Lincomycin Hydrochloride: Difference between revisions
(Created page with "{{DrugProjectFormSinglePage |authorTag={{SG}} |blackBoxWarningTitle=<b><span style="color:#FF0000;">TITLE</span></b> |blackBoxWarningBody=<i><span style="color:#FF0000;">Condi...") |
No edit summary |
||
| Line 1: | Line 1: | ||
{{DrugProjectFormSinglePage | {{DrugProjectFormSinglePage | ||
|authorTag={{SG}} | |authorTag={{SG}} | ||
|blackBoxWarningTitle=<b><span style="color:#FF0000;"> | |genericName=Lincomycin hydrochloride | ||
|blackBoxWarningBody=<i><span style="color:#FF0000;"> | |aOrAn=a | ||
|drugClass=[[Lincosamide]] | |||
|indicationType=treatment | |||
|indication=serious infections due to susceptible strains of [[streptococci]], [[pneumococci]], and [[staphylococci]] | |||
|hasBlackBoxWarning=Yes | |||
|adverseReactions=[[Diarrhea]], [[colitis]] and [[pruritus ani]] | |||
|blackBoxWarningTitle=<b><span style="color:#FF0000;">WARNING</span></b> | |||
|blackBoxWarningBody=<i><span style="color:#FF0000;"></span></i> Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including Lincomycin and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile. | |||
Because lincomycin therapy has been associated with severe colitis which may end fatally, it should be reserved for serious infections where less toxic antimicrobial agents are inappropriate. It should not be used in patients with nonbacterial infections such as most upper respiratory tract infections. | |||
C.diffficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents. | |||
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated. | |||
|offLabelAdultGuideSupport=There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of Lincomycin Hydrochloride in adult patients. | |offLabelAdultGuideSupport=There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of Lincomycin Hydrochloride in adult patients. | ||
|offLabelAdultNoGuideSupport=There is limited information regarding <i>Off-Label Non–Guideline-Supported Use</i> of Lincomycin Hydrochloride in adult patients. | |offLabelAdultNoGuideSupport=There is limited information regarding <i>Off-Label Non–Guideline-Supported Use</i> of Lincomycin Hydrochloride in adult patients. | ||
|offLabelPedGuideSupport=There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of Lincomycin Hydrochloride in pediatric patients. | |offLabelPedGuideSupport=There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of Lincomycin Hydrochloride in pediatric patients. | ||
|offLabelPedNoGuideSupport=There is limited information regarding <i>Off-Label Non–Guideline-Supported Use</i> of Lincomycin Hydrochloride in pediatric patients. | |offLabelPedNoGuideSupport=There is limited information regarding <i>Off-Label Non–Guideline-Supported Use</i> of Lincomycin Hydrochloride in pediatric patients. | ||
|contraindications=*This drug is contraindicated in patients previously found to be hypersensitive to lincomycin or [[clindamycin]]. | |||
|warnings=Clostridium difficile associated diarrhea | |||
Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including Lincomycin, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile. | |||
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents. | |||
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated. | |||
Hypersensitivity | |||
Serious hypersensitivity reactions, including anaphylaxis and erythema multiforme, have been reported with use of LINCOCIN. If an allergic reaction to LINCOCIN occurs, discontinue the drug. (See ADVERSE REACTIONS) | |||
Benzyl Alcohol Toxicity in Pediatric Patients (Gasping Syndrome") | |||
This product contains benzyl alcohol as a preservative. | |||
The preservative benzyl alcohol has been associated with serious adverse events, including the "gasping syndrome", and death in pediatric patients. Although normal therapeutic doses of this product ordinarily deliver amounts of benzyl alcohol that are substantially lower than those reported in association with the "gasping syndrome", the minimum amount of benzyl alcohol at which toxicity may occur is not known. The risk of benzyl alcohol toxicity depends on the quantity administered and the hepatic capacity to detoxify the chemical. Premature and low-birth weight infants may be more likely to develop toxicity. | |||
Use in Meningitis — Although lincomycin appears to diffuse into cerebrospinal fluid, levels of lincomycin in the CSF may be inadequate for the treatment of meningitis. | |||
