Levocabastine
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Rabin Bista, M.B.B.S. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Levocabastine is a histamine H1-receptor antagonist that is FDA approved for the treatment of seasonal allergic conjunctivitis.. Common adverse reactions include stinging and burning sensation in the eye and headache.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications=
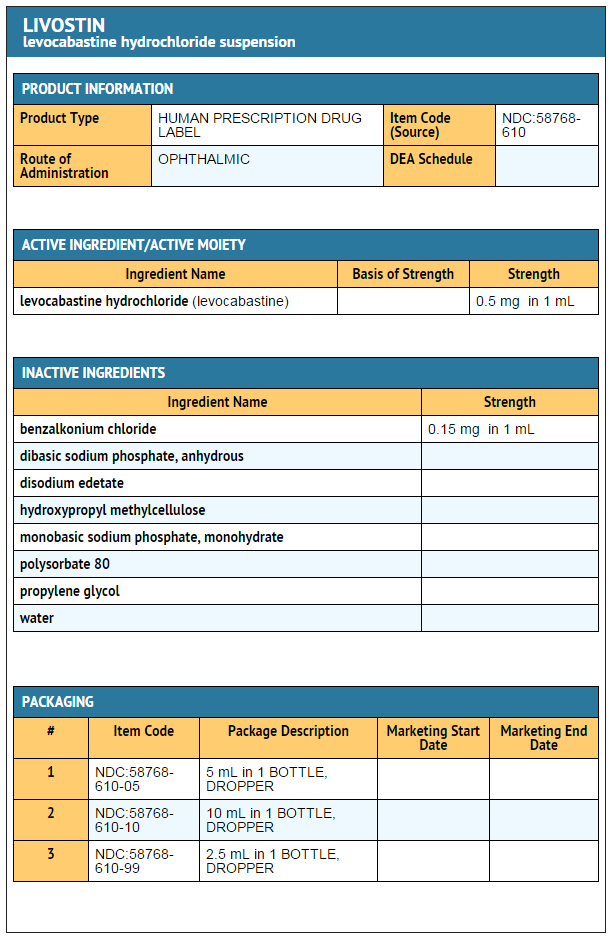
- LIVOSTIN™ 0.05% (levocabastine hydrochloride ophthalmic suspension) is indicated for the temporary relief of the signs and symptoms of seasonal allergic conjunctivitis.
=Dosage
- SHAKE WELL BEFORE USING. The usual dose is one drop instilled in affected eyes four times per day.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Levocabastine in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Levocabastine in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Levocabastine in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Levocabastine in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Levocabastine in pediatric patients.
Contraindications
- This product is contraindicated in persons with known or suspected hypersensitivity to any of its components. It should not be used while soft contact lenses are being worn.
Warnings
- For topical use only. Not for injection.
Adverse Reactions
Clinical Trials Experience
- The most frequent adverse experiences reported with the use of LIVOSTIN™ 0.05% (levocabastine hydrochloride ophthalmic suspension) were mild, transient stinging and burning (29%) and headache (5%).
- Other adverse experiences reported in approximately 1-3% of patients treated with LIVOSTIN™ were visual disturbances, dry mouth, fatigue, pharyngitis, eye pain/dryness, somnolence, red eyes, lacrimation/discharge, cough, nausea, rash/erythema, eyelid edema, and dyspnea.
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Levocabastine in the drug label.
Drug Interactions
There is limited information regarding Levocabastine Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
- Levocabastine has been shown to be teratogenic (polydactyly) in rats when given in doses 16,500 times the maximum recommended human ocular dose. Teratogenicity (polydactyly, hydrocephaly, brachygnathia), embryotoxicity, and maternal toxicity were observed in rats at 66,000 times the maximum recommended ocular human dose. There are no adequate and well-controlled studies in pregnant women. Levocabastine should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Levocabastine in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Levocabastine during labor and delivery.
Nursing Mothers
- Based on determinations of levocabastine in breast milk after ophthalmic administration of the drug to one nursing woman, it was calculated that the daily dose of levocabastine in the infant was about 0.5 µg.
Pediatric Use
- Safety and effectiveness in pediatric patients below the age of 12 have not been established.
Geriatic Use
There is no FDA guidance on the use of Levocabastine with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Levocabastine with respect to specific gender populations.
Race
There is no FDA guidance on the use of Levocabastine with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Levocabastine in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Levocabastine in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Levocabastine in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Levocabastine in patients who are immunocompromised.
Administration and Monitoring
Administration
- Topical ophthalmic application
Monitoring
There is limited information regarding Monitoring of Levocabastine in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Levocabastine in the drug label.
Overdosage
There is limited information regarding Overdose of Levocabastine in the drug label.
