinue Trabectedin for rhabdomyolysis.
Hepatotoxicity
Hepatotoxicity, including hepatic failure, can occur with Trabectedin. Patients with serum bilirubin levels above the upper limit of normal or AST or ALT levels >2.5 × upper limit of normal were not enrolled in Trial 1. In Trial 1, the incidence of Grade 3–4 elevated liver function tests (LFTs; defined as elevations in ALT, AST, total bilirubin, or alkaline phosphatase) was 35% (134/378) in patients receiving Trabectedin. The median time to development of Grade 3–4 elevation in ALT or AST was 29 days (range: 3 days to 11.5 months). Of the 134 patients with Grade 3–4 elevations in LFTs, 114 (85%) experienced complete resolution with the median time to complete resolution of 13 days (range: 4 days to 4.4 months).
In Trial 1, the incidence of drug-induced liver injury (defined as concurrent elevation in ALT or AST of more than three times the upper limit of normal, alkaline phosphatase less than two times the upper limit of normal, and total bilirubin at least two times the upper limit of normal) was 1.3% (5/378) in patients receiving Trabectedin. ALT or AST elevation greater than eight times the upper limit of normal occurred in 18% (67/378) of patients receiving Trabectedin.
Assess LFTs prior to each administration of Trabectedin and as clinically indicated based on underlying severity of pre-existing hepatic impairment. Manage elevated LFTs with treatment interruption, dose reduction, or permanent discontinuation based on severity and duration of LFT abnormality.
Cardiomyopathy
Cardiomyopathy including cardiac failure, congestive heart failure, ejection fraction decreased, diastolic dysfunction, or right ventricular dysfunction can occur with Trabectedin. In Trial 1, patients with a history of New York Heart Association Class II to IV heart failure or abnormal left ventricular ejection fraction (LVEF) at baseline were ineligible. In Trial 1, cardiomyopathy occurred in 23 patients (6%) receiving Trabectedin and in four patients (2.3%) receiving dacarbazine. Grade 3 or 4 cardiomyopathy occurred in 15 patients (4%) receiving Trabectedin and 2 patients (1.2%) receiving dacarbazine; cardiomyopathy leading to death occurred in 1 patient (0.3%) receiving Trabectedin and in none of the patients receiving dacarbazine. The median time to development of Grade 3 or 4 cardiomyopathy in patients receiving Trabectedin was 5.3 months (range: 26 days to 15.3 months).
Assess LVEF by echocardiogram or multigated acquisition (MUGA) scan before initiation of Trabectedin and at 2- to 3-month intervals thereafter until Trabectedin is discontinued. Withhold Trabectedin for LVEF below lower limit of normal. Permanently discontinue Trabectedin for symptomatic cardiomyopathy or persistent left ventricular dysfunction that does not recover to lower limit of normal within 3 weeks.
Extravasation Resulting in Tissue Necrosis
Extravasation of Trabectedin, resulting in tissue necrosis requiring debridement, can occur. Evidence of tissue necrosis can occur more than 1 week after the extravasation. There is no specific antidote for extravasation of Trabectedin. Administer Trabectedin through a central venous line.
Embryofetal Toxicity
Based on its mechanism of action, Trabectedin can cause fetal harm when administered to a pregnant woman. Advise females of reproductive potential to use effective contraception during therapy and for at least 2 months after the last dose of Trabectedin. Advise males with female partners of reproductive potential to use effective contraception during therapy and for at least 5 months after the last dose of Trabectedin.
Adverse Reactions
Clinical Trials Experience
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Anaphylaxis
- Neutropenic Sepsis
- Rhabdomyolysis
- Hepatotoxicity
- Cardiomyopathy
- Extravasation Resulting in Tissue Necrosis
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure to Trabectedin in 755 patients with soft tissue sarcoma including 197 (26%) patients exposed to Trabectedin for greater than or equal to 6 months and 57 (8%) patients exposed to Trabectedin for greater than or equal to 1 year. The safety of Trabectedin was evaluated in six open-label, single-arm trials, in which 377 patients received Trabectedin and one open-label, randomized, active-controlled clinical trial in which 378 patients received Trabectedin (Trial 1). All patients received Trabectedin at the recommended dosing regimen of 1.5 mg/m2 administered as an intravenous infusion over 24 hours once every 3 weeks (q3wk, 24-h). The median age was 54 years (range: 18 to 81 years), 63% were female, and all patients had metastatic soft tissue sarcoma.
