Fibroadenoma pathophysiology: Difference between revisions
Ahmed Younes (talk | contribs) No edit summary |
No edit summary |
||
| Line 1: | Line 1: | ||
__NOTOC__ | __NOTOC__ | ||
{{Fibroadenoma}} | {{Fibroadenoma}} | ||
{{CMG}} {{AE}} {{HL}} | {{CMG}} {{AE}} {{IO}} {{HL}} | ||
==Overview== | ==Overview== | ||
Fibroadenoma is a common benign [[tumor]] of the [[breast]]. Fibroadenoma arises from [[connective tissue]] [[cell]]s, which are cells that are normally involved in the functional and mechanical support of the surrounding tissues. Fibroadenomas may demonstrate [[estrogen]] and [[progesterone]] sensitivity and may grow during [[pregnancy]]. The mediator complex subunit 12 (MED12) [[gene]] is the most common gene involved in the pathogenesis of fibroadenoma. On [[gross pathology]], a rubbery, tan colored, and lobulated mass is a characteristic finding of fibroadenoma. On [[microscopic]] pathology, charectersitic findings of fibroadenoma include a biphasic proliferation of both [[stromal]] and [[epithelial]] components that can be arranged in two growth patterns; a pericanalicular growth pattern and an intracanalicular growth pattern. | Fibroadenoma is a common benign [[tumor]] of the [[breast]]. Fibroadenoma arises from [[connective tissue]] [[cell]]s, which are cells that are normally involved in the functional and mechanical support of the surrounding tissues. Fibroadenomas may demonstrate [[estrogen]] and [[progesterone]] sensitivity and may grow during [[pregnancy]]. The mediator complex subunit 12 (MED12) [[gene]] is the most common gene involved in the pathogenesis of fibroadenoma. On [[gross pathology]], a rubbery, tan colored, and lobulated mass is a characteristic finding of fibroadenoma. On [[microscopic]] pathology, charectersitic findings of fibroadenoma include a biphasic proliferation of both [[stromal]] and [[epithelial]] components that can be arranged in two growth patterns; a pericanalicular growth pattern and an intracanalicular growth pattern. | ||
Revision as of 03:27, 14 September 2019
|
Fibroadenoma Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Fibroadenoma pathophysiology On the Web |
|
American Roentgen Ray Society Images of Fibroadenoma pathophysiology |
|
Risk calculators and risk factors for Fibroadenoma pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1] Associate Editor(s)-in-Chief: Ifeoma Odukwe, M.D. [2] Haytham Allaham, M.D. [3]
Overview
Fibroadenoma is a common benign tumor of the breast. Fibroadenoma arises from connective tissue cells, which are cells that are normally involved in the functional and mechanical support of the surrounding tissues. Fibroadenomas may demonstrate estrogen and progesterone sensitivity and may grow during pregnancy. The mediator complex subunit 12 (MED12) gene is the most common gene involved in the pathogenesis of fibroadenoma. On gross pathology, a rubbery, tan colored, and lobulated mass is a characteristic finding of fibroadenoma. On microscopic pathology, charectersitic findings of fibroadenoma include a biphasic proliferation of both stromal and epithelial components that can be arranged in two growth patterns; a pericanalicular growth pattern and an intracanalicular growth pattern.
Pathophysiology
Breast physiology
- Hormones and growth factors act on connective tissue cells in the breast to regulate the development, maturation, and differentiation of mammary glands.[1]
- Estrogen is responsible for mediation of the development of ductal tisuue. Progesterone promotes ductal branching and lobulo-alveolar development. While prolactin regulates the production of milk protein.
- Estrogen and progesterone levels increase at puberty and initiates breast development. This development results in the formation of a complex tree-like structure which comprises of primary milk ducts (about 5 to 10) that originate from the nipple, segmental ducts (about 20 to 40), and sub-segmental ducts (about 10 to 100) that end in terminal duct lobular units. The increase in these hormones during the menstrual cycle stimulates cell proliferation in the luteal phase.
Pathogenesis
- Fibroadenoma is a common benign tumor of the breast. The pathogenesis is not completely understood.
- Fibroadenoma is a proliferation of stromal and epithelial connective tissue cells (biphasic) originating from the terminal duct-lobular unit. Analysis of the stromal and epithelial cells showed that both are polyclonal, which supports the theory that fibroadenomas are hyperplastic lesions associated with a deviation from the normal maturation of the breast, rather than a true neoplasm.[2][3]
- In some patients, fibroadenomas may express estrogen and progesterone receptors. These hormones stimulate the fibroadenomas via hormone-receptor mechanism leading to excessive proliferation of epithelial and stromal cells. They undergo atrophy during menopause.[4]
- Some fibroadenomas may express epidermal growth factor (EGF) receptors.[4]
- More than 70% of fibroadenomas present as a single mass, and 10%–25% of fibroadenomas present as multiple masses.[5]
- Although fibroadenomas may be develop in any part of the breast, there is a significant predilection for the upper outer quadrant. The mass may enlarge slowly without associated pain or nipple and skin changes, but fluctuations in size may occur with the menstrual cycle. [2]
Genetics
- The mediator complex subunit 12 (MED12) gene is involved in the pathophysiology of fibroadenoma. The MED12 gene helps in producing the MED12 protein which with other proteins, is essential for eukaryotic transcriptional regulation.[6][7]
Associated Conditions
Conditions associated with fibroadenoma include:[5]
Gross Pathology
- On gross pathology, a painless, firm, solitary, mobile, and slowly growing breast lump is a characteristic finding of fibroadenoma.
