Phenoxybenzamine
 | |
| Clinical data | |
|---|---|
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Pharmacokinetic data | |
| Elimination half-life | 24 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
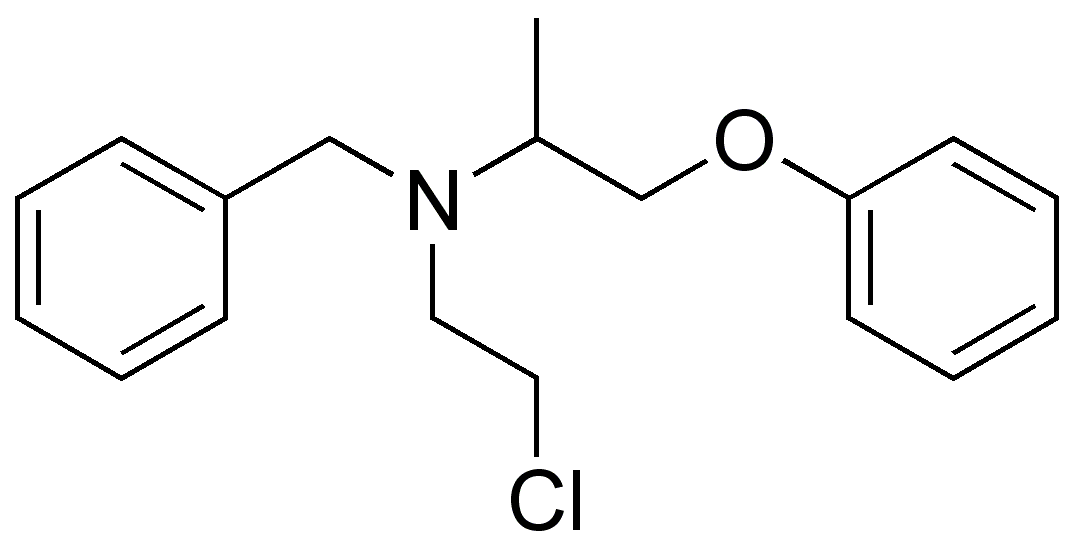
| Formula | C18H22ClNO |
| Molar mass | 303.826 g/mol |
Phenoxybenzamine (marketed under the trade name Dibenzyline) is a non-specific, irreversible alpha blocker used in the treatment of hypertension, and specifically that caused by pheochromocytoma. It has a slower onset and a longer lasting effect compared with other alpha blockers. It was also the first alpha blocker to be used for treatment of benign prostatic hyperplasia,[1] although it is currently seldom used for that indication due to an unfavourable side effects profile.
Pharmacology
Phenoxybenzamine is used as an anti-hypertensive due to its efficacy in reducing the vasoconstriction caused by adrenaline and noradrenaline. Phenoxybenzamine forms a permanent covalent bond with adrenergic receptors. Based on what we know about the structures of these receptors, it likely involves attack by the cysteine at position 3.36 in transmembrane helix 3 to form a stable linkage.[2] Thus, it remains permanently bound to the receptor, preventing adrenaline and noradrenaline from binding. This causes vasodilatation in blood vessels, due to its antagonistic effect at the alpha-1 adrenoceptor found in the walls of blood vessels, resulting in a drop in blood pressure.
It will also affect the alpha 1 and 2 receptors found in the nervous system, where this will reduce sympathetic activity. This is due to its effect on the postsynaptic membrane adrenoceptors in the second synapse of the sympathetic nervous pathway, and will therefore reduce the 'fight or flight' output of the sympathetic nervous system. This results in further general vasodilation, pupil constriction, an increase in GI tract motility and secretions, and glycogen synthesis.
It also has partial agonist/antagonist properties at the serotonin 5-HT2A receptor.
References
- ↑ Caine M, Perlberg S, Meretyk S (1978). "A placebo-controlled double-blind study of the effect of phenoxybenzamine in benign prostatic obstruction". British journal of urology. 50 (7): 551–4. PMID 88984.
- ↑ Frang H, Cockcroft V, Karskela T, Scheinin M, Marjamäki A (2001). "Phenoxybenzamine binding reveals the helical orientation of the third transmembrane domain of adrenergic receptors". J. Biol. Chem. 276 (33): 31279–84. doi:10.1074/jbc.M104167200. PMID 11395517.
- Day, M.D. (1979). Autonomic Pharmacology Experimental and Clinical Aspects. Churchill Livingstone.
- Bullock, S. Galbraith, A. Hunt, B. Manias, E. and Richards, A. (1999). Fundamentals of Pharmacology. Pearson Education Limited publishing as Prentice Hall.
- Pages with script errors
- CS1 maint: Multiple names: authors list
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs with no legal status
- Articles containing unverified chemical infoboxes
- Alpha blockers