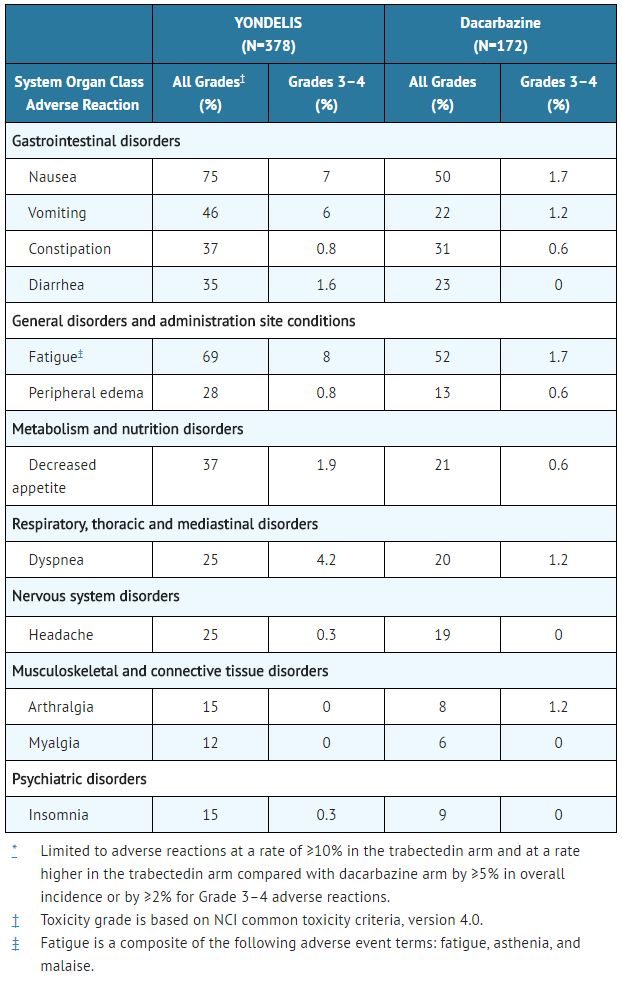
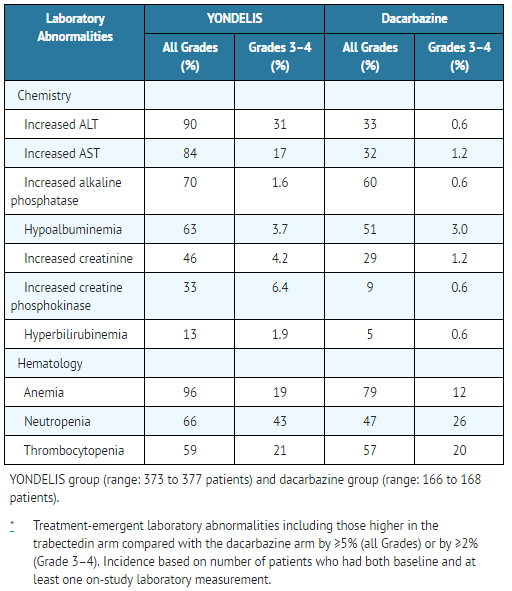
Tables 3 and 4 present selected adverse reactions and laboratory abnormalities, respectively, observed in Trial 1, an open-label, randomized (2:1), active-controlled trial in which 550 patients with previously treated leiomyosarcoma or liposarcoma (dedifferentiated, myxoid round cell, or pleomorphic) received Trabectedin 1.5 mg/m2 intravenous infusion over 24 hours once every 3 weeks (n=378) or dacarbazine 1000 mg/m2 intravenous infusion over 20 to 120 minutes once every 3 weeks (n=172). All patients treated with Trabectedin were required to receive dexamethasone 20 mg intravenous injection 30 minutes prior to start of the Trabectedin infusion.