|clinicalTrials=Gastrointestinal | |||
Glossitis, stomatitis, nausea, vomiting, antibacterial -associated diarrhea and colitis, and pruritus ani. Onset of pseudomembranous colitis symptoms may occur during or after antibacterial treatment (see WARNINGS). | |||
Hematopoietic | |||
Neutropenia, leukopenia, agranulocytosis and thrombocytopenic purpura have been reported. There have been rare reports of aplastic anemia and pancytopenia in which LINCOCIN could not be ruled out as the causative agent. | |||
Hypersensitivity Reactions | |||
Hypersensitivity reactions such as angioneurotic edema, serum sickness and anaphylaxis have been reported. Cases of erythema multiforme, some resembling Stevens-Johnson syndrome, have been associated with LINCOCIN (see WARNINGS). | |||
Skin and Mucous Membranes | |||
Skin rashes, urticaria and vaginitis and rare instances of exfoliative and vesiculobullous dermatitis have been reported. | |||
Liver | |||
Although no direct relationship of LINCOCIN to liver dysfunction has been established, jaundice and abnormal liver function tests (particularly elevations of serum transaminase) have been observed. | |||
Renal | |||
Although no direct relationship of lincomycin to renal damage has been established, renal dysfunction as evidenced by azotemia, oliguria, and/or proteinuria has been observed in rare instances. | |||
Cardiovascular | |||
After too rapid intravenous administration, rare instances of cardiopulmonary arrest and hypotension have been reported. (See DOSAGE AND ADMINISTRATION.) | |||
Special Senses | |||
Tinnitus and vertigo have been reported occasionally. | |||
Local Reactions | |||
Patients have demonstrated excellent local tolerance to intramuscularly administered LINCOCIN. Reports of pain following injection have been infrequent. Intravenous administration of LINCOCIN in 250 to 500 mL of 5% dextrose injection or 0.9% sodium chloride injection produced no local irritation or phlebitis. | |||
|drugInteractions=Lincomycin has been shown to have neuromuscular blocking properties that may enhance the action of other neuromuscular blocking agents. Therefore, it should be used in caution in patients receiving such agents. | |||
Antagonism between lincomycin and erythromycin in vitro has been demonstrated. Because of possible clinical significance, the two drugs should not be administered concurrently. | |||
|FDAPregCat=C | |||
|useInPregnancyFDA=LINCOCIN Sterile Solution contains benzyl alcohol as a preservative. Benzyl alcohol can cross the placenta. See WARNINGS. | |||
Teratogenic Effects | |||
There are no studies on the teratogenic potential of lincomycin in animals or adequate and well-controlled studies of pregnant women. | |||
Nonteratogenic Effects | |||
Reproduction studies have been performed in rats using oral doses of lincomycin up to 1000 mg/kg (1.2 times the maximum daily human dose based on mg/m2) and have revealed no adverse effects on survival of offspring from birth to weaning. | |||
|useInNursing=Lincomycin has been reported to appear in human milk in concentrations of 0.5 to 2.4 mcg/mL. Because of the potential for serious adverse reactions in nursing infants from LINCOCIN, a decision should be made whether to discontinue nursing, or to discontinue the drug, taking into account the importance of the drug to the mother. | |||
|useInPed=LINCOCIN Sterile Solution contains benzyl alcohol as a preservative. Benzyl alcohol has been associated with a fatal "Gasping Syndrome" in premature infants. See WARNINGS. Safety and effectiveness in pediatric patients below the age of one month have not been established. | |||
|useInGeri=Review of experience to date suggests that a subgroup of older patients with associated severe illness may tolerate diarrhea less well. When LINCOCIN is indicated in these patients, they should be carefully monitored for change in bowel frequency. | |||
|useInRenalImpair=The serum half-life of lincomycin may be prolonged in patients with severe impairment of renal function compared to patients with normal renal function. | |||
When therapy with LINCOCIN is required in individuals with severe impairment of renal function, an appropriate dose is 25 to 30% of that recommended for patients with normally functioning kidneys. | |||
|useInHepaticImpair=In patients with abnormal hepatic function, serum half-life may be twofold longer than in patients with normal hepatic function. | |||
|administration=*Intramuscular | |||
