Esomeprazole (oral): Difference between revisions
No edit summary |
No edit summary |
||
| Line 285: | Line 285: | ||
<!--Structure--> | <!--Structure--> | ||
|structure= | |structure=: [[File:Esome structure.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | ||
: [[File:Esome structure.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
<!--Pharmacodynamics--> | <!--Pharmacodynamics--> | ||
| Line 320: | Line 319: | ||
:* NDC 0186-4040–01 unit dose packages of 30: 40 mg packets. | :* NDC 0186-4040–01 unit dose packages of 30: 40 mg packets. | ||
|storage=* Store at 25°C (77°F); excursions permitted to 15 to 30°C (59 to 86°F). Keep NEXIUM Delayed-Release Capsules container tightly closed. Dispense in a tight container if the NEXIUM Delayed-Release Capsules product package is subdivided. | |storage=* Store at 25°C (77°F); excursions permitted to 15 to 30°C (59 to 86°F). Keep NEXIUM Delayed-Release Capsules container tightly closed. Dispense in a tight container if the NEXIUM Delayed-Release Capsules product package is subdivided. | ||
|packLabel=[[File: | |packLabel=[[File:Eso 01.jpg|thumb|none|400px|This image is provided by the National Library of Medicine.]] | ||
[[File: | [[File:Eso 02.jpg|thumb|none|400px|This image is provided by the National Library of Medicine.]] | ||
[[File: | [[File:Eso 03.jpg|thumb|none|400px|This image is provided by the National Library of Medicine.]] | ||
[[File: | [[File:Eso 04.jpg|thumb|none|400px|This image is provided by the National Library of Medicine.]] | ||
[[File: | [[File:Eso 05.jpg|thumb|none|400px|This image is provided by the National Library of Medicine.]] | ||
[[File: | [[File:Eso 06.jpg|thumb|none|400px|This image is provided by the National Library of Medicine.]] | ||
[[File: | [[File:Eso 07.jpg|thumb|none|400px|This image is provided by the National Library of Medicine.]] | ||
[[File: | [[File:Eso 08.jpg|thumb|none|400px|This image is provided by the National Library of Medicine.]] | ||
[[File: | [[File:Eso 09.jpg|thumb|none|400px|This image is provided by the National Library of Medicine.]] | ||
[[File: | [[File:DailyMed NEXIUM esomeprazole magnesium capsule delayed release NEXIUM esomeprazole magnesium granule delayed release.png|thumb|none|400px|This image is provided by the National Library of Medicine.]] | ||
|fdaPatientInfo=* Advise patients to let you know if they are taking, or begin taking, other medications, because NEXIUM can interfere with antiretroviral drugs and drugs that are affected by gastric pH changes. | |fdaPatientInfo=* Advise patients to let you know if they are taking, or begin taking, other medications, because NEXIUM can interfere with antiretroviral drugs and drugs that are affected by gastric pH changes. | ||
* Let patients know that antacids may be used while taking NEXIUM. | * Let patients know that antacids may be used while taking NEXIUM. | ||
Revision as of 13:29, 22 May 2015
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Ammu Susheela, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Esomeprazole (oral) is a proton pump inhibitor that is FDA approved for the treatment of erosive esophagitis due to acid-mediated GERD, Zollinger-Ellison Syndrome. Common adverse reactions include abdominal pain, constipation, diarrhea, flatulence, nausea, xerostomia, headache, somnolence.
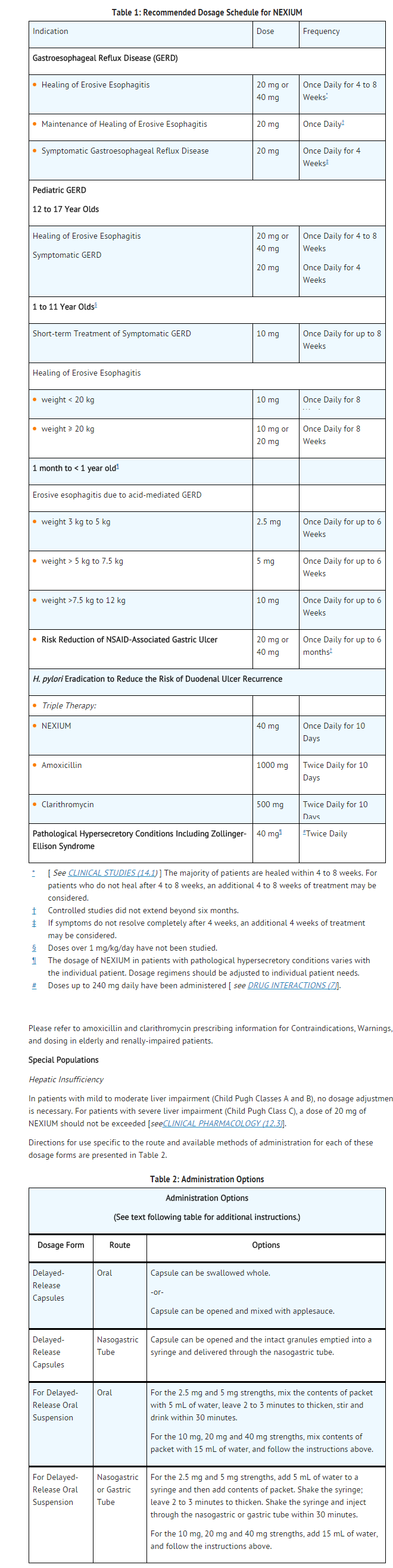
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indication
Treatment of Gastroesophageal Reflux Disease (GERD)
- Healing of Erosive Esophagitis
- NEXIUM is indicated for the short-term treatment (4 to 8 weeks) in the healing and symptomatic resolution of diagnostically confirmed erosive esophagitis. For those patients who have not healed after 4 to 8 weeks of treatment, an additional 4 to 8 week course of NEXIUM may be considered.
