Terpenoid
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
The terpenoids, sometimes referred to as isoprenoids, are a large and diverse class of naturally occurring organic chemicals similar to terpenes, derived from five-carbon isoprene units assembled and modified in thousands of ways. Most are multicyclic structures which differ from one another not only in functional groups, but also in their basic carbon skeletons. These lipids can be found in all classes of living things, and are the largest group of natural products.
Plant terpenoids are extensively used for their aromatic qualities. They play a role in traditional herbal remedies and are under investigation for antibacterial, antineoplastic and other pharmaceutical effects. Terpenoids contribute to the scent of eucalyptus, the flavors of cinnamon, cloves and ginger and the color of yellow flowers. Well-known terpenoids include citral, menthol, camphor and the cannabinoids found in the Cannabis plant.
The steroids and sterols in animals are biologically produced from terpenoid precursors. Sometimes terpenoids are added to proteins, e.g. to enhance their attachment to the cell membrane; this is known as isoprenylation.
Many of these are substrates for plant Cytochrome P450.
Structure and classification
Terpenes are hydrocarbons resulting from the combination of several isoprene units. Terpenoids can be thought of as modified terpenes, where methyl groups have been moved or removed, or oxygen atoms added. (Some authors use the term "terpene" more broadly, to include the terpenoids.) Just like terpenes, the terpenoids can be classified according to the number of isoprene units used:
- Monoterpenoids, 2 isoprene units
- Sesquiterpenoids, 3 isoprene units
- Diterpenoids, 4 isoprene units
- Sesterterpenoids, 5 isoprene units
- Triterpenoids, 6 isoprene units
- Tetraterpenoids, 8 isoprene units
- Polyterpenoids with a larger number of isoprene units
Terpenoids can also be classified according to the number of cyclic structures they contain.
Biosynthesis
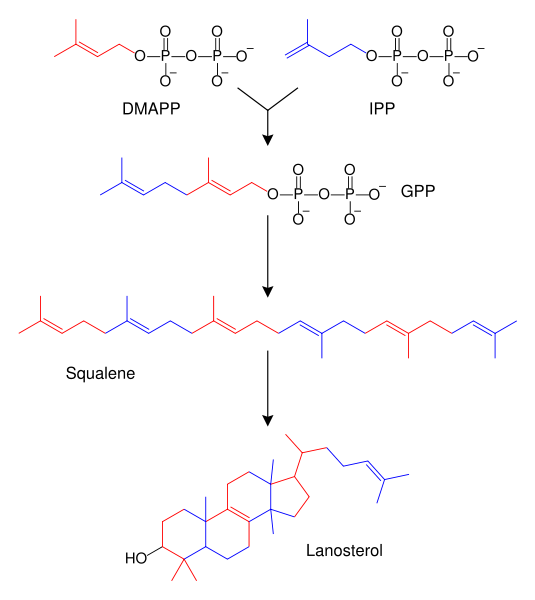
There are two metabolic pathways of creating terpenoids:
Mevalonic acid pathway
Many organisms manufacture terpenoids through the HMG-CoA reductase pathway, the pathway that also produces cholesterol. The reactions take place in the cytosol. The pathway was discovered in the 1950s.
MEP/DOXP pathway
The 2-C-methyl-D-erythritol 4-phosphate/1-deoxy-D-xylulose 5-phosphate pathway (MEP/DOXP pathway), also known as non-mevalonate pathway or mevalonic acid independent pathway, takes place in the plastids of plants and apicomplexan protozoa as well as in many bacteria. It was discovered in the late 1980s.
Pyruvate and glyceraldehyde 3-phosphate are converted by DOXP synthase (Dxs) to 1-deoxy-D-xylulose 5-phosphate, and by DOXP reductase (Dxr, IspC) to 2-C-methyl-D-erythritol 4-phosphate (MEP). The subsequent three reaction steps catalyzed by 4-diphosphocytidyl-2-C-methyl-D-erythritol synthase (YgbP, IspD), 4-diphosphocytidyl-2-C-methyl-D-erythritol kinase (YchB, IspE), and 2-C-methyl-D-erythritol 2,4-cyclodiphosphate synthase (YgbB, IspF) mediate the formation of 2-C-methyl-D-erythritol 2,4-cyclopyrophosphate (MEcPP). Finally, MEcPP is converted to (E)-4-hydroxy-3-methyl-but-2-enyl pyrophosphate (HMB-PP) by HMB-PP synthase (GcpE, IspG), and HMB-PP is converted to isopentenyl pyrophosphate (IPP) and dimethylallyl pyrophosphate (DMAPP) by HMB-PP reductase (LytB, IspH).
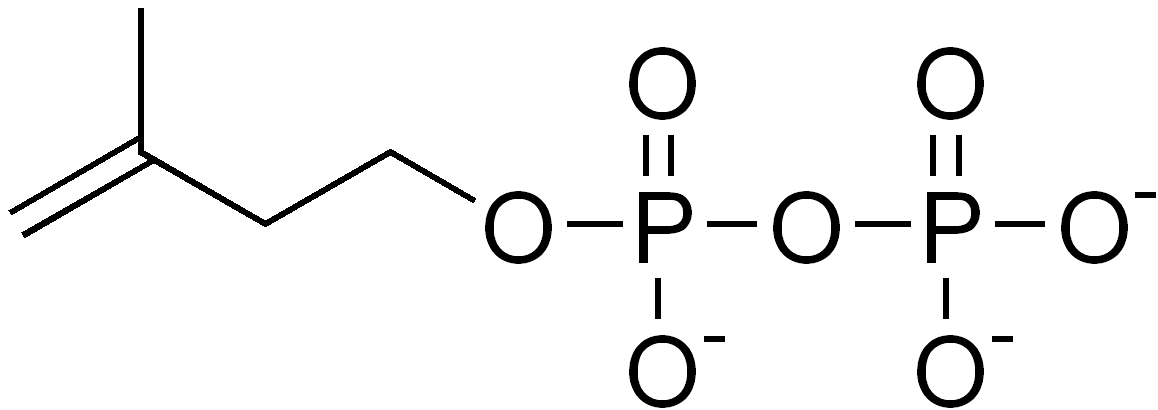
IPP and DMAPP are the end products in either pathway, and are the precursors of isoprene, monoterpenoids (10-carbon), diterpenoids (20-carbon), carotenoids (40-carbon), chlorophylls and plastoquinone-9 (45-carbon). Synthesis of all higher terpenoids proceeds via formation of geranyl pyrophosphate (GPP), farnesyl pyrophosphate (FPP), and geranylgeranyl pyrophosphate (GGPP).
Although both pathways, MVA and MEP, are mutually exclusive in most organisms, interactions between them have been reported in plants and few bacteria species.
| Organism | Pathways |
|---|---|
| Eubacteria | MVA or MEP |
| Archaea | MVA |
| Green Algae | MEP |
| Plants | MVA and MEP |
| Animals | MVA |
| Fungi | MVA |