*Intravenous | |||
*Subconjuctival | |||
|monitoring=*During prolonged therapy with LINCOCIN, periodic liver and kidney function tests and blood counts should be performed. | |||
*Older patients with associated severe illness may tolerate diarrhea less well. When LINCOCIN is indicated in these patients, they should be carefully monitored for change in bowel frequency. | |||
*Patients with severe impairment of renal function and/or abnormal hepatic function should monitored during high-dose therapy. | |||
|IVCompat=Physically compatible for 24 hours at room temperature unless otherwise indicated. | |||
Infusion Solutions | |||
5% Dextrose Injection | |||
10% Dextrose Injection | |||
5% Dextrose and 0.9% Sodium Chloride Injection | |||
10% Dextrose and 0.9% Sodium Chloride Injection | |||
Ringer's Injection | |||
1/6 M Sodium Lactate Injection | |||
Travert 10%-Electrolyte No. 1 | |||
Dextran in Saline 6% w/v | |||
Vitamins in Infusion Solutions | |||
B-Complex | |||
B-Complex with Ascorbic Acid | |||
Antibacterial in Infusion Solutions | |||
Penicillin G Sodium (Satisfactory for 4 hours) | |||
Cephalothin | |||
Tetracycline HCl | |||
Cephaloridine | |||
Colistimethate (Satisfactory for 4 hours) | |||
Ampicillin | |||
Methicillin | |||
Chloramphenicol | |||
Polymyxin B Sulfate | |||
Physically Incompatible with: | |||
Novobiocin | |||
Kanamycin | |||
IT SHOULD BE EMPHASIZED THAT THE COMPATIBLE AND INCOMPATIBLE DETERMINATIONS ARE PHYSICAL OBSERVATIONS ONLY, NOT CHEMICAL DETERMINATIONS. ADEQUATE CLINICAL EVALUATION OF THE SAFETY AND EFFICACY OF THESE COMBINATIONS HAS NOT BEEN PERFORMED. | |||
|overdose=Serum levels of lincomycin are not appreciably affected by hemodialysis and peritoneal dialysis. | |||
|drugBox={{Drugbox | |||
| Verifiedfields = changed | |||
| Watchedfields = changed | |||
| verifiedrevid = 408578167 | |||
| IUPAC_name = (2''S'',4''R'')-''N''-[(1''R'',2''R'')-2-hydroxy-1-[(2''R'',3''R'',4''S'',5''R'',6''R'')-3,4,5-trihydroxy-6-(methylsulfanyl)oxan-2-yl]propyl]-1-methyl-4-propylpyrrolidine-2-carboxamide | |||
| image = Lincomycin.png | |||
<!--Clinical data--> | |||
| tradename = | |||
| Drugs.com = {{drugs.com|monograph|lincomycin-hydrochloride}} | |||
| MedlinePlus = a609005 | |||
| pregnancy_AU = <!-- A / B1 / B2 / B3 / C / D / X --> | |||
| pregnancy_US = C | |||
| pregnancy_category = | |||
| legal_AU = <!-- Unscheduled / S2 / S3 / S4 / S5 / S6 / S7 / S8 / S9 --> | |||
| legal_CA = <!-- / Schedule I, II, III, IV, V, VI, VII, VIII --> | |||
| legal_UK = <!-- GSL / P / POM / CD / Class A, B, C --> | |||
| legal_US = Rx-only | |||
| legal_status = | |||
| routes_of_administration = IM/IV | |||
<!--Pharmacokinetic data--> | |||
| bioavailability = N/A | |||
| protein_bound = | |||
| metabolism = | |||
| elimination_half-life = 5.4 ± 1.0 hours after IM or IV administration | |||
| excretion = renal and biliary | |||
<!--Identifiers--> | |||
| CASNo_Ref = {{cascite|correct|CAS}} | |||
| CAS_number_Ref = {{cascite|correct|??}} | |||
| CAS_number = 154-21-2 | |||
| ATC_prefix = J01 | |||
| ATC_suffix = FF02 | |||
| PubChem = 3000540 | |||
| DrugBank_Ref = {{drugbankcite|changed|drugbank}} | |||
| DrugBank = DB01627 | |||
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} | |||
| ChemSpiderID = 2272112 | |||
| UNII_Ref = {{fdacite|correct|FDA}} | |||
| UNII = BOD072YW0F | |||
| KEGG_Ref = {{keggcite|correct|kegg}} | |||
| KEGG = D00223 | |||
| ChEBI_Ref = {{ebicite|changed|EBI}} | |||
| ChEBI = 6472 | |||
| ChEMBL_Ref = {{ebicite|correct|EBI}} | |||
| ChEMBL = 1447 | |||
<!--Chemical data--> | |||
| C=18 | H=34 | N=2 | O=6 | S=1 | |||
| molecular_weight = 406.538 g/mol | |||
| smiles = O=C(N[C@@H]([C@H]1O[C@H](SC)[C@H](O)[C@@H](O)[C@H]1O)[C@H](O)C)[C@H]2N(C)C[C@H](CCC)C2 | |||
| InChI = 1/C18H34N2O6S/c1-5-6-10-7-11(20(3)8-10)17(25)19-12(9(2)21)16-14(23)13(22)15(24)18(26-16)27-4/h9-16,18,21-24H,5-8H2,1-4H3,(H,19,25)/t9-,10-,11+,12-,13+,14-,15-,16-,18-/m1/s1 | |||
| InChIKey = OJMMVQQUTAEWLP-KIDUDLJLBO | |||
| StdInChI_Ref = {{stdinchicite|correct|chemspider}} | |||
| StdInChI = 1S/C18H34N2O6S/c1-5-6-10-7-11(20(3)8-10)17(25)19-12(9(2)21)16-14(23)13(22)15(24)18(26-16)27-4/h9-16,18,21-24H,5-8H2,1-4H3,(H,19,25)/t9-,10-,11+,12-,13+,14-,15-,16-,18-/m1/s1 | |||
| StdInChIKey_Ref = {{stdinchicite|correct|chemspider}} | |||
| StdInChIKey = OJMMVQQUTAEWLP-KIDUDLJLSA-N | |||
}} | |||