- In infants 1 month to less than 1 year, NEXIUM is indicated for short-term treatment (up to 6 weeks) of erosive esophagitis due to acid-mediated GERD.
Maintenance of Healing of Erosive Esophagitis
- NEXIUM is indicated to maintain symptom resolution and healing of erosive esophagitis. Controlled studies do not extend beyond 6 months.
Symptomatic Gastroesophageal Reflux Disease
- NEXIUM is indicated for short-term treatment (4 to 8 weeks) of heartburn and other symptoms associated with GERD in adults and children 1 year or older.
Risk Reduction of NSAID-Associated Gastric Ulcer
- NEXIUM is indicated for the reduction in the occurrence of gastric ulcers associated with continuous NSAID therapy in patients at risk for developing gastric ulcers. Patients are considered to be at risk due to their age (≥ 60) and/or documented history of gastric ulcers. Controlled studies do not extend beyond 6 months.
H. pylori Eradication to Reduce the Risk of Duodenal Ulcer Recurrence
- Triple Therapy (NEXIUM plus amoxicillin and clarithromycin): NEXIUM, in combination with amoxicillin and clarithromycin, is indicated for the treatment of patients with H. pylori infection and duodenal ulcer disease (active or history of within the past 5 years) to eradicate H. pylori. Eradication of H. pylori has been shown to reduce the risk of duodenal ulcer recurrence.
- In patients who fail therapy, susceptibility testing should be done. If resistance to clarithromycin is demonstrated or susceptibility testing is not possible, alternative antimicrobial therapy should be instituted.
Pathological Hypersecretory Conditions Including Zollinger-Ellison Syndrome
- NEXIUM is indicated for the long-term treatment of pathological hypersecretory conditions, including Zollinger-Ellison Syndrome.
Dosage
NEXIUM Delayed-Release Capsules
- NEXIUM Delayed-Release Capsules should be swallowed whole.
- Alternatively, for patients who have difficulty swallowing capsules, one tablespoon of applesauce can be added to an empty bowl and the NEXIUM Delayed-Release Capsule can be opened, and the granules inside the capsule carefully emptied onto the applesauce. The granules should be mixed with the applesauce and then swallowed immediately: do not store for future use. The applesauce used should not be hot and should be soft enough to be swallowed without chewing. The granules should not be chewed or crushed. If the granules/applesauce mixture is not used in its entirety, the remaining mixture should be discarded immediately.
- For patients who have a nasogastric tube in place, NEXIUM Delayed-Release Capsules can be opened and the intact granules emptied into a 60 mL catheter tipped syringe and mixed with 50 mL of water. It is important to only use a catheter tipped syringe when administering NEXIUM through a nasogastric tube. Replace the plunger and shake the syringe vigorously for 15 seconds. Hold the syringe with the tip up and check for granules remaining in the tip. Attach the syringe to a nasogastric tube and deliver the contents of the syringe through the nasogastric tube into the stomach. After administering the granules, the nasogastric tube should be flushed with additional water. Do not administer the granules if they have dissolved or disintegrated.
- The mixture must be used immediately after preparation.
- NEXIUM For Delayed-Release Oral Suspension
- NEXIUM For Delayed-Release Oral Suspension should be administered as follows:
- Empty the contents of a 2.5 mg or 5 mg packet into a container containing 5 mL of water. For the 10 mg, 20 mg, and 40 mg strengths, the contents of a packet should be emptied into a container containing 15 mL of water.
Stir.
- Leave 2 to 3 minutes to thicken.
- Stir and drink within 30 minutes.
- If any medicine remains after drinking, add more water, stir, and drink immediately.
- In cases where there is a need to use two packets, they may be mixed in a similar way by adding twice the required amount of water or follow the mixing instructions provided by your pharmacist or doctor.
- For patients who have a nasogastric or gastric tube in place, NEXIUM For Delayed-Release Oral Suspension can be administered as follows:
- Add 5 mL of water to a catheter tipped syringe and then add the contents of a 2.5 mg or 5 mg NEXIUM packet. For the 10 mg, 20 mg, and 40 mg strengths, the volume of water in the syringe should be 15 mL. It is important to only use a catheter tipped syringe when administering NEXIUM through a nasogastric tube or gastric tube.
- Immediately shake the syringe and leave 2 to 3 minutes to thicken.
- Shake the syringe and inject through the nasogastric or gastric tube, French size 6 or larger, into the stomach within 30 minutes.
- Refill the syringe with an equal amount of water (5 mL or 15 mL).
- Shake and flush any remaining contents from the nasogastric or gastric tube into the stomach.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Esomeprazole (oral) in adult patients.
