Isoxsuprine: Difference between revisions
No edit summary |
No edit summary |
||
| (19 intermediate revisions by 5 users not shown) | |||
| Line 1: | Line 1: | ||
{{DrugProjectFormSinglePage | {{DrugProjectFormSinglePage | ||
|authorTag= | |authorTag={{SS}}; {{TA}} | ||
|genericName=Isoxsuprine | |||
|aOrAn=a | |||
|drugClass=[[vasodilator]] | |||
|indicationType=treatment | |||
|indication=cerebral [[vascular insufficiency]], [[arteriosclerosis obliterans]], [[thromboangitis obliterans]], and [[Raynaud's disease]] | |||
|genericName= | |adverseReactions=[[hypotension]], [[tachyarrhythmia]], [[rash]], [[abdominal discomfort]], [[nausea]], and [[dizziness]] | ||
|blackBoxWarningTitle=Title | |||
Isoxsuprine | |blackBoxWarningBody=<i><span style="color:#FF0000;">ConditionName: </span></i> | ||
|aOrAn= | |||
a | |||
|drugClass= | |||
[[vasodilator]] | |||
|indication= | |||
cerebral [[vascular insufficiency]], [[arteriosclerosis obliterans]], [[thromboangitis obliterans]], and [[Raynaud's disease]] | |||
|adverseReactions= | |||
|blackBoxWarningTitle= | |||
Title | |||
|blackBoxWarningBody= | |||
<i><span style="color:#FF0000;">ConditionName: </span></i> | |||
* Content | * Content | ||
| Line 41: | Line 15: | ||
<!--FDA-Labeled Indications and Dosage (Adult)--> | <!--FDA-Labeled Indications and Dosage (Adult)--> | ||
|fdaLIADAdult======Cerebral Vascular Insufficiency, Arteriosclerosis Obliterans, Thromboangitis Obliterans, and Raynaud's Disease===== | |||
|fdaLIADAdult= | |||
===== | |||
* Dosing Information | * Dosing Information | ||
:* | :* 10 to 20 mg three or four times daily | ||
<!--Off-Label Use and Dosage (Adult)--> | <!--Off-Label Use and Dosage (Adult)--> | ||
<!--Guideline-Supported Use (Adult)--> | <!--Guideline-Supported Use (Adult)--> | ||
|offLabelAdultGuideSupport=There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of {{PAGENAME}} in adult patients. | |||
|offLabelAdultGuideSupport= | |||
There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of {{PAGENAME}} in adult patients. | |||
<!--Non–Guideline-Supported Use (Adult)--> | <!--Non–Guideline-Supported Use (Adult)--> | ||
|offLabelAdultNoGuideSupport=There is limited information regarding <i>Off-Label Non–Guideline-Supported Use</i> of {{PAGENAME}} in adult patients. | |||
|offLabelAdultNoGuideSupport= | |||
There is limited information regarding <i>Off-Label Non–Guideline-Supported Use</i> of {{PAGENAME}} in adult patients. | |||
<!--Pediatric Indications and Dosage--> | <!--Pediatric Indications and Dosage--> | ||
<!--FDA-Labeled Indications and Dosage (Pediatric)--> | <!--FDA-Labeled Indications and Dosage (Pediatric)--> | ||
|fdaLIADPed=There is limited information regarding <i>FDA-Labeled Use</i> of {{PAGENAME}} in pediatric patients. | |||
|fdaLIADPed= | |||
There is limited information regarding <i>FDA-Labeled Use</i> of {{PAGENAME}} in pediatric patients. | |||
<!--Off-Label Use and Dosage (Pediatric)--> | <!--Off-Label Use and Dosage (Pediatric)--> | ||
<!--Guideline-Supported Use (Pediatric)--> | <!--Guideline-Supported Use (Pediatric)--> | ||
|offLabelPedGuideSupport=There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of {{PAGENAME}} in pediatric patients. | |||
|offLabelPedGuideSupport= | |||
There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of {{PAGENAME}} in pediatric patients. | |||
<!--Non–Guideline-Supported Use (Pediatric)--> | <!--Non–Guideline-Supported Use (Pediatric)--> | ||
|offLabelPedNoGuideSupport=There is limited information regarding <i>Off-Label Non–Guideline-Supported Use</i> of {{PAGENAME}} in pediatric patients. | |||