|structure=LINCOCIN Sterile Solution contains lincomycin hydrochloride which is the monohydrated salt of lincomycin, a substance produced by the growth of a member of the lincolnensis group of Streptomyces lincolnensis (Fam. Streptomycetaceae). The chemical name for lincomycin hydrochloride is Methyl 6,8-dideoxy-6-(1-methyl-trans-4-propyl-L-2-pyrolidinecarboxamido)-1-thio-D-erythro-α-D-galacto-octopyranoside monohydrochloride monohydrate. The molecular formula of lincomycin hydrochloride is C18H34N2O6S.HCl.H2O and the molecular weight is 461.01. | |||
The structural formula is represented below: | |||
[[File:Lincomycin chemical structure.png|none|300px]] | |||
Lincomycin hydrochloride is a white or practically white, crystalline powder and is odorless or has a faint odor. Its solutions are acid and are dextrorotatory. Lincomycin hydrochloride is freely soluble in water; soluble in dimethylformamide and very slightly soluble in acetone. | |||
|PK=Intramuscular administration of a single dose of 600 mg of lincomycin produces average peak serum levels of 11.6 µg/mL at 60 minutes and maintains therapeutic levels for 17 to 20 hours for most susceptible gram-positive organisms. Urinary excretion after this dose ranges from 1.8 to 24.8 percent (mean: 17.3 percent). | |||
A two hour intravenous infusion of 600 mg of lincomycin achieves average peak serum levels of 15.9 µg/mL and yields therapeutic levels for 14 hours for most susceptible gram-positive organisms. Urinary excretion ranges from 4.9 to 30.3 percent (mean: 13.8 percent). | |||
The biological half-life after intramuscular or intravenous administration is 5.4 ± 1.0 hours. The serum half-life of lincomycin may be prolonged in patients with severe impairment of renal function compared to patients with normal renal function. In patients with abnormal hepatic function, serum half-life may be twofold longer than in patients with normal hepatic function. Hemodialysis and peritoneal dialysis are not effective in removing lincomycin from the serum. | |||
Tissue level studies indicate that bile is an important route of excretion. Significant levels have been demonstrated in the majority of body tissues. Although lincomycin appears to diffuse into cerebrospinal fluid (CSF), levels of lincomycin in the CSF appear inadequate for the treatment of meningitis. | |||
|nonClinToxic=Carcinogenesis, Mutagenesis, Impairment of Fertility | |||
The carcinogenic potential of lincomycin has not been evaluated. | |||
Lincomycin was not found to be mutagenic in the Ames Salmonella reversion assay or the V79 Chinese hamster lung cells at the HGPRT locus. It did not induce DNA strand breaks in V79 Chinese hamster lung cells as measured by alkaline elution or chromosomal abnormalities in cultured human lymphocytes. In vivo, lincomycin was negative in both the rat and mouse micronucleus assays and it did not induce sex-linked recessive lethal mutations in the offspring of male Drosophila. However, lincomycin did cause unscheduled DNA syntheses in freshly isolated rat hepatocytes. | |||
Impairment of fertility was not observed in male or female rats given oral 300 mg/kg doses of lincomycin (0.36 times the highest recommended human dose based on mg/m2). | |||
|clinicalStudies=*Experience with 345 obstetrical patients receiving this drug revealed no ill effects related to pregnancy. | |||
|howSupplied=LINCOCIN Sterile Solution is available in the following strength and package sizes: | |||
300 mg | |||
2 mL Vials — NDC 0009-0555-01 | |||
10 mL Vials — NDC 0009-0555-02 | |||
Each mL of LINCOCIN Sterile Solution contains lincomycin hydrochloride equivalent to lincomycin 300 mg; also benzyl alcohol, 9.45 mg added as preservative. | |||
|storage=*Store at controlled room temperature 20° to 25°C (68° to 77°F) | |||
|packLabel=[[File:Lincomycin FDA label.png|none|450px]] | |||
|fdaPatientInfo=Patients should be counseled that antibacterial drugs including LINCOCIN should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When LINCOCIN is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by LINCOCIN or other antibacterial drugs in the future. | |||
Diarrhea is a common problem caused by antibacterial which usually ends when the antibacterial is discontinued. Sometimes after starting treatment with antibacterial, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibacterial. If this occurs, patients should contact their physician as soon as possible | |||