In Trial 1, patients had been previously treated with an anthracycline- and ifosfamide-containing regimen or with an anthracycline-containing regimen and one additional cytotoxic chemotherapy regimen. The trial excluded patients with known central nervous system metastasis, elevated serum bilirubin or significant chronic liver disease, such as cirrhosis or active hepatitis, and history of myocardial infarction within 6 months, history of New York Heart Association Class II to IV heart failure, or abnormal left ventricular ejection fraction at baseline. The median age of patients in Trial 1 was 57 years (range: 17 to 81 years), with 69% female, 77% White, 12% Black or African American, 4% Asian, and <1% American Indian or Alaska Native. The median duration of exposure to trabectedin was 13 weeks (range: 1 to 127 weeks) with 30% of patients exposed to Trabectedin for greater than 6 months and 7% of patients exposed to Trabectedin for greater than 1 year.
In Trial 1, adverse reactions resulting in permanent discontinuation of Trabectedin occurred in 26% (98/378) of patients; the most common were increased liver tests (defined as ALT, AST, alkaline phosphatase, bilirubin) (5.6%), thrombocytopenia (3.4%), fatigue (1.6%), increased creatine phosphokinase (1.1%), and decreased ejection fraction (1.1%). Adverse reactions that led to dose reductions occurred in 42% (158/378) of patients treated with Trabectedin; the most common were increased liver tests (24%), neutropenia (including febrile neutropenia) (8%), thrombocytopenia (4.2%), fatigue (3.7%), increased creatine phosphokinase (2.4%), nausea (1.1%), and vomiting (1.1%). Adverse reactions led to dose interruptions in 52% (198/378) of patients treated with Trabectedin; the most common were neutropenia (31%), thrombocytopenia (15%), increased liver tests (6%), fatigue (2.9%), anemia (2.6%), increased creatinine (1.1%), and nausea (1.1%).
The most common adverse reactions (≥20%) were nausea, fatigue, vomiting, constipation, decreased appetite, diarrhea, peripheral edema, dyspnea, and headache. The most common laboratory abnormalities (≥20%) were increases in AST or ALT, increased alkaline phosphatase, hypoalbuminemia, increased creatinine, increased creatine phosphokinase, anemia, neutropenia, and thrombocytopenia.
- Table 3: Selected Adverse Reactions* Occurring in ≥10% of Patients Receiving Trabectedin and at a Higher Incidence than in the Control Arm - Trial 1
YONDELIS: Trabectedin's Brand name
Other clinically important adverse reactions observed in <10% of patients (N=755) with soft tissue sarcoma receiving Trabectedin were:
- Nervous system disorders: peripheral neuropathy, paresthesia, hypoesthesia.
- Respiratory, thoracic, and mediastinal disorders: pulmonary embolism.
- Table 4: Incidence of Selected Treatment-Emergent Laboratory Abnormalities* - Trial 1
YONDELIS: Trabectedin's Brand name
Postmarketing Experience
There is limited information regarding Trabectedin Postmarketing Experience in the drug label.
Drug Interactions
Effect of Cytochrome CYP3A Inhibitors
Coadministration of Trabectedin with ketoconazole, a strong CYP3A inhibitor, increased systemic exposure of trabectedin by 66%. Avoid using strong CYP3A inhibitors (e.g., oral ketoconazole, itraconazole, posaconazole, voriconazole, clarithromycin, telithromycin, indinavir,lopinavir, ritonavir, boceprevir, nelfinavir,saquinavir, telaprevir, nefazodone, conivaptan) in patients taking Trabectedin. Avoid taking grapefruit or grapefruit juice during Trabectedin treatment. If a strong CYP3A inhibitor for short-term use (i.e., less than 14 days) must be used, administer the strong CYP3A inhibitor 1 week after the Trabectedin infusion, and discontinue it the day prior to the next Trabectedin infusion.
Effect of Cytochrome CYP3A Inducers
Coadministration of Trabectedin with rifampin, a strong CYP3A inducer, decreased systemic exposure of trabectedin by 31%. Avoid using strong CYP3A inducers (e.g., rifampin, phenobarbital, St. John's wort) in patients taking Trabectedin.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): N
. Based on its mechanism of action, trabectedin can cause fetal harm when administered during pregnancy. There are no available data with the use of Trabectedin during pregnancy. Animal reproductive and developmental studies at relevant doses have not been conducted with trabectedin; however, placental transfer of trabectedin was demonstrated in pregnant rats. Advise pregnant woman of the potential risk to a fetus. The background risk of major birth defects and miscarriage for the indicated population are unknown; however, the background risk in the U.S. general population of major birth defects is 2 to 4% and of miscarriage is 15 to 20% of clinically recognized pregnancies.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Trabectedin in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Trabectedin during labor and delivery.