Non–Guideline-Supported Use
- Duodenal ulcer disease - Helicobacter pylori gastrointestinal tract infection
- Gastric ulcer; Prophylaxis - NSAID-associated gastropathy; Prophylaxis
- Zollinger-Ellison syndrome
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Esomeprazole (oral) in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Esomeprazole (oral) in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Esomeprazole (oral) in pediatric patients.
Contraindications
- NEXIUM is contraindicated in patients with known hypersensitivity to substituted benzimidazoles or to any component of the formulation. Hypersensitivity reactions may include anaphylaxis, anaphylactic shock, angioedema, bronchospasm, acute interstitial nephritis, and urticaria.
- For information about contraindications of antibacterial agents (clarithromycin and amoxicillin) indicated in combination with NEXIUM, refer to the CONTRAINDICATIONS section of their package inserts.
Warnings
Concurrent Gastric Malignancy
- Symptomatic response to therapy with NEXIUM does not preclude the presence of gastric malignancy.
Atrophic Gastritis
- Atrophic gastritis has been noted occasionally in gastric corpus biopsies from patients treated long-term with omeprazole, of which esomeprazole is an enantiomer.
Acute Interstitial Nephritis
- Acute interstitial nephritis has been observed in patients taking PPIs including NEXIUM. Acute interstitial nephritis may occur at any point during PPI therapy and is generally attributed to an idiopathic hypersensitivity reaction. Discontinue NEXIUM if acute interstitial nephritis develops.
Cyanocobalamin (vitamin B-12) Deficiency
- Daily treatment with any acid-suppressing medications over a long period of time (e.g., longer than 3 years) may lead to malabsorption of cyanocobalamin (vitamin B-12) caused by hypo- or achlorhydria. Rare reports of cyanocobalamin deficiency occurring with acid-supressing therapy have been reported in the literature. This diagnosis should be considered if clinical symptoms consistent with cyanocobalamin deficiency are observed.
Clostridium difficile associated diarrhea
- Published observational studies suggest that PPI therapy like NEXIUM may be associated with an increased risk of Clostridium difficile associated diarrhea, especially in hospitalized patients. This diagnosis should be considered for diarrhea that does not improve.
- Patients should use the lowest dose and shortest duration of PPI therapy appropriate to the condition being treated.
- Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents. For more information specific to antibacterial agents (clarithromycin and amoxicillin) indicated for use in combination with NEXIUM, refer to WARNINGS and PRECAUTIONS sections of those package inserts.
Interaction with Clopidogrel
- Avoid concomitant use of NEXIUM with clopidogrel. Clopidogrel is a prodrug. Inhibition of platelet aggregation by clopidogrel is entirely due to an active metabolite. The metabolism of clopidogrel to its active metabolite can be impaired by use with concomitant medications, such as esomeprazole, that inhibit CYP2C19 activity. Concomitant use of clopidogrel with 40 mg esomeprazole reduces the pharmacological activity of clopidogrel. When using NEXIUM consider alternative anti-platelet therapy.
Bone Fracture
- Several published observational studies suggest that proton pump inhibitor (PPI) therapy may be associated with an increased risk for osteoporosis-related fractures of the hip, wrist, or spine. The risk of fracture was increased in patients who received high-dose, defined as multiple daily doses, and long-term PPI therapy (a year or longer). Patients should use the lowest dose and shortest duration of PPI therapy appropriate to the condition being treated. Patients at risk for osteoporosis-related fractures should be managed according to established treatment guidelines.
Hypomagnesemia
- Hypomagnesemia, symptomatic and asymptomatic, has been reported rarely in patients treated with PPIs for at least three months, in most cases after a year of therapy. Serious adverse events include tetany, arrhythmias, and seizures. In most patients, treatment of hypomagnesemia required magnesium replacement and discontinuation of the PPI.
- For patients expected to be on prolonged treatment or who take PPIs with medications such as digoxin or drugs that may cause hypomagnesemia (e.g., diuretics), health care professionals may consider monitoring magnesium levels prior to initiation of PPI treatment and periodically.
Concomitant use of NEXIUM with St. John’s Wort or Rifampin
Drugs which induce CYP2C19 or CYP3A4 (such as St. John’s Wort or rifampin) can substantially decrease esomeprazole concentration. Avoid concomitant use of NEXIUM with St. John’s Wort or rifampin.
Interactions with Diagnostic Investigations for Neuroendocrine Tumors
- Serum chromogranin A (CgA) levels increase secondary to drug-induced decreases in gastric acidity. The increased CgA level may cause false positive results in diagnostic investigations for neuroendocrine tumors. Healthcare providers should temporarily stop esomeprazole treatment at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high. If serial tests are performed (e.g., for monitoring), the same commercial laboratory should be used for testing, as reference ranges between tests may vary.
Concomitant use of NEXIUM with Methotrexate
- Literature suggests that concomitant use of PPIs with methotrexate (primarily at high dose; see methotrexate prescribing information) may elevate and prolong serum levels of methotrexate and/or its metabolite, possibly leading to methotrexate toxicities. In high-dose methotrexate administration a temporary withdrawal of the PPI may be considered in some patients.
Adverse Reactions
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adults
- The safety of NEXIUM was evaluated in over 15,000 patients (aged 18 to 84 years) in clinical trials worldwide including over 8,500 patients in the United States and over 6,500 patients in Europe and Canada. Over 2,900 patients were treated in long-term studies for up to 6-12 months. In general, NEXIUM was well tolerated in both short and long-term clinical trials.