- Other charectersitic findings on gross examination of fibroadenoma include:[8][9][10][11]
- Rubbery texture
- Tan/white colored
- Lobulated appearance
- Short slit-like spaces present
- Calcifications
- Fibroadenomas may sometimes be referred to as a "breast mouse", owing to the high mobility of the tumor through out the breast tissue.
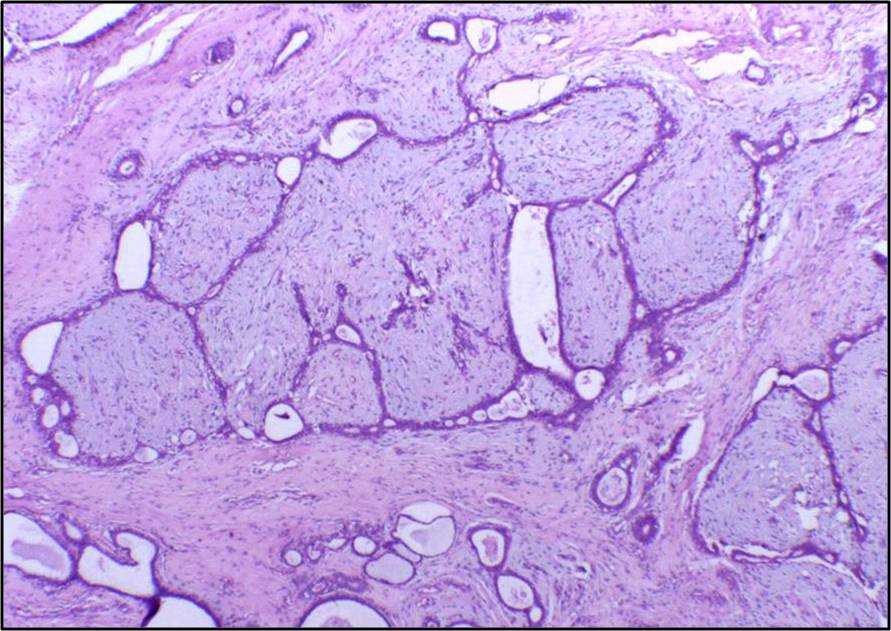
Microscopic Pathology
- Uniformly distributed sheets of epithelial cells arranged in a honeycomb pattern
- Presence of foam and apocrine cells
- Intact basement membrane
- A hypovascular stroma
- Calcification may be present
- Absence of excessive mitotic figures or anaplasia
- Biphasic proliferation of both stromal and epithelial components that can be arranged in two growth patterns:
- Pericanalicular growth pattern: stromal proliferation around epithelial structures
- Intracanalicular growth pattern: stromal proliferation compressing the epithelial structures into clefts
References
- ↑ Feingold KR, Anawalt B, Boyce A, Chrousos G, Dungan K, Grossman A, Hershman JM, Kaltsas G, Koch C, Kopp P, Korbonits M, McLachlan R, Morley JE, New M, Perreault L, Purnell J, Rebar R, Singer F, Trence DL, Vinik A, Wilson DP, Santen RJ. PMID 25905225. Missing or empty
|title=(help) - ↑ 2.0 2.1 Cerrato F, Labow BI (February 2013). "Diagnosis and management of fibroadenomas in the adolescent breast". Semin Plast Surg. 27 (1): 23–5. doi:10.1055/s-0033-1343992. PMC 3706050. PMID 24872735.
- ↑ Noguchi S, Motomura K, Inaji H, Imaoka S, Koyama H (September 1993). "Clonal analysis of fibroadenoma and phyllodes tumor of the breast". Cancer Res. 53 (17): 4071–4. PMID 8395336.
- ↑ 4.0 4.1 Greenberg R, Skornick Y, Kaplan O (September 1998). "Management of breast fibroadenomas". J Gen Intern Med. 13 (9): 640–5. PMC 1497021. PMID 9754521.
- ↑ 5.0 5.1 Lee M, Soltanian HT (2015). "Breast fibroadenomas in adolescents: current perspectives". Adolesc Health Med Ther. 6: 159–63. doi:10.2147/AHMT.S55833. PMC 4562655. PMID 26366109.
- ↑ Ajmal M, Van Fossen K. PMID 30570966. Missing or empty
|title=(help) - ↑ Philibert RA, Madan A (2007). "Role of MED12 in transcription and human behavior". Pharmacogenomics. 8 (8): 909–16. doi:10.2217/14622416.8.8.909. PMID 17716226.
- ↑ 8.0 8.1 Fibroadenoma. Wikipedia (2015) https://en.wikipedia.org/wiki/Fibroadenoma Accessed on January, 29 2016
- ↑ 9.0 9.1 Fibroadenoma. Libre Pathology (2015) http://librepathology.org/wiki/index.php/Fibroadenoma Accessed on January, 29 2016
- ↑ 10.0 10.1 Breast-nonmalignant-Fibroadenoma. PathologyOutlines (2015) http://www.pathologyoutlines.com/topic/breastfibroadenoma.html Accessed on January, 29 2016
- ↑ 11.0 11.1 Fibroadenoma. Radiopaedia (2015) http://radiopaedia.org/articles/fibroadenoma-of-the-breast-1 Accessed on January, 29 2016