|alcohol=Alcohol-Lincomycin Hydrochloride interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication. | |alcohol=Alcohol-Lincomycin Hydrochloride interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication. | ||
|brandNames=*Lincocin | |||
}} | |||
{{LabelImage | |||
|fileName=Lincomycin Package.png | |||
}} | |||
{{LabelImage | |||
|fileName=Lincomycin pakcage 2.png | |||
}} | }} | ||
Revision as of 16:34, 23 January 2015
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Stefano Giannoni [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
WARNING
See full prescribing information for complete Boxed Warning.
Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including Lincomycin and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
Because lincomycin therapy has been associated with severe colitis which may end fatally, it should be reserved for serious infections where less toxic antimicrobial agents are inappropriate. It should not be used in patients with nonbacterial infections such as most upper respiratory tract infections. C.diffficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents. If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated. |
Overview
Lincomycin Hydrochloride is a Lincosamide that is FDA approved for the treatment of serious infections due to susceptible strains of streptococci, pneumococci, and staphylococci. There is a Black Box Warning for this drug as shown here. Common adverse reactions include Diarrhea, colitis and pruritus ani.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
There is limited information regarding Lincomycin Hydrochloride FDA-Labeled Indications and Dosage (Adult) in the drug label.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Lincomycin Hydrochloride in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Lincomycin Hydrochloride in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding Lincomycin Hydrochloride FDA-Labeled Indications and Dosage (Pediatric) in the drug label.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Lincomycin Hydrochloride in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Lincomycin Hydrochloride in pediatric patients.
Contraindications
- This drug is contraindicated in patients previously found to be hypersensitive to lincomycin or clindamycin.
Warnings
|
WARNING
See full prescribing information for complete Boxed Warning.
Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including Lincomycin and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
Because lincomycin therapy has been associated with severe colitis which may end fatally, it should be reserved for serious infections where less toxic antimicrobial agents are inappropriate. It should not be used in patients with nonbacterial infections such as most upper respiratory tract infections. C.diffficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents. If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated. |
Clostridium difficile associated diarrhea Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including Lincomycin, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
Hypersensitivity Serious hypersensitivity reactions, including anaphylaxis and erythema multiforme, have been reported with use of LINCOCIN. If an allergic reaction to LINCOCIN occurs, discontinue the drug. (See ADVERSE REACTIONS)
Benzyl Alcohol Toxicity in Pediatric Patients (Gasping Syndrome") This product contains benzyl alcohol as a preservative.
The preservative benzyl alcohol has been associated with serious adverse events, including the "gasping syndrome", and death in pediatric patients. Although normal therapeutic doses of this product ordinarily deliver amounts of benzyl alcohol that are substantially lower than those reported in association with the "gasping syndrome", the minimum amount of benzyl alcohol at which toxicity may occur is not known. The risk of benzyl alcohol toxicity depends on the quantity administered and the hepatic capacity to detoxify the chemical. Premature and low-birth weight infants may be more likely to develop toxicity.
Use in Meningitis — Although lincomycin appears to diffuse into cerebrospinal fluid, levels of lincomycin in the CSF may be inadequate for the treatment of meningitis.
Adverse Reactions
Clinical Trials Experience
Gastrointestinal Glossitis, stomatitis, nausea, vomiting, antibacterial -associated diarrhea and colitis, and pruritus ani. Onset of pseudomembranous colitis symptoms may occur during or after antibacterial treatment (see WARNINGS).