- The safety in the treatment of healing of erosive esophagitis was assessed in four randomized comparative clinical trials, which included 1,240 patients on NEXIUM 20 mg, 2,434 patients on NEXIUM 40 mg, and 3,008 patients on omeprazole 20 mg daily. The most frequently occurring adverse reactions (≥1%) in all three groups were headache (5.5, 5, and 3.8, respectively) and diarrhea (no difference among the three groups). Nausea, flatulence, abdominal pain, constipation, and dry mouth occurred at similar rates among patients taking NEXIUM or omeprazole.
- Additional adverse reactions that were reported as possibly or probably related to NEXIUM with an incidence <1% are listed below by body system:
- Body as a Whole: abdomen enlarged, allergic reaction, asthenia, back pain, chest pain, substernal chest pain, facial edema, peripheral edema, hot flushes, fatigue, fever, flu-like disorder, generalized edema, leg edema, malaise, pain, rigors;
- Cardiovascular: flushing, hypertension, tachycardia;
- Endocrine: goiter;
- Gastrointestinal: bowel irregularity, constipation aggravated, dyspepsia, dysphagia, dysplasia GI, epigastric pain, eructation, esophageal disorder, frequent stools, gastroenteritis, GI hemorrhage, GI symptoms not otherwise specified, hiccup, melena, mouth disorder, pharynx disorder, rectal disorder, serum gastrin increased, tongue disorder, tongue edema, ulcerative stomatitis, vomiting;
- Hearing: earache, tinnitus;
- Hematologic: anemia, anemia hypochromic, cervical lymphadenopathy, epistaxis, leukocytosis, leukopenia, thrombocytopenia;
- Hepatic: bilirubinemia, hepatic function abnormal, SGOT increased, SGPT increased;
- Metabolic/Nutritional: glycosuria, hyperuricemia, hyponatremia, increased alkaline phosphatase, thirst, vitamin B12 deficiency, weight increase, weight decrease;
- Musculoskeletal: arthralgia, arthritis aggravated, arthropathy, cramps, fibromyalgia syndrome, hernia, polymyalgia rheumatica;
- Nervous System/Psychiatric: anorexia, apathy, appetite increased, confusion, depression aggravated, dizziness, hypertonia, nervousness, hypoesthesia, impotence, insomnia, migraine, migraine aggravated, paresthesia, sleep disorder, somnolence, tremor, vertigo, visual field defect;
- Reproductive: dysmenorrhea, menstrual disorder, vaginitis;
- Respiratory: asthma aggravated, coughing, dyspnea, larynx edema, pharyngitis, rhinitis, sinusitis;
- Skin and Appendages: acne, angioedema, dermatitis, pruritus, pruritus ani, rash, rash erythematous, rash maculo-papular, skin inflammation, sweating increased, urticaria;
- Special Senses: otitis media, parosmia, taste loss, taste perversion;
- Urogenital: abnormal urine, albuminuria, cystitis, dysuria, fungal infection, hematuria, micturition frequency, moniliasis, genital moniliasis, polyuria;
- Visual: conjunctivitis, vision abnormal.
- The following potentially clinically significant laboratory changes in clinical trials, irrespective of relationship to NEXIUM, were reported in ≤1% of patients: increased creatinine, uric acid, total bilirubin, alkaline phosphatase, ALT, AST, hemoglobin, white blood cell count, platelets, serum gastrin, potassium, sodium, thyroxine and thyroid stimulating hormone. Decreases were seen in hemoglobin, white blood cell count, platelets, potassium, sodium, and thyroxine.
- Endoscopic findings that were reported as adverse reactions include: duodenitis, esophagitis, esophageal stricture, esophageal ulceration, esophageal varices, gastric ulcer, gastritis, hernia, benign polyps or nodules, Barrett’s esophagus, and mucosal discoloration.
- The incidence of treatment-related adverse reactions during 6-month maintenance treatment was similar to placebo. There were no differences in types of related adverse reactions seen during maintenance treatment up to 12 months compared to short-term treatment.
- Two placebo-controlled studies were conducted in 710 patients for the treatment of symptomatic gastroesophageal reflux disease. The most common adverse reactions that were reported as possibly or probably related to NEXIUM were diarrhea (4.3%), headache (3.8%), and abdominal pain (3.8%).
Pediatrics
- The safety of NEXIUM was evaluated in 316 pediatric and adolescent patients aged 1 to 17 years in four clinical trials for the treatment of symptomatic GERD. In 109 pediatric patients aged 1 to 11 years, the most frequently reported (at least 1%) treatment-related adverse reactions in these patients were diarrhea (2.8%), headache (1.9%) and somnolence (1.9%). In 149 pediatric patients aged 12 to 17 years the most frequently reported (at least 2%) treatment-related adverse reactions in these patients were headache (8.1%), abdominal pain (2.7%), diarrhea (2%), and nausea (2%).