Pharmacology
| |
| |
Levocabastine
| |
| Systematic (IUPAC) name | |
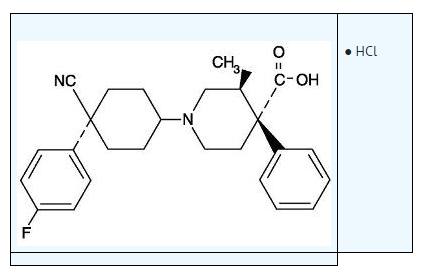
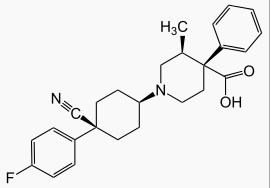
| (3S,4R)-1-[4-cyano-4-(4-fluorophenyl)cyclohexyl]-3-methyl-4-phenylpiperidine-4-carboxylic acid | |
| Identifiers | |
| CAS number | |
| ATC code | R01 S01GX02 (WHO) |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 420.519 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | 3 |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. | |
| Legal status |
Template:Unicode Prescription only |
| Routes | Ophthalmic, intranasal[1] |
Mechanism of Action
- Levocabastine is a potent, selective histamine H1-antagonist.
Structure
- LIVOSTIN™ 0.05% (levocabastine hydrochloride ophthalmic suspension) is a selective histamine H1-receptor antagonist for topical ophthalmic use. Each mL contains 0.54 mg levocabastine hydrochloride equivalent to 0.5 mg levocabastine; 0.15 mg benzalkonium chloride; propylene glycol; polysorbate 80; dibasic sodium phosphate, anhydrous; monobasic sodium phosphate, monohydrate; disodium edetate; hydroxypropyl methylcellulose; and purified water. It has a pH of 6.0 to 8.0.
- The chemical name for levocabastine hydrochloride is (–)-trans-1-[cis-4-Cyano-4- (p-fluorophenyl)cyclohexyl]-3-methyl-4-phenylisonipecotic acid monohydrochloride, and is represented by the following chemical structure:
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Levocabastine in the drug label.
Pharmacokinetics
- Antigen challenge studies performed two and four hours after initial drug instillation indicated activity was maintained for at least two hours.
- In an environmental study, LIVOSTIN™ 0.05% (levocabastine hydrochloride ophthalmic suspension) instilled four times daily was shown to be significantly more effective than its vehicle in reducing ocular itching associated with seasonal allergic conjunctivitis.
- After instillation in the eye, levocabastine is systemically absorbed. However, the amount of systemically absorbed levocabastine after therapeutic ocular doses is low (mean plasma concentrations in the range of 1-2 ng/mL).
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
- Levocabastine was not carcinogenic in male or female rats or in male mice when administered in the diet for up to 24 months. In female mice, levocabastine doses of 5,000 and 21,500 times the maximum recommended ocular human use level resulted in an increased incidence of pituitary gland adenoma and mammary gland adenocarcinoma possibly produced by increased prolactin levels. The clinical relevance of this finding is unknown with regard to the interspecies differences in prolactin physiology and the very low plasma concentrations of levocabastine following ocular administration.
- Mutagenic potential was not demonstrated for levocabastine when tested in Ames’ Salmonella reversion test or in Escherichia coli, Drosophila melanogaster, a mouse Dominant Lethal Assay or in rat Micronucleus test.
- In reproduction studies in rats, levocabastine showed no effects on fertility at oral doses of 20 mg/kg/day (8,300 times the maximum recommended human ocular dose).
Clinical Studies
There is limited information regarding Clinical Studies of Levocabastine in the drug label.
How Supplied
- LIVOSTIN™ 0.05% (levocabastine hydrochloride ophthalmic suspension), 2.5 mL, 5 mL, and 10 mL is provided in white, polyethylene dropper tip squeeze bottles.
- Keep tightly closed when not in use.
- Do not use if the suspension has discolored.
- NDC 58768-610-10 (10.0 mL)
- NDC 58768-610-05 (5.0 mL)
- NDC 58768-610-99 (2.5 mL)
- Rx Only
Storage
- Store at controlled room temperature 15°to 30°C (59° to 86°F).
- Protect from freezing.
Images
Drug Images
{{#ask: Page Name::Levocabastine |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
Ingredients and Appearance
{{#ask: Label Page::Levocabastine |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- SHAKE WELL BEFORE USING. To prevent contaminating the dropper tip and suspension, care should be taken not to touch the eyelids or surrounding areas with the dropper tip of the bottle. Keep bottle tightly closed when not in use. Do not use if the suspension has discolored. Store at controlled room temperature. Protect from freezing.
Precautions with Alcohol
- Alcohol-Levocabastine interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- LIVOSTIN®[2]
Look-Alike Drug Names
There is limited information regarding Levocabastine Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "RxMed: Pharmaceutical Information - LIVOSTIN NASAL SPRAY". Retrieved 13 November 2005.
- ↑ "LEVOCABASTINE".