|offLabelPedNoGuideSupport= | |||
There is limited information regarding <i>Off-Label Non–Guideline-Supported Use</i> of {{PAGENAME}} in pediatric patients. | |||
<!--Contraindications--> | <!--Contraindications--> | ||
|contraindications=* There are no known contraindications to oral use when administered in recommended doses. | |||
|contraindications= | |||
* | |||
<!--Warnings--> | <!--Warnings--> | ||
|warnings=* Should not be given immediately postpartum or in the presence of arterial [[bleeding]]. | |||
|warnings= | |||
* | |||
<!--Adverse Reactions--> | <!--Adverse Reactions--> | ||
<!--Clinical Trials Experience--> | <!--Clinical Trials Experience--> | ||
|clinicalTrials=There is limited information regarding <i>Clinical Trial Experience</i> of {{PAGENAME}} in the drug label. | |||
|clinicalTrials= | |||
There is limited information regarding <i>Clinical Trial Experience</i> of {{PAGENAME}} in the drug label. | |||
<!--Postmarketing Experience--> | <!--Postmarketing Experience--> | ||
|postmarketing=* On rare occasions oral administration of the drug has been associated in time with the occurrences of [[hypotension]], [[tachycardia]], [[nausea]], [[vomiting]], [[dizziness]], abdominal distress, and severe [[rash]]. If rash appears the drug should be discontinued. | |||
|postmarketing= | * Although available evidence suggests a temporal association of these reactions with Isoxsuprine, a casual relationship can be neither confirmed nor refused. | ||
* Maternal and fetal [[tachycardia]] may occur under such use. [[Hypocalcemia]], [[hypoglycemia]], [[hypotension]] and [[ileus]] have been reported to occur in infants whose mothers received Isoxsuprine Hydrochloride. [[Pulmonary edema]] has been reported in mothers treated with beta stimulants. Isoxsuprine Hydrochloride is neither approved nor recommended for use in the treatment of premature labor. | |||
<!--Drug Interactions--> | <!--Drug Interactions--> | ||
|drugInteractions=There is limited information regarding <i>Drug Interactions</i> of {{PAGENAME}} in the drug label. | |||
|drugInteractions= | |||
<!--Use in Specific Populations--> | <!--Use in Specific Populations--> | ||
|useInPregnancyFDA=There is no FDA guidance on the use of {{PAGENAME}} with respect to pregnant patients. | |||
|useInPregnancyFDA= | |useInPregnancyAUS=* '''Australian Drug Evaluation Committee (ADEC) Pregnancy Category''' | ||
|useInPregnancyAUS= | |||
* '''Australian Drug Evaluation Committee (ADEC) Pregnancy Category''' | |||
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of {{PAGENAME}} in women who are pregnant. | There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of {{PAGENAME}} in women who are pregnant. | ||
|useInLaborDelivery=There is no FDA guidance on use of {{PAGENAME}} during labor and delivery. | |||
|useInLaborDelivery= | |useInNursing=There is no FDA guidance on the use of {{PAGENAME}} with respect to nursing mothers. | ||
There is no FDA guidance on use of {{PAGENAME}} during labor and delivery. | |useInPed=There is no FDA guidance on the use of {{PAGENAME}} with respect to pediatric patients. | ||
|useInGeri=There is no FDA guidance on the use of {{PAGENAME}} with respect to geriatric patients. | |||
|useInNursing= | |useInGender=There is no FDA guidance on the use of {{PAGENAME}} with respect to specific gender populations. | ||
There is no FDA guidance on the use of {{PAGENAME}} with respect to nursing mothers. | |useInRace=There is no FDA guidance on the use of {{PAGENAME}} with respect to specific racial populations. | ||