- The safety of NEXIUM was evaluated in 167 pediatric patients from birth to <1 year of age in three clinical trials [seeCLINICAL STUDIES (14.3)]. In a study that included 26 pediatric patients aged birth to 1 month there were no treatment related adverse reactions. In a study that included 43 pediatric patients age 1 to 11 months, inclusive the most frequently reported (at least 5%) adverse reactions, irrespective of causality, were irritability and vomiting. In a study that included 98 pediatric patients, age 1 to 11 months, inclusive exposed to esomeprazole for up to 6 weeks (including 39 patients randomized to the withdrawal phase), there were 4 treatment-related adverse reactions: abdominal pain (1%), regurgitation (1%), tachypnea (1%), and increased ALT (1%).
- No new safety concerns were identified in pediatric patients.
- Combination Treatment with Amoxicillin and Clarithromycin
- In clinical trials using combination therapy with NEXIUM plus amoxicillin and clarithromycin, no additional adverse reactions specific to these drug combinations were observed. Adverse reactions that occurred were limited to those observed when using NEXIUM, amoxicillin, or clarithromycin alone.
- The most frequently reported drug-related adverse reactions for patients who received triple therapy for 10 days were diarrhea (9.2%), taste perversion (6.6%), and abdominal pain (3.7%). No treatment-emergent adverse reactions were observed at higher rates with triple therapy than were observed with NEXIUM alone.
- For more information on adverse reactions with amoxicillin or clarithromycin, refer to their package inserts, Adverse Reactions sections.
- In clinical trials using combination therapy with NEXIUM plus amoxicillin and clarithromycin, no additional increased laboratory abnormalities particular to these drug combinations were observed.
- For more information on laboratory changes with amoxicillin or clarithromycin, refer to their package inserts, Adverse Reactions section.
Postmarketing Experience
- The following adverse reactions have been identified during post-approval use of NEXIUM. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These reports are listed below by body system:
- Blood and Lymphatic: agranulocytosis, pancytopenia;
- Eye: blurred vision;
- Gastrointestinal: pancreatitis; stomatitis; microscopic colitis
- Hepatobiliary: hepatic failure, hepatitis with or without jaundice;
- Immune System: anaphylactic reaction/shock;
- Infections and Infestations: GI candidiasis; Clostridium difficile associated diarrhea;
- Metabolism and nutritional disorders: hypomagnesemia, with or without hypocalcemia and/or hypokalemia
- Musculoskeletal and Connective Tissue: muscular weakness, myalgia, bone fracture;
- Nervous System: hepatic encephalopathy, taste disturbance;
- Psychiatric: aggression, agitation, depression, hallucination;
- Renal and Urinary: interstitial nephritis;
- Reproductive System and Breast: gynecomastia;
- Respiratory, Thoracic, and Mediastinal: bronchospasm;
- Skin and Subcutaneous Tissue: alopecia, erythema multiforme, hyperhidrosis, photosensitivity, Stevens-Johnson syndrome, toxic epidermal necrolysis (some fatal).
Drug Interactions
Interference with Antiretroviral Therapy
- Concomitant use of atazanavir and nelfinavir with proton pump inhibitors is not recommended. Co-administration of atazanavir with proton pump inhibitors is expected to substantially decrease atazanavir plasma concentrations and may result in a loss of therapeutic effect and the development of drug resistance. Co-administration of saquinavir with proton pump inhibitors is expected to increase saquinavir concentrations, which may increase toxicity and require dose reduction.
- Omeprazole, of which esomeprazole is an enantiomer, has been reported to interact with some antiretroviral drugs. The clinical importance and the mechanisms behind these interactions are not always known. Increased gastric pH during omeprazole treatment may change the absorption of the antiretroviral drug. Other possible interaction mechanisms are via CYP2C19.
- Reduced concentrations of atazanavir and nelfinavir
- For some antiretroviral drugs, such as atazanavir and nelfinavir, decreased serum levels have been reported when given together with omeprazole. Following multiple doses of nelfinavir (1250 mg, twice daily) and omeprazole (40 mg daily), AUC was decreased by 36% and 92%, Cmax by 37% and 89% and Cmin by 39% and 75% respectively for nelfinavir and M8. Following multiple doses of atazanavir (400 mg, daily) and omeprazole (40 mg, daily, 2 hr before atazanavir), AUC was decreased by 94%, Cmax by 96%, and Cmin by 95%. Concomitant administration with omeprazole and drugs such as atazanavir and nelfinavir is therefore not recommended.
- Increased concentrations of saquinavir
- For other antiretroviral drugs, such as saquinavir, elevated serum levels have been reported, with an increase in AUC by 82%, in Cmax by 75%, and in Cmin by 106%, following multiple dosing of saquinavir/ritonavir (1000/100 mg) twice daily for 15 days with omeprazole 40 mg daily co-administered days 11 to 15. Therefore, clinical and laboratory monitoring for saquinavir toxicity is recommended during concurrent use with NEXIUM. Dose reduction of saquinavir should be considered from the safety perspective for individual patients.
- There are also some antiretroviral drugs of which unchanged serum levels have been reported when given with omeprazole.
Drugs for Which Gastric pH Can Affect Bioavailability
- Due to its effects on gastric acid secretion, esomeprazole can reduce the absorption of drugs where gastric pH is an important determinant of their bioavailability. Like with other drugs that decrease the intragastric acidity, the absorption of drugs such as ketoconazole, atazanavir, iron salts, erlotinib, and mycophenolate mofetil (MMF) can decrease, while the absorption of drugs such as digoxin can increase during treatment with esomeprazole. Esomeprazole is an enantiomer of omeprazole. Concomitant treatment with omeprazole (20 mg daily) and digoxin in healthy subjects increased the bioavailability of digoxin by 10% (30% in two subjects). Co-administration of digoxin with NEXIUM is expected to increase the systemic exposure of digoxin. Therefore, patients may need to be monitored when digoxin is taken concomitantly with NEXIUM.