Hematopoietic Neutropenia, leukopenia, agranulocytosis and thrombocytopenic purpura have been reported. There have been rare reports of aplastic anemia and pancytopenia in which LINCOCIN could not be ruled out as the causative agent.
Hypersensitivity Reactions Hypersensitivity reactions such as angioneurotic edema, serum sickness and anaphylaxis have been reported. Cases of erythema multiforme, some resembling Stevens-Johnson syndrome, have been associated with LINCOCIN (see WARNINGS).
Skin and Mucous Membranes Skin rashes, urticaria and vaginitis and rare instances of exfoliative and vesiculobullous dermatitis have been reported.
Liver Although no direct relationship of LINCOCIN to liver dysfunction has been established, jaundice and abnormal liver function tests (particularly elevations of serum transaminase) have been observed.
Renal Although no direct relationship of lincomycin to renal damage has been established, renal dysfunction as evidenced by azotemia, oliguria, and/or proteinuria has been observed in rare instances.
Cardiovascular After too rapid intravenous administration, rare instances of cardiopulmonary arrest and hypotension have been reported. (See DOSAGE AND ADMINISTRATION.)
Special Senses Tinnitus and vertigo have been reported occasionally.
Local Reactions Patients have demonstrated excellent local tolerance to intramuscularly administered LINCOCIN. Reports of pain following injection have been infrequent. Intravenous administration of LINCOCIN in 250 to 500 mL of 5% dextrose injection or 0.9% sodium chloride injection produced no local irritation or phlebitis.
Postmarketing Experience
There is limited information regarding Lincomycin Hydrochloride Postmarketing Experience in the drug label.
Drug Interactions
Lincomycin has been shown to have neuromuscular blocking properties that may enhance the action of other neuromuscular blocking agents. Therefore, it should be used in caution in patients receiving such agents.
Antagonism between lincomycin and erythromycin in vitro has been demonstrated. Because of possible clinical significance, the two drugs should not be administered concurrently.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): C LINCOCIN Sterile Solution contains benzyl alcohol as a preservative. Benzyl alcohol can cross the placenta. See WARNINGS.
Teratogenic Effects
There are no studies on the teratogenic potential of lincomycin in animals or adequate and well-controlled studies of pregnant women.
Nonteratogenic Effects
Reproduction studies have been performed in rats using oral doses of lincomycin up to 1000 mg/kg (1.2 times the maximum daily human dose based on mg/m2) and have revealed no adverse effects on survival of offspring from birth to weaning.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Lincomycin Hydrochloride in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Lincomycin Hydrochloride during labor and delivery.
Nursing Mothers
Lincomycin has been reported to appear in human milk in concentrations of 0.5 to 2.4 mcg/mL. Because of the potential for serious adverse reactions in nursing infants from LINCOCIN, a decision should be made whether to discontinue nursing, or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
LINCOCIN Sterile Solution contains benzyl alcohol as a preservative. Benzyl alcohol has been associated with a fatal "Gasping Syndrome" in premature infants. See WARNINGS. Safety and effectiveness in pediatric patients below the age of one month have not been established.
Geriatic Use
Review of experience to date suggests that a subgroup of older patients with associated severe illness may tolerate diarrhea less well. When LINCOCIN is indicated in these patients, they should be carefully monitored for change in bowel frequency.
Gender
There is no FDA guidance on the use of Lincomycin Hydrochloride with respect to specific gender populations.
Race
There is no FDA guidance on the use of Lincomycin Hydrochloride with respect to specific racial populations.
Renal Impairment
The serum half-life of lincomycin may be prolonged in patients with severe impairment of renal function compared to patients with normal renal function. When therapy with LINCOCIN is required in individuals with severe impairment of renal function, an appropriate dose is 25 to 30% of that recommended for patients with normally functioning kidneys.
Hepatic Impairment
In patients with abnormal hepatic function, serum half-life may be twofold longer than in patients with normal hepatic function.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Lincomycin Hydrochloride in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Lincomycin Hydrochloride in patients who are immunocompromised.
Administration and Monitoring
Administration
- Intramuscular
- Intravenous
- Subconjuctival
Monitoring
- During prolonged therapy with LINCOCIN, periodic liver and kidney function tests and blood counts should be performed.
- Older patients with associated severe illness may tolerate diarrhea less well. When LINCOCIN is indicated in these patients, they should be carefully monitored for change in bowel frequency.