|useInRenalImpair=There is no FDA guidance on the use of {{PAGENAME}} in patients with renal impairment. | |||
|useInPed= | |useInHepaticImpair=There is no FDA guidance on the use of {{PAGENAME}} in patients with hepatic impairment. | ||
There is no FDA guidance on the use of {{PAGENAME}} with respect to pediatric patients. | |useInReproPotential=There is no FDA guidance on the use of {{PAGENAME}} in women of reproductive potentials and males. | ||
|useInImmunocomp=There is no FDA guidance one the use of {{PAGENAME}} in patients who are immunocompromised. | |||
|useInGeri= | |||
There is no FDA guidance on the use of {{PAGENAME}} with respect to geriatric patients. | |||
|useInGender= | |||
There is no FDA guidance on the use of {{PAGENAME}} with respect to specific gender populations. | |||
|useInRace= | |||
There is no FDA guidance on the use of {{PAGENAME}} with respect to specific racial populations. | |||
|useInRenalImpair= | |||
There is no FDA guidance on the use of {{PAGENAME}} in patients with renal impairment. | |||
|useInHepaticImpair= | |||
There is no FDA guidance on the use of {{PAGENAME}} in patients with hepatic impairment. | |||
|useInReproPotential= | |||
There is no FDA guidance on the use of {{PAGENAME}} in women of reproductive potentials and males. | |||
|useInImmunocomp= | |||
There is no FDA guidance one the use of {{PAGENAME}} in patients who are immunocompromised. | |||
<!--Administration and Monitoring--> | <!--Administration and Monitoring--> | ||
|administration=* Oral | |||
|administration= | |monitoring=There is limited information regarding <i>Monitoring</i> of {{PAGENAME}} in the drug label. | ||
* Oral | |||
|monitoring= | |||
There is limited information regarding <i>Monitoring</i> of {{PAGENAME}} in the drug label. | |||
<!--IV Compatibility--> | <!--IV Compatibility--> | ||
|IVCompat=There is limited information regarding <i>IV Compatibility</i> of {{PAGENAME}} in the drug label. | |||
|IVCompat= | |||
There is limited information regarding <i>IV Compatibility</i> of {{PAGENAME}} in the drug label. | |||
<!--Overdosage--> | <!--Overdosage--> | ||
|overdose====Acute Overdose=== | |||
There is limited information regarding <i>Acute Overdose</i> of {{PAGENAME}} in the drug label. | |||
===Chronic Overdose=== | ===Chronic Overdose=== | ||
| Line 390: | Line 96: | ||
<!--Drug box 2--> | <!--Drug box 2--> | ||
|drugBox=<!--Mechanism of Action--> | |||
|drugBox= | |mechAction=<!--Structure--> | ||
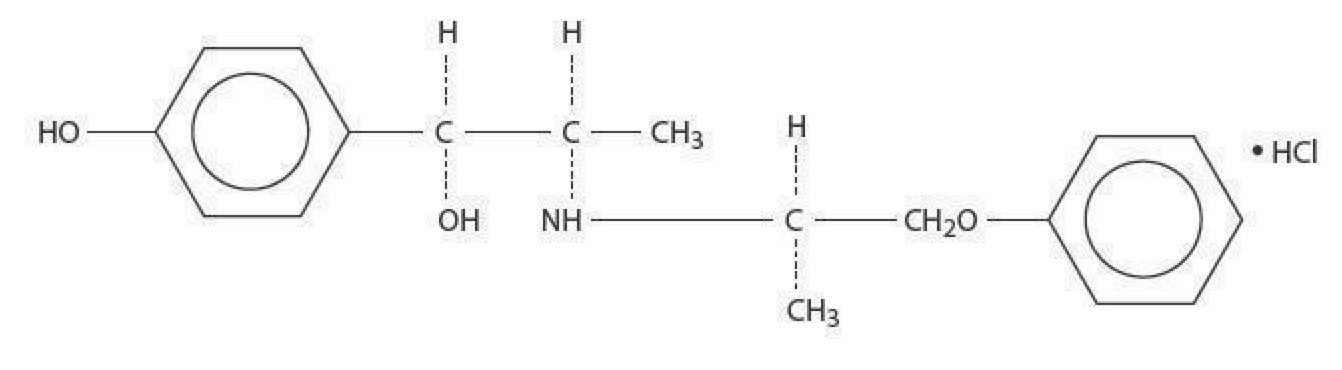
|structure=* lsoxsuprine HCI occurs as a white odorless, crystalline powder, having a bitter taste, It has a following structural formula | |||
<!--Mechanism of Action--> | |||
|mechAction= | |||
<!--Structure--> | |||
|structure= | |||
* | |||
: [[File:{{PAGENAME}}01.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | : [[File:{{PAGENAME}}01.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | ||
<!--Pharmacodynamics--> | <!--Pharmacodynamics--> | ||
|PD=There is limited information regarding <i>Pharmacodynamics</i> of {{PAGENAME}} in the drug label. | |||
|PD= | |||
There is limited information regarding <i>Pharmacodynamics</i> of {{PAGENAME}} in the drug label. | |||
<!--Pharmacokinetics--> | <!--Pharmacokinetics--> | ||
|PK=There is limited information regarding <i>Pharmacokinetics</i> of {{PAGENAME}} in the drug label. | |||
|PK= | |||
There is limited information regarding <i>Pharmacokinetics</i> of {{PAGENAME}} in the drug label. | |||
<!--Nonclinical Toxicology--> | <!--Nonclinical Toxicology--> | ||
