Splenic infarction pathophysiology
|
Splenic infarction Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Splenic infarction pathophysiology On the Web |
|
American Roentgen Ray Society Images of Splenic infarction pathophysiology |
|
Risk calculators and risk factors for Splenic infarction pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Pathophysiology
Related Anatomy
The arterial supply to the spleen consists of the splenic artery (a branch of the celiac axis) and the short gastric arteries (branches of the left gastroepiploic artery), which supply the upper pole of the spleen. Even with occlusion of the main splenic artery, collateral flow from the short gastric arteries often may preserve some or all of the splenic parenchyma.
Within the spleen, the arterial supply is segmental. Occlusion of these secondary branches results in the classic wedge-shaped infarct. Most commonly, these infarcts contract and fibrose over time, as demonstrated by the sickle hemoglobinopathies in which repeated episodes of infarction ultimately result in auto-infarction of the spleen.
Pathophysiology
- In diseases such as chronic myeloid leukemia that result in massive splenomegaly secondary to the malignant infiltrative process, segmental infarcts are considered the result of outstripping the available blood supply.
- Splenic vein thrombosis, most commonly the result of pancreatitis or surgery, can result in venous infarction.
- Unusual causes of splenic infarction include malaria, pancreatitis, cocaine use, and splenic infarction occurring as a late complication of liver transplantation.
- An anatomic variant that renders the spleen more susceptible to global infarction is that of the wandering spleen. The spleen is attached by a long vascular pedicle, without the usual fixating ligaments, to the diaphragm, colon, left kidney, and lateral abdominal wall. This allows torsion of the freely mobile spleen around its vascular pedicle, occluding the blood supply and leading to infarction. The treatment is surgical fixation of the spleen (i.e., splenopexy), allowing preservation of the spleen if it has not infarcted as a result of occlusion of the splenic hilum. Techniques include suturing the spleen to the surrounding structures, wrapping it in omentum or mesh prior to suture fixation, or placing it in a surgically created retroperitoneal pouch.
Pathology
A. Brief Descriptions:
- Nearly 99% of infarcts are caused by thromboembolic events, and almost all are the result of arterial occlusions.
- White infarcts are encountered with arterial occlusion and in solid tissues.
B. Gross Findings:
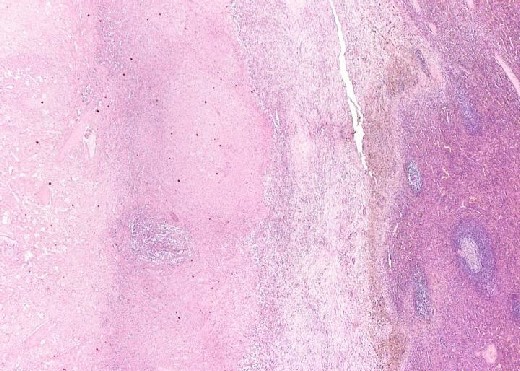
- Recent infarcts are hemorrhagic,whereas older, more fibrotic infarcts are pale yellow-gray.
C. Micro Findings:
- Necrotic area with homogenous pinkish appearance.
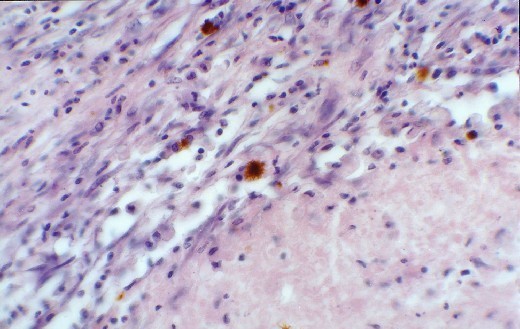
- Hematoidin crystals can be found in this section.
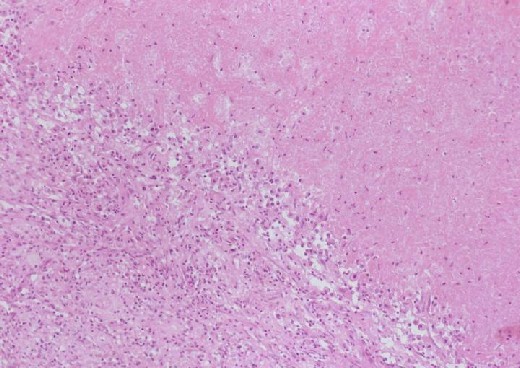
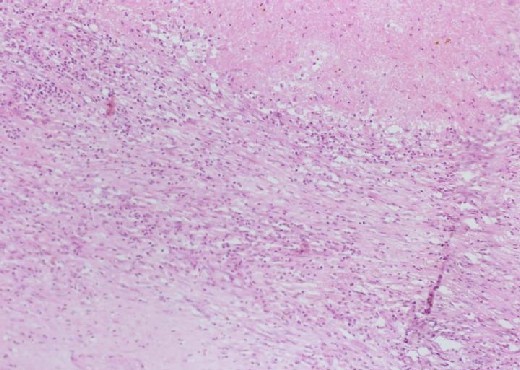
- Inflammatory cells seated on the margin of infarct area.
Histolopathological Findings
- Pathologic examination of the resected spleen may provide information regarding the pathogenesis of the infarct, e.g. evidence of septic or atheromatous emboli or the presence of an infectious etiology.
-
-
Inflammatory cells seated on the margin of infarct area
-
Inflammatory cells
-
Hemosiderin laden macrophages in the more fibrous areas (junction of infarction)
-
Multiple infarcts in a spleen greatly enlarged by malignant lymphoma. The multiple areas of infarction are well demarcated