- Patients with severe impairment of renal function and/or abnormal hepatic function should monitored during high-dose therapy.
IV Compatibility
Physically compatible for 24 hours at room temperature unless otherwise indicated.
Infusion Solutions 5% Dextrose Injection 10% Dextrose Injection 5% Dextrose and 0.9% Sodium Chloride Injection 10% Dextrose and 0.9% Sodium Chloride Injection Ringer's Injection
1/6 M Sodium Lactate Injection Travert 10%-Electrolyte No. 1 Dextran in Saline 6% w/v
Vitamins in Infusion Solutions B-Complex B-Complex with Ascorbic Acid
Antibacterial in Infusion Solutions Penicillin G Sodium (Satisfactory for 4 hours) Cephalothin Tetracycline HCl Cephaloridine Colistimethate (Satisfactory for 4 hours) Ampicillin Methicillin Chloramphenicol Polymyxin B Sulfate
Physically Incompatible with: Novobiocin Kanamycin
IT SHOULD BE EMPHASIZED THAT THE COMPATIBLE AND INCOMPATIBLE DETERMINATIONS ARE PHYSICAL OBSERVATIONS ONLY, NOT CHEMICAL DETERMINATIONS. ADEQUATE CLINICAL EVALUATION OF THE SAFETY AND EFFICACY OF THESE COMBINATIONS HAS NOT BEEN PERFORMED.
Overdosage
Serum levels of lincomycin are not appreciably affected by hemodialysis and peritoneal dialysis.
Pharmacology
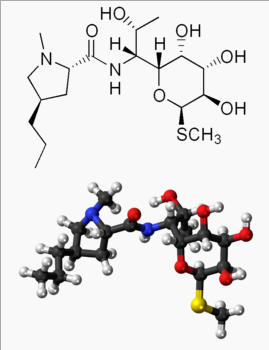 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a609005 |
| Pregnancy category |
|
| Routes of administration | IM/IV |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | N/A |
| Elimination half-life | 5.4 ± 1.0 hours after IM or IV administration |
| Excretion | renal and biliary |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C18H34N2O6S |
| Molar mass | 406.538 g/mol |
| 3D model (JSmol) | |
| |
| |
| | |
Mechanism of Action
There is limited information regarding Lincomycin Hydrochloride Mechanism of Action in the drug label.
Structure
LINCOCIN Sterile Solution contains lincomycin hydrochloride which is the monohydrated salt of lincomycin, a substance produced by the growth of a member of the lincolnensis group of Streptomyces lincolnensis (Fam. Streptomycetaceae). The chemical name for lincomycin hydrochloride is Methyl 6,8-dideoxy-6-(1-methyl-trans-4-propyl-L-2-pyrolidinecarboxamido)-1-thio-D-erythro-α-D-galacto-octopyranoside monohydrochloride monohydrate. The molecular formula of lincomycin hydrochloride is C18H34N2O6S.HCl.H2O and the molecular weight is 461.01.
The structural formula is represented below:
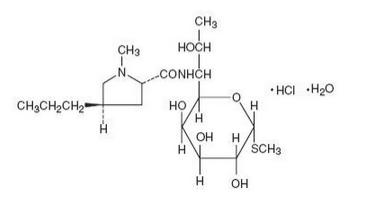
Lincomycin hydrochloride is a white or practically white, crystalline powder and is odorless or has a faint odor. Its solutions are acid and are dextrorotatory. Lincomycin hydrochloride is freely soluble in water; soluble in dimethylformamide and very slightly soluble in acetone.
Pharmacodynamics
There is limited information regarding Lincomycin Hydrochloride Pharmacodynamics in the drug label.
Pharmacokinetics
Intramuscular administration of a single dose of 600 mg of lincomycin produces average peak serum levels of 11.6 µg/mL at 60 minutes and maintains therapeutic levels for 17 to 20 hours for most susceptible gram-positive organisms. Urinary excretion after this dose ranges from 1.8 to 24.8 percent (mean: 17.3 percent).
A two hour intravenous infusion of 600 mg of lincomycin achieves average peak serum levels of 15.9 µg/mL and yields therapeutic levels for 14 hours for most susceptible gram-positive organisms. Urinary excretion ranges from 4.9 to 30.3 percent (mean: 13.8 percent).