|nonClinToxic=There is limited information regarding <i>Nonclinical Toxicology</i> of {{PAGENAME}} in the drug label. | |||
|nonClinToxic= | |||
There is limited information regarding <i>Nonclinical Toxicology</i> of {{PAGENAME}} in the drug label. | |||
<!--Clinical Studies--> | <!--Clinical Studies--> | ||
|clinicalStudies=There is limited information regarding <i>Clinical Studies</i> of {{PAGENAME}} in the drug label. | |||
|clinicalStudies= | |||
There is limited information regarding <i>Clinical Studies</i> of {{PAGENAME}} in the drug label. | |||
<!--How Supplied--> | <!--How Supplied--> | ||
|howSupplied=* lsoxsuprine HCI 20mg tablets are supplied in HDPE containers of 1,000's. | |||
|howSupplied= | |||
* | |||
<!--Patient Counseling Information--> | <!--Patient Counseling Information--> | ||
|packLabel=Bi-Coastal | |||
Pharmaceutical Corp.® | |||
NDC 42582-100-10 | |||
Isoxsuprine | |||
Hydrochloride | |||
Tablets, USP | |||
10 mg | |||
Rx only | |||
100 Tablets | |||
[[File:TA 1- ISOXSUPRINE HYDROCHLORIDE- isoxsuprine hydrochloride tablet.png|thumb|none|400px|This image is provided by the National Library of Medicine.]] | |||
Bi-Coastal | |||
Pharmaceutical Corp.® | |||
NDC 42582-200-10 | |||
Isoxsuprine | |||
Hydrochloride | |||
Tablets, USP | |||
20 mg | |||
Rx only | |||
| | 100 Tablets | ||
[[File:Ta2- ISOXSUPRINE HYDROCHLORIDE- isoxsuprine hydrochloride tablet.png|thumb|none|400px|This image is provided by the National Library of Medicine.]] | |||
[[File:TA- ISOXSUPRINE HYDROCHLORIDE- isoxsuprine hydrochloride tablet.png|thumb|none|400px|This image is provided by the National Library of Medicine.]] | |||
|fdaPatientInfo=There is limited information regarding <i>Patient Counseling Information</i> of {{PAGENAME}} in the drug label. | |||
<!-- | <!--Precautions with Alcohol--> | ||
|alcohol=* Alcohol-{{PAGENAME}} interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication. | |||
| | <!--Brand Names--> | ||
|brandNames=* Vasodilan®<ref>{{Cite web | title = Isoxsuprine Hydrochloride (isoxsuprine hydrochloride) tablet | url = http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=e6582e24-9414-43b8-ac6f-b1422e3197cf }}</ref> | |||
|lookAlike=* N/A<ref name="www.ismp.org">{{Cite web | last = | first = | title = http://www.ismp.org | url = http://www.ismp.org | publisher = | date = }}</ref> | |||
}} | }} | ||
<!--Pill Image--> | |||
<!--Label Display Image--> | <!--Label Display Image--> | ||
<!--Category--> | <!--Category--> | ||
Latest revision as of 16:31, 24 March 2015
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Sheng Shi, M.D. [2]; Turky Alkathery, M.D. [3]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Isoxsuprine is a vasodilator that is FDA approved for the treatment of cerebral vascular insufficiency, arteriosclerosis obliterans, thromboangitis obliterans, and Raynaud's disease. Common adverse reactions include hypotension, tachyarrhythmia, rash, abdominal discomfort, nausea, and dizziness.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Cerebral Vascular Insufficiency, Arteriosclerosis Obliterans, Thromboangitis Obliterans, and Raynaud's Disease
- Dosing Information
- 10 to 20 mg three or four times daily
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Isoxsuprine in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Isoxsuprine in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Isoxsuprine in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Isoxsuprine in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Isoxsuprine in pediatric patients.