Nursing Mothers
There are no data on the presence of trabectedin in human milk, the effects on the breastfed infant, or the effects on milk production. Because of the potential for serious adverse reactions from Trabectedin in breastfed infants, advise a nursing woman to discontinue nursing during treatment with Trabectedin.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Geriatic Use
Clinical studies of Trabectedin did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
Gender
There is no FDA guidance on the use of Trabectedin with respect to specific gender populations.
Race
There is no FDA guidance on the use of Trabectedin with respect to specific racial populations.
Renal Impairment
No dose adjustment is recommended in patients with mild (creatinine clearance (CLcr) 60–89 mL/min) or moderate (CLcr of 30–59 mL/min) renal impairment.
The pharmacokinetics of trabectedin has not been evaluated in patients with severe renal impairment (CLcr <30 mL/min) or end stage renal disease.
Hepatic Impairment
The mean trabectedin exposure was (97%) higher in patients with moderate (bilirubin levels 1.5 to 3.0 times the upper limit of normal, and AST and ALT less than 8 times the upper limit of normal) hepatic impairment compared to patients with normal (total bilirubin ≤ the upper limit of normal, and AST and ALT ≤ the upper limit of normal) liver function. Reduce Trabectedin dose in patients with moderate hepatic impairment.
Do not administer Trabectedin to patients with severe hepatic impairment (bilirubin levels above 3 times to 10 times the upper limit of normal, and any AST and ALT).
Females of Reproductive Potential and Males
-
- Females: Advise female patients of reproductive potential to use effective contraception during and for 2 months after the last dose of Trabectedin.
- Males: Trabectedin may damage spermatozoa, resulting in possible genetic and fetal abnormalities. Advise males with a female sexual partner of reproductive potential to use effective contraception during and for 5 months after the last dose of Trabectedin.
- Infertility: Trabectedin may result in decreased fertility in males and females.
Immunocompromised Patients
There is no FDA guidance one the use of Trabectedin in patients who are immunocompromised.
Administration and Monitoring
Administration
- Preparation
- Trabectedin is a cytotoxic drug. Follow applicable special handling and disposal procedures.
- Using aseptic technique, inject 20 mL of Sterile Water for Injection, USP into the vial. Shake the vial until complete dissolution. The reconstituted solution is clear, colorless to pale brownish-yellow, and contains 0.05 mg/mL of trabectedin.
- Inspect for particulate matter and discoloration prior to further dilution. Discard vial if particles or discoloration are observed.
- Immediately following reconstitution, withdraw the calculated volume of trabectedin and further dilute in 500 mL of 0.9% Sodium Chloride, USP or 5% Dextrose Injection, USP.
- Do not mix Trabectedin with other drugs.
- Discard any remaining solution within 30 hours of reconstituting the lyophilized powder.
- Trabectedin diluted solution is compatible with Type I colorless glass vials, polyvinylchloride (PVC) and polyethylene (PE) bags and tubing, PE and polypropylene (PP) mixture bags, polyethersulfone (PES) in-line filters, titanium, platinum or plastic ports, silicone and polyurethane catheters, and pumps having contact surfaces made of PVC, PE, or PE/PP.
- Administration
- Infuse the reconstituted, diluted solution over 24 hours through a central venous line using an infusion set with a 0.2 micron polyethersulfone (PES) in-line filter to reduce the risk of exposure to adventitious pathogens that may be introduced during solution preparation.
- Complete infusion within 30 hours of initial reconstitution. Discard any unused portion of the reconstituted product or of the infusion solution.
Monitoring
There is limited information regarding Trabectedin Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Trabectedin and IV administrations.
Overdosage
There is no specific antidote for Trabectedin. Hemodialysis is not expected to enhance the elimination of Trabectedin because trabectedin is highly bound to plasma proteins (97%) and not significantly renally excreted.