The biological half-life after intramuscular or intravenous administration is 5.4 ± 1.0 hours. The serum half-life of lincomycin may be prolonged in patients with severe impairment of renal function compared to patients with normal renal function. In patients with abnormal hepatic function, serum half-life may be twofold longer than in patients with normal hepatic function. Hemodialysis and peritoneal dialysis are not effective in removing lincomycin from the serum.
Tissue level studies indicate that bile is an important route of excretion. Significant levels have been demonstrated in the majority of body tissues. Although lincomycin appears to diffuse into cerebrospinal fluid (CSF), levels of lincomycin in the CSF appear inadequate for the treatment of meningitis.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility The carcinogenic potential of lincomycin has not been evaluated.
Lincomycin was not found to be mutagenic in the Ames Salmonella reversion assay or the V79 Chinese hamster lung cells at the HGPRT locus. It did not induce DNA strand breaks in V79 Chinese hamster lung cells as measured by alkaline elution or chromosomal abnormalities in cultured human lymphocytes. In vivo, lincomycin was negative in both the rat and mouse micronucleus assays and it did not induce sex-linked recessive lethal mutations in the offspring of male Drosophila. However, lincomycin did cause unscheduled DNA syntheses in freshly isolated rat hepatocytes.
Impairment of fertility was not observed in male or female rats given oral 300 mg/kg doses of lincomycin (0.36 times the highest recommended human dose based on mg/m2).
Clinical Studies
- Experience with 345 obstetrical patients receiving this drug revealed no ill effects related to pregnancy.
How Supplied
LINCOCIN Sterile Solution is available in the following strength and package sizes:
300 mg
2 mL Vials — NDC 0009-0555-01 10 mL Vials — NDC 0009-0555-02
Each mL of LINCOCIN Sterile Solution contains lincomycin hydrochloride equivalent to lincomycin 300 mg; also benzyl alcohol, 9.45 mg added as preservative.
Storage
- Store at controlled room temperature 20° to 25°C (68° to 77°F)
Images
Drug Images
{{#ask: Page Name::Lincomycin Hydrochloride |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
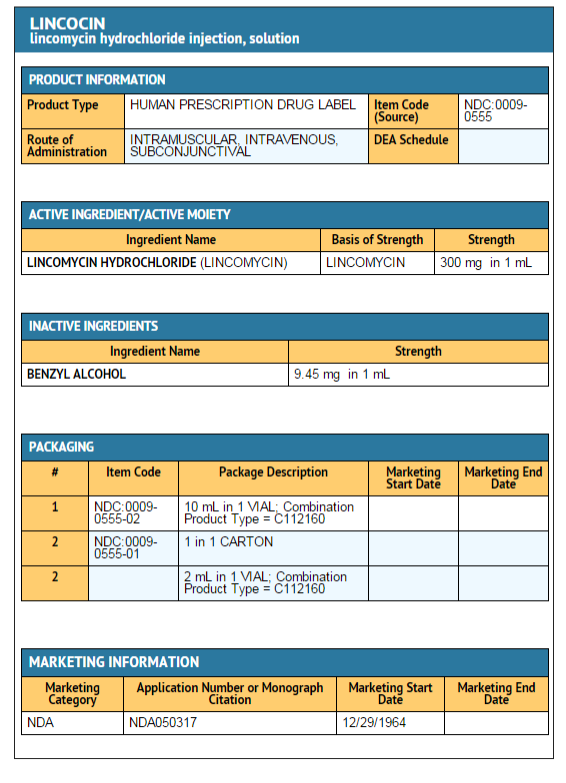
{{#ask: Label Page::Lincomycin Hydrochloride |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Patients should be counseled that antibacterial drugs including LINCOCIN should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When LINCOCIN is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by LINCOCIN or other antibacterial drugs in the future.
Diarrhea is a common problem caused by antibacterial which usually ends when the antibacterial is discontinued. Sometimes after starting treatment with antibacterial, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibacterial. If this occurs, patients should contact their physician as soon as possible
Precautions with Alcohol
Alcohol-Lincomycin Hydrochloride interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Lincocin
Look-Alike Drug Names
There is limited information regarding Lincomycin Hydrochloride Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Label Page=Lincomycin Hydrochloride |Label Name=Lincomycin Package.png
}}
{{#subobject:
|Label Page=Lincomycin Hydrochloride |Label Name=Lincomycin pakcage 2.png
}}
- Pages with script errors
- Template:drugs.com link with non-standard subpage
- Articles with changed DrugBank identifier
- Articles with changed EBI identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields