Q fever
For patient information click here
| Q fever | |
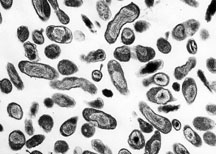 | |
|---|---|
| Organism Responsible for Q fever, Rocky Mountain Laboratories, NIAID, NIH | |
| ICD-10 | A78 |
| ICD-9 | 083.0 |
| MeSH | D011778 |
|
Q fever Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Q fever On the Web |
|
American Roentgen Ray Society Images of Q fever |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Historical perspective
Epidemiology & Demographics
Pathophysiology
History & Symptoms
Diagnosis
Lab Tests
Natural history, Complications, and Prognosis
Treatment
Medical Therapy
Primary Prevention
Other
Because of its route of infection it can be used as biological warfare agent. See also bioterrorism.Q-fever is category "B" agent. It is highly contagious and very stable in aerosols in a wide range of temperatures. Just 1-2 particles are enough to infect an individual. Q-fever microorganisms may survive on surfaces up to 60 days (like sporulating bacteria) and C. burnetii is known to reproduce and grow well in chicken egg embryos reaching very high concentrations. Protection against disease is offered by Q-Vax, a whole cell inactivated vaccine developed by a leading Australian vaccine manufacturing company CSL. (http://www.csl.com.au/QFever.asp)
History
It was first described by Edward Holbrook Derrick in abattoir workers in Brisbane, Queensland, Australia. The "Q" stands for “query” and was applied historically at a time when the causative agent was unknown.
In 1937 the bacterium was isolated by Frank Macfarlane Burnet and Mavis Freeman from one of Derrick’s patients for the first time and identified as Rickettsia-species. H.R. Cox and Davis isolated the pathogen from ticks in Montana, USA in 1938, called it Rickettsia diasporica, it was considered nonpathogenic until laboratory investigators were infected; it was officially named Coxiella burnetii the same year. It is a zoonotic disease and most common animal reservoirs are cattle, sheep and goats. Coxiella burnetii is no longer regarded as closely related to Rickettsiae.
Acknowledgements
List of contributors:
Pilar Almonacid
References
http://www.cdc.gov/ncidod/diseases/submenus/sub_q_fever.htm http://www.cdc.gov/ncidod/dvrd/qfever/index.htm http://www.cdc.gov/healthypets/diseases/qfever.htm
Template:Bacterial diseases
cs:Q-horečka
de:Q-Fieber
hr:Q groznica
it:Febbre Q
he:קדחת Q
nl:Q-koorts
fi:Q-kuume