Pharmacology
Mechanism of Action
Trabectedin is an alkylating drug that binds guanine residues in the minor groove of DNA, forming adducts and resulting in a bending of the DNA helix towards the major groove. Adduct formation triggers a cascade of events that can affect the subsequent activity of DNA binding proteins, including some transcription factors, and DNA repair pathways, resulting in perturbation of the cell cycle and eventual cell death.
Structure
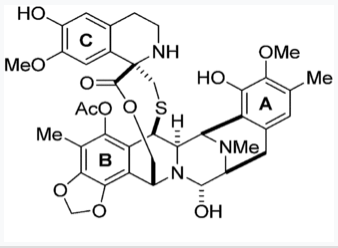
Trabectedin is an alkylating agent with the chemical name (1'R,6R,6aR,7R,13S,14S,16R)-5-(acetyloxy)-3',4',6,6a,7,13,14,16-octahydro-6',8,14-trihydroxy-7',9-dimethoxy-4,10,23-trimethyl-spiro[6,16-(epithiopropanoxymethano)-7,13-imino-12H-1,3-dioxolo[7,8]isoquino[3,2-b][3]benzazocine-20,1'(2'H)-isoquinolin]-19-one. The molecular formula is C39H43N3O11S. The molecular weight is 761.84 daltons. The chemical structure is shown below:
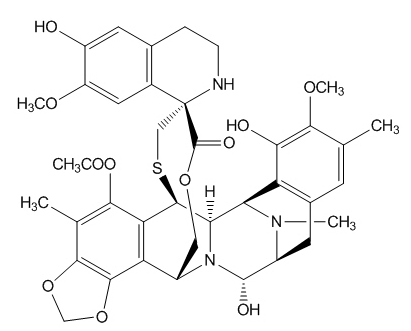
Trabectedin is hydrophobic and has a low solubility in water.
Trabectedin for injection is supplied as a sterile lyophilized white to off-white powder/cake in a single-dose vial. Each single-dose vial contains 1 mg of trabectedin, 27.2 mg potassium dihydrogen phosphate, 400 mg sucrose, and phosphoric acid and potassium hydroxide (for pH adjustment to 3.6 – 4.2).
Pharmacodynamics
- The effect of trabectedin on the QT/QTc interval was evaluated in 75 patients who received placebo on day 1 and trabectedin (1.3 mg/m2) as a 3-hour intravenous infusion on day 2. No patients in the study showed a QTc interval exceeding 500 msec or more than 60 msec increase from baseline, and no large changes in the mean QTc interval (i.e., >20 msec) were observed.
Pharmacokinetics
The pharmacokinetics of trabectedin is characterized by a rapid decline phase at the end of the infusion and slower exponential phases. Population pharmacokinetic analyses suggest that the pharmacokinetics of trabectedin is dose-proportional (over the dose range of 0.024 to 1.8 mg/m2) and exposure is time-independent. No accumulation of trabectedin in plasma is observed upon repeated administrations every 3 weeks.
- Binding of trabectedin to human plasma proteins was approximately 97%, independent of trabectedin concentrations ranging from 10 ng/mL to 100 ng/mL. Steady state volume of distribution of trabectedin exceeds 5000 L.
- Elimination
- Trabectedin was extensively metabolized with negligible unchanged drug in urine and feces following administration of trabectedin to humans.
- Excretion
- In patients with solid tumors, following a 3-hour or a 24-hour intravenous infusion of 14C-labeled trabectedin, 64% of the total administered radioactive dose was recovered in 24 days, with 58% in feces and 6% in urine.
- Specific Populations
- The following population characteristics are not associated with a clinically significant effect on the pharmacokinetics of trabectedin: sex, age (19 to 83 years), body weight (36 to 148 kg), body surface area (0.9 to 2.8 m2), mild hepatic impairment, or mild to moderate renal impairment. The effects of severe hepatic impairment, severe renal impairment or end stage renal disease on trabectedin exposure are unknown.
- Hepatic Impairment
- The geometric mean dose normalized trabectedin exposure (AUC) increased by 97% (90% CI: 20%, 222%) in patients with moderate hepatic impairment following administration of a single Trabectedin dose of 0.58 mg/m2 or 0.9 mg/m2 compared to patients with normal liver function following administration of a single Trabectedin dose of 1.3 mg/m2.