Contraindications
- There are no known contraindications to oral use when administered in recommended doses.
Warnings
- Should not be given immediately postpartum or in the presence of arterial bleeding.
Adverse Reactions
Clinical Trials Experience
There is limited information regarding Clinical Trial Experience of Isoxsuprine in the drug label.
Postmarketing Experience
- On rare occasions oral administration of the drug has been associated in time with the occurrences of hypotension, tachycardia, nausea, vomiting, dizziness, abdominal distress, and severe rash. If rash appears the drug should be discontinued.
- Although available evidence suggests a temporal association of these reactions with Isoxsuprine, a casual relationship can be neither confirmed nor refused.
- Maternal and fetal tachycardia may occur under such use. Hypocalcemia, hypoglycemia, hypotension and ileus have been reported to occur in infants whose mothers received Isoxsuprine Hydrochloride. Pulmonary edema has been reported in mothers treated with beta stimulants. Isoxsuprine Hydrochloride is neither approved nor recommended for use in the treatment of premature labor.
Drug Interactions
There is limited information regarding Drug Interactions of Isoxsuprine in the drug label.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA):
There is no FDA guidance on the use of Isoxsuprine with respect to pregnant patients.
Pregnancy Category (AUS):
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Isoxsuprine in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Isoxsuprine during labor and delivery.
Nursing Mothers
There is no FDA guidance on the use of Isoxsuprine with respect to nursing mothers.
Pediatric Use
There is no FDA guidance on the use of Isoxsuprine with respect to pediatric patients.
Geriatic Use
There is no FDA guidance on the use of Isoxsuprine with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Isoxsuprine with respect to specific gender populations.
Race
There is no FDA guidance on the use of Isoxsuprine with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Isoxsuprine in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Isoxsuprine in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Isoxsuprine in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Isoxsuprine in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
Monitoring
There is limited information regarding Monitoring of Isoxsuprine in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Isoxsuprine in the drug label.
Overdosage
Acute Overdose
There is limited information regarding Acute Overdose of Isoxsuprine in the drug label.
Chronic Overdose
There is limited information regarding Chronic Overdose of Isoxsuprine in the drug label.
Pharmacology
There is limited information regarding Isoxsuprine Pharmacology in the drug label.
Mechanism of Action
There is limited information regarding Isoxsuprine Mechanism of Action in the drug label.
Structure
- lsoxsuprine HCI occurs as a white odorless, crystalline powder, having a bitter taste, It has a following structural formula
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Isoxsuprine in the drug label.
Pharmacokinetics
There is limited information regarding Pharmacokinetics of Isoxsuprine in the drug label.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Isoxsuprine in the drug label.
Clinical Studies
There is limited information regarding Clinical Studies of Isoxsuprine in the drug label.
How Supplied
- lsoxsuprine HCI 20mg tablets are supplied in HDPE containers of 1,000's.
Storage
There is limited information regarding Isoxsuprine Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Isoxsuprine |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
Bi-Coastal Pharmaceutical Corp.®
NDC 42582-100-10
Isoxsuprine Hydrochloride Tablets, USP
10 mg
Rx only
100 Tablets

Bi-Coastal Pharmaceutical Corp.®
NDC 42582-200-10
Isoxsuprine Hydrochloride Tablets, USP
20 mg
Rx only
100 Tablets

{{#ask: Label Page::Isoxsuprine |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Isoxsuprine in the drug label.
Precautions with Alcohol
- Alcohol-Isoxsuprine interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Vasodilan®[1]
Look-Alike Drug Names
- N/A[2]
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "Isoxsuprine Hydrochloride (isoxsuprine hydrochloride) tablet".
- ↑ "http://www.ismp.org". External link in
|title=(help)