- Co-administration of omeprazole in healthy subjects and in transplant patients receiving MMF has been reported to reduce the exposure to the active metabolite, mycophenolic acid (MPA), possibly due to a decrease in MMF solubility at an increased gastric pH. The clinical relevance of reduced MPA exposure on organ rejection has not been established in transplant patients receiving NEXIUM and MMF. Use NEXIUM with caution in transplant patients receiving MMF.
Effects on Hepatic Metabolism/Cytochrome P-450 Pathways
- Esomeprazole is extensively metabolized in the liver by CYP2C19 and CYP3A4. In vitro and in vivo studies have shown that esomeprazole is not likely to inhibit CYPs 1A2, 2A6, 2C9, 2D6, 2E1, and 3A4. No clinically relevant interactions with drugs metabolized by these CYP enzymes would be expected. Drug interaction studies have shown that esomeprazole does not have any clinically significant interactions with phenytoin, warfarin, quinidine, clarithromycin, or amoxicillin.
- However, post-marketing reports of changes in prothrombin measures have been received among patients on concomitant warfarin and esomeprazole therapy. Increases in INR and prothrombin time may lead to abnormal bleeding and even death. Patients treated with proton pump inhibitors and warfarin concomitantly may need to be monitored for increases in INR and prothrombin time.
- Esomeprazole may potentially interfere with CYP2C19, the major esomeprazole metabolizing enzyme. Co-administration of esomeprazole 30 mg and diazepam, a CYP2C19 substrate, resulted in a 45% decrease in clearance of diazepam.
Clopidogrel
- Clopidogrel is metabolized to its active metabolite in part by CYP2C19. Concomitant use of esomeprazole 40 mg results in reduced plasma concentrations of the active metabolite of clopidogrel and a reduction in platelet inhibition. Avoid concomitant administration of NEXIUM with clopidogrel. When using NEXIUM, consider use of alternative anti-platelet therapy.
- Omeprazole acts as an inhibitor of CYP2C19. Omeprazole, given in doses of 40 mg daily for one week to 20 healthy subjects in cross-over study, increased Cmax and AUC of cilostazol by 18% and 26% respectively. Cmax and AUC of one of its active metabolites, 3,4-dihydrocilostazol, which has 4-7 times the activity of cilostazol, were increased by 29% and 69% respectively. Co-administration of cilostazol with esomeprazole is expected to increase concentrations of cilostazol and its above mentioned active metabolite. Therefore a dose reduction of cilostazol from 100 mg twice daily to 50 mg twice daily should be considered.
- Concomitant administration of esomeprazole and a combined inhibitor of CYP2C19 and CYP3A4, such as voriconazole, may result in more than doubling of the esomeprazole exposure. Dose adjustment of esomeprazole is not normally required. However, in patients with Zollinger-Ellison’s Syndrome, who may require higher doses up to 240 mg/day, dose adjustment may be considered.
- Drugs known to induce CYP2C19 or CYP3A4 or both (such as rifampin) may lead to decreased esomeprazole serum levels. Omeprazole, of which esomeprazole is an enantiomer, has been reported to interact with St. John’s Wort, an inducer of CYP3A4. In a cross-over study in 12 healthy male subjects, St. John’s Wort (300 mg three times daily for 14 days) significantly decreased the systemic exposure of omeprazole in CYP2C19 poor metabolisers (Cmax and AUC decreased by 37.5% and 37.9%, respectively) and extensive metabolisers (Cmax and AUC decreased by 49.6 % and 43.9%, respectively). Avoid concomitant use of St. John’s Wort or rifampin with NEXIUM.
Interactions With Investigations of Neuroendocrine Tumors
- Drug-induced decrease in gastric acidity results in enterochromaffin-like cell hyperplasia and increased Chromogranin A levels which may interfere with investigations for neuroendocrine tumors.
Tacrolimus
- Concomitant administration of esomeprazole and tacrolimus may increase the serum levels of tacrolimus.
Combination Therapy with Clarithromycin
- Co-administration of esomeprazole, clarithromycin, and amoxicillin has resulted in increases in the plasma levels of esomeprazole and 14-hydroxyclarithromycin.
- Concomitant administration of clarithromycin with other drugs can lead to serious adverse reactions due to drug interactions [see Warnings and Precautions in prescribing information for clarithromycin]. Because of these drug interactions, clarithromycin is contraindicated for co-administration with certain drugs.
Methotrexate
- Case reports, published population pharmacokinetic studies, and retrospective analyses suggest that concomitant administration of PPIs and methotrexate (primarily at high dose; see methotrexate prescribing information) may elevate and prolong serum levels of methotrexate and/or its metabolite hydroxymethotrexate. However, no formal drug interaction studies of methotrexate with PPIs have been conducted.