- Drug Interactions
- Effect of Strong CYP3A Inhibitors on Trabectedin
- Coadministration of multiple doses of ketoconazole (200 mg twice daily for 7.5 days) with a single dose of Trabectedin (0.58 mg/m2) on day 1 increased trabectedin dose-normalized AUC by 66% and Cmax by 22% compared to a single Trabectedin dose (1.3 mg/m2) given alone.
- Effect of Strong CYP3A Inducers on Trabectedin
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
Trabectedin is genotoxic in both in vitro and in vivo studies. Long-term carcinogenicity studies have not been performed.
Fertility studies with trabectedin were not performed. In male rats there were limited histopathological signs of hemorrhage and degeneration in the testes following repeated administration of trabectedin at doses approximately 0.2 times the 1.5 mg/m2 human dose based on body surface area.
Clinical Studies
The clinical efficacy and safety of Trabectedin in patients with metastatic or recurrent leiomyosarcoma or liposarcoma were demonstrated in Trial 1, a randomized (2:1), open-label, active-controlled trial comparing treatment with Trabectedin 1.5 mg/m2 as a 24-hour continuous intravenous infusion once every 3 weeks to dacarbazine 1000 mg/m2 intravenous infusion (20 to 120 minutes) once every 3 weeks. Treatment continued in both arms until disease progression or unacceptable toxicity; all patients in the Trabectedin arm were required to receive dexamethasone 20 mg intravenous injection prior to each Trabectedin infusion. Patients were required to have unresectable, locally advanced or metastatic leiomyosarcoma or liposarcoma (dedifferentiated, myxoid round cell, or pleomorphic) and previous treatment with an anthracycline- and ifosfamide-containing regimen or an anthracycline-containing regimen and one additional cytotoxic chemotherapy regimen. Randomization was stratified by subtype of soft tissue sarcoma (leiomyosarcoma vs. liposarcoma), ECOG performance status (0 vs. 1), and number of prior chemotherapy regimens (1 vs. ≥2). The efficacy outcome measures were investigator-assessed progression-free survival (PFS) according to the Response Evaluation Criteria in Solid Tumors (RECIST v1.1), overall survival (OS), objective response rate (ORR), and duration of response (DOR). Patients in the dacarbazine arm were not offered Trabectedin at the time of disease progression.
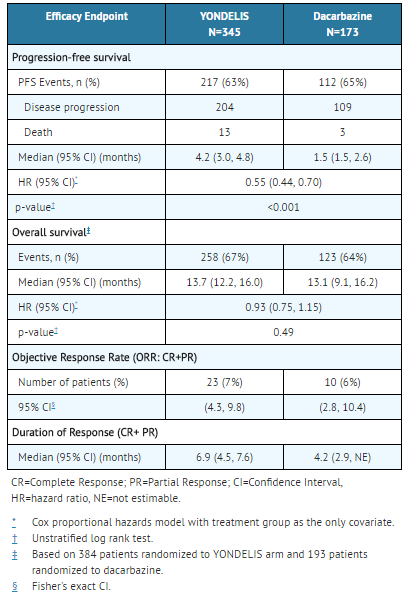
A total of 518 patients were randomized, 345 to the Trabectedin arm and 173 patients to the dacarbazine arm. The median patient age was 56 years (range: 17 to 81); 30% were male; 76% White, 12% Black, and 4% Asian; 73% had leiomyosarcomas and 27% liposarcomas; 49% had an ECOG PS of 0; and 89% received ≥2 prior chemotherapy regimens. The most common (≥20%) pre-study chemotherapeutic agents administered were doxorubicin (90%), gemcitabine (81%), docetaxel (74%), and ifosfamide (59%). Approximately 10% of patients had received pazopanib.
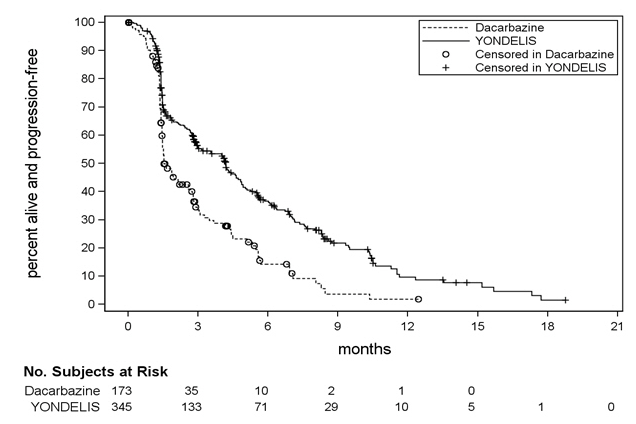
Trial 1 demonstrated a statistically significant improvement in PFS. An exploratory analysis of independent radiology committee-determined PFS, in a subgroup consisting of approximately 60% of the total population, provided similar results to the investigator-determined PFS. Efficacy results from Trial 1 are presented in the table below.
