Immunoglobulin G
|
WikiDoc Resources for Immunoglobulin G |
|
Articles |
|---|
|
Most recent articles on Immunoglobulin G Most cited articles on Immunoglobulin G |
|
Media |
|
Powerpoint slides on Immunoglobulin G |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Immunoglobulin G at Clinical Trials.gov Trial results on Immunoglobulin G Clinical Trials on Immunoglobulin G at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Immunoglobulin G NICE Guidance on Immunoglobulin G
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Immunoglobulin G Discussion groups on Immunoglobulin G Patient Handouts on Immunoglobulin G Directions to Hospitals Treating Immunoglobulin G Risk calculators and risk factors for Immunoglobulin G
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Immunoglobulin G |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
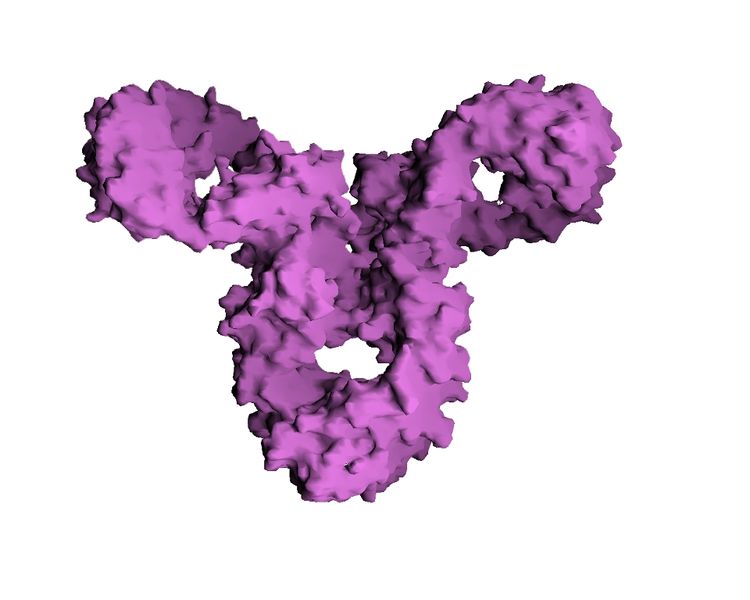
Immunoglobulin G (IgG) is a multimeric immunoglobulin, built of two heavy chains <math>\gamma</math> and two light chains. Each complex has two antigen binding sites. This is the most abundant immunoglobulin and is approximately equally distributed in blood and in tissue liquids, constituting 75% of serum immunoglobulins in humans.[1]
In birds, IgG is often called IgY, and is found in serum and yolk.[2]
Functions
This is the only isotype that can pass through the human placenta, thereby providing protection to the fetus in its first weeks of life before its own immune system has developed.
It can bind to many kinds of pathogens, for example viruses, bacteria, and fungi, and protects the body against them by complement activation (classic pathway), opsonization for phagocytosis and neutralisation of their toxins.
IgG can cause food allergy, and in such causes delayed-onset food allergy, in contrast to food allergy by IgE, whose effects appear rapidly.
Subclasses
| Name | Percent | Crosses placenta easily | Complement activator | Binds to Fc receptors on phagocytic cells |
| IgG1 | 66% | yes | second highest | high affinity |
| IgG2 | 23% | no | third highest | extremely low affinity |
| IgG3 | 7% | yes | highest | high affinity |
| IgG4 | 4% | yes | no | intermediate affinity |
Note: IgG affinity to Fc receptors on phagocytic cells is specific to individual species from which the antibody comes as well as the class.
Related chapters
Resources
References
- ↑ Junqueira, Luiz C. (2003). Basic Histology. McGraw-Hill. ISBN 0838505902. Unknown parameter
|coauthors=ignored (help) - ↑ http://www.sigmaaldrich.com/Brands/Sigma_Genosys/Custom_Antisera/Key_Resources/Antisera_FAQ/Chicken_Antibody.html