Use in Specific Populations
Pregnancy
Risk Summary
- There are no adequate and well-controlled studies with NEXIUM in pregnant women. Esomeprazole is the S-isomer of omeprazole. Available epidemiologic data fail to demonstrate an increased risk of major congenital malformations or other adverse pregnancy outcomes with first trimester omeprazole use.
- Teratogenicity was not observed in animal reproduction studies with administration of oral esomeprazole magnesium in rats and rabbits with doses about 68 times and 42 times, respectively, an oral human dose of 40 mg (based on a body surface area basis for a 60 kg person). However, changes in bone morphology were observed in offspring of rats dosed through most of pregnancy and lactation at doses equal to or greater than approximately 34 times an oral human dose of 40 mg (see Animal Data). Because of the observed effect at high doses of esomeprazole magnesium on developing bone in rat studies, NEXIUM should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Human Data
- Esomeprazole is the S-isomer of omeprazole. Four epidemiological studies compared the frequency of congenital abnormalities among infants born to women who used omeprazole during pregnancy with the frequency of abnormalities among infants of women exposed to H2-receptor antagonists or other controls.
- A population-based retrospective cohort epidemiological study from the Swedish Medical Birth Registry, covering approximately 99% of pregnancies, from 1995-99, reported on 955 infants (824 exposed during the first trimester with 39 of these exposed beyond first trimester, and 131 exposed after the first trimester) whose mothers used omeprazole during pregnancy. The number of infants exposed in utero to omeprazole that had any malformation, low birth weight, low Apgar score, or hospitalization was similar to the number observed in this population. The number of infants born with ventricular septal defects and the number of stillborn infants was slightly higher in the omeprazole-exposed infants than the expected number in this population.
- A population-based retrospective cohort study covering all live births in Denmark from 1996-2009, reported on 1,800 live births whose mothers used omeprazole during the first trimester of pregnancy and 837, 317 live births whose mothers did not use any proton pump inhibitor. The overall rate of birth defects in infants born to mothers with first trimester exposure to omeprazole was 2.9% and 2.6% in infants born to mothers not exposed to any proton pump inhibitor during the first trimester.
- A retrospective cohort study reported on 689 pregnant women exposed to either H2-blockers or omeprazole in the first trimester (134 exposed to omeprazole) and 1,572 pregnant women unexposed to either during the first trimester. The overall malformation rate in offspring born to mothers with first trimester exposure to omeprazole, an H2-blocker, or were unexposed was 3.6%, 5.5%, and 4.1% respectively.
- A small prospective observational cohort study followed 113 women exposed to omeprazole during pregnancy (89% first trimester exposures). The reported rate of major congenital malformations was 4% in the omeprazole group, 2% in controls exposed to non-teratogens, and 2.8% in disease paired controls. Rates of spontaneous and elective abortions, preterm deliveries, gestational age at delivery, and mean birth weight were similar among the groups.
- Several studies have reported no apparent adverse short-term effects on the infant when single dose oral or intravenous omeprazole was administered to over 200 pregnant women as premedication for cesarean section under general anesthesia.
Animal Data
- Reproduction studies have been performed with esomeprazole magnesium in rats at oral doses up to 280 mg/kg/day (about 68 times an oral human dose of 40 mg on a body surface area basis) and in rabbits at oral doses up to 86 mg/kg/day (about 41 times an oral human dose of 40 mg on a body surface area basis) and have revealed no evidence of impaired fertility or harm to the fetus due to esomeprazole magnesium.
- A pre- and postnatal developmental toxicity study in rats with additional endpoints to evaluate bone development was performed with esomeprazole magnesium at oral doses of 14 to 280 mg/kg/day (about 3.4 to 68 times an oral human dose of 40 mg on a body surface area basis). Neonatal/early postnatal (birth to weaning) survival was decreased at doses equal to or greater than 138 mg/kg/day (about 34 times an oral human dose of 40 mg on a body surface area basis). Body weight and body weight gain were reduced and neurobehavioral or general developmental delays in the immediate post-weaning timeframe were evident at doses equal to or greater than 69 mg/kg/day (about 17 times an oral human dose of 40 mg on a body surface area basis). In addition, decreased femur length, width and thickness of cortical bone, decreased thickness of the tibial growth plate and minimal to mild bone marrow hypocellularity were noted at doses equal to or greater than 14 mg/kg/day (about 3.4 times an oral human dose of 40 mg on a body surface area basis). Physeal dysplasia in the femur was observed in offspring of rats treated with oral doses of esomeprazole magnesium at doses equal to or greater than 138 mg/kg/day (about 34 times an oral human dose of 40 mg on a body surface area basis).
- Effects on maternal bone were observed in pregnant and lactating rats in a pre- and postnatal toxicity study when esomeprazole magnesium was administered at oral doses of 14 to 280 mg/kg/day (about 3.4 to 68 times an oral human dose of 40 mg on a body surface area basis). When rats were dosed from gestational day 7 through weaning on postnatal day 21, a statistically significant decrease in maternal femur weight of up to 14% (as compared to placebo treatment) was observed at doses equal to or greater than 138 mg/kg/day (about 34 times an oral human dose of 40 mg on a body surface area basis).
- A pre- and postnatal development study in rats with esomeprazole strontium (using equimolar doses compared to esomeprazole magnesium study) produced similar results in dams and pups as described above.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Esomeprazole (oral) in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Esomeprazole (oral) during labor and delivery.