- Table 5: Efficacy Results for Trial 1
YONDELIS: Trabectedin's Brand name
- Figure 1: Kaplan-Meier Curves of Progression-Free Survival in Trial 1
YONDELIS: Trabectedin's Brand name
How Supplied
Trabectedin is supplied in a glass vial containing 1 mg trabectedin. Each carton contains one vial (NDC: 59676-610-01).
Storage
Store Trabectedin vials in a refrigerator at 2ºC to 8ºC (36ºF to 46ºF).
Trabectedin is a cytotoxic drug. Follow applicable special handling and disposal procedures.
Images
Drug Images
{{#ask: Page Name::Trabectedin |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
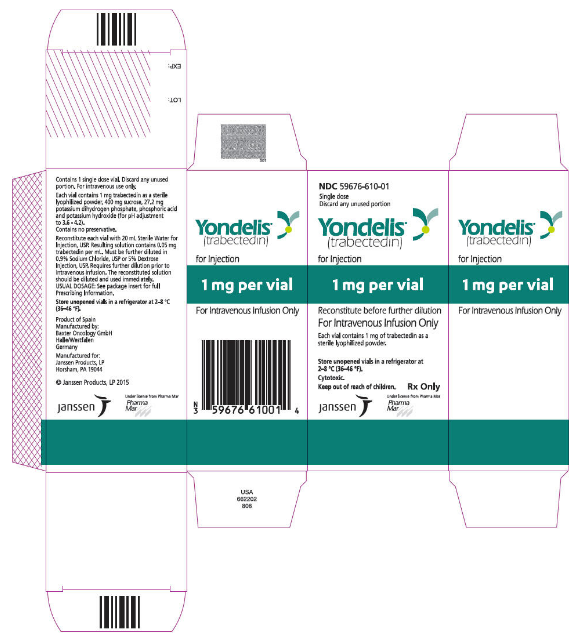
{{#ask: Label Page::Trabectedin |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
- Myelosuppression: Inform patients of the risks of myelosuppression. Instruct patients to immediately contact their healthcare provider for fever or unusual bruising, bleeding, tiredness, or paleness.
- Rhabdomyolysis: Advise patients to contact their healthcare provider if they experience severe muscle pain or weakness.
- Hepatotoxicity: Advise patients to contact their healthcare provider immediately for yellowing of skin and eyes (jaundice), pain in the upper right quadrant, severe nausea or vomiting, difficulty in concentrating, disorientation, or confusion.
- Cardiomyopathy: Advise patients to contact their healthcare provider for new onset chest pain, shortness of breath, fatigue, lower extremity edema, or heart palpitations.
- Hypersensitivity: Advise patients to seek immediate medical attention for symptoms of allergic reactions including difficulty breathing, chest tightness, wheezing, severe dizziness or light-headedness, swelling of the lips or skin rash.
- Extravasation: Inform patients of the risks of extravasation and to notify their healthcare provider for redness, swelling, itchiness and discomfort or leakage at the injection site.
- Embryofetal toxicity: Advise pregnant women of the potential risk to a fetus. Advise females to contact their healthcare provider if they become pregnant, or if pregnancy is suspected, during treatment with Trabectedin.
- Females and males of reproductive potential: Advise females of reproductive potential to use effective contraception during treatment with Trabectedin and for at least 2 months after last dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with Trabectedin and for at least 5 months after the last dose.
- Lactation: Advise females not to breastfeed during treatment with Trabectedin.

Precautions with Alcohol
Alcohol-Trabectedin interaction has not been established. Talk to your doctor regarding the effects of taking alcohol with this medication.
Brand Names
YONDELIS®
Look-Alike Drug Names
There is limited information regarding Trabectedin Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.