Nursing Mothers
- Esomeprazole is likely present in human milk. Esomeprazole is the S-isomer of omeprazole and limited data indicate that maternal doses of omeprazole 20 mg daily produce low levels in human milk. Caution should be exercised when Nexium is administered to a nursing woman.
Pediatric Use
- The safety and effectiveness of NEXIUM have been established in pediatric patients 1 to 17 years of age for short-term treatment (up to eight weeks) of GERD. The safety and effectiveness of NEXIUM have been established in pediatric patients 1 month to less than 1 year for short-term treatment (up to 6 weeks) of erosive esophagitis due to acid-mediated GERD. However, the safety and effectiveness of NEXIUM have not been established in patients less than 1 month of age.
- 1 to 17 years of age
- Use of NEXIUM in pediatric and adolescent patients 1 to 17 years of age for short-term treatment (up to eight weeks) of GERD is supported by extrapolation of results from adequate and well-controlled studies for adults and safety and pharmacokinetic studies performed in pediatric and adolescent patients. The safety and effectiveness of NEXIUM for other pediatric uses have not been established.
- Erosive esophagitis due to acid-mediated GERD in infants 1 month to less than on year of age.
- Use of NEXIUM in pediatric patients 1 month to less than 1 year of age for treatment (up to 6 weeks) of erosive esophagitis due to acid-mediated GERD is supported by extrapolation of results from adequate and well-controlled studies for adults and safety, pharmacokinetic, and pharmacodynamic studies performed in pediatric patients.
- Symptomatic GERD in infants 1 month to less than one year of age
- There was no statistically significant difference between NEXIUM and placebo in the rate of discontinuation due to symptom worsening in a multicenter, randomized, double-blind, controlled, treatment-withdrawal study of 98 patients ages 1 to 11 months, inclusive. Patients were enrolled if they had either a clinical diagnosis of suspected GERD, symptomatic GERD, or endoscopically proven GERD. Twenty of 98 enrolled patients underwent endoscopy, and 6 patients were found to have erosive esophagitis on endoscopy at baseline. All patients received NEXIUM Delayed-Release Oral Suspension once daily during a two-week, open-label phase of the study.
- There were 80 patients who attained a pre-specified level of symptom improvement and who entered the double-blind phase, in which they were randomized in equal proportions to receive NEXIUM or placebo for the next four weeks. Efficacy was assessed by observing the time from randomization to study discontinuation due to symptom worsening during the four-week, treatment-withdrawal phase.
- The following pharmacokinetic and pharmacodynamic information was obtained in pediatric patients with GERD aged birth to less than one year of age. In infants (1 to 11 months old, inclusive) given NEXIUM 1 mg/kg once daily, the percent time with intragastric pH > 4 increased from 29% at baseline to 69% on Day 7, which is similar to the pharmacodynamic effect in adults. Apparent clearance (CL/F) increases with age in pediatric patients from birth to 2 years of age.
- Neonates 0 to 1 month of age
- Following administration of oral NEXIUM in neonates the geometric mean (range) for the apparent clearance (CL/F) was 0.55 L/h/kg (0.25-1.6 L/h/kg).
- The safety and effectiveness of NEXIUM in neonates have not been established.
Juvenile Animal Data
- In a juvenile rat toxicity study, esomeprazole was administered with both magnesium and strontium salts at oral doses about 34 to 68 times a daily human dose of 40 mg based on body surface area. Increases in death were seen at the high dose, and at all doses of esomeprazole, there were decreases in body weight, body weight gain, femur weight and femur length, and decreases in overall growth.
Geriatic Use
- Of the total number of patients who received NEXIUM in clinical trials, 1459 were 65 to 74 years of age and 354 patients were ≥ 75 years of age.
- No overall differences in safety and efficacy were observed between the elderly and younger individuals, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Gender
There is no FDA guidance on the use of Esomeprazole (oral) with respect to specific gender populations.
Race
There is no FDA guidance on the use of Esomeprazole (oral) with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Esomeprazole (oral) in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Esomeprazole (oral) in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Esomeprazole (oral) in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Esomeprazole (oral) in patients who are immunocompromised.
Administration and Monitoring
Administration
Monitoring
There is limited information regarding Monitoring of Esomeprazole (oral) in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Esomeprazole (oral) in the drug label.
Overdosage
- A single oral dose of esomeprazole at 510 mg/kg (about 124 times the human dose on a body surface area basis), was lethal to rats. The major signs of acute toxicity were reduced motor activity, changes in respiratory frequency, tremor, ataxia, and intermittent clonic convulsions.
- The symptoms described in connection with deliberate NEXIUM overdose (limited experience of doses in excess of 240 mg/day) are transient. Single doses of 80 mg of esomeprazole were uneventful. Reports of overdosage with omeprazole in humans may also be relevant. Doses ranged up to 2,400 mg (120 times the usual recommended clinical dose). Manifestations were variable, but included confusion, drowsiness, blurred vision, tachycardia, nausea, diaphoresis, flushing, headache, dry mouth, and other adverse reactions similar to those seen in normal clinical experience (see omeprazole package insert - ADVERSE REACTIONS). No specific antidote for esomeprazole is known. Since esomeprazole is extensively protein bound, it is not expected to be removed by dialysis. In the event of overdosage, treatment should be symptomatic and supportive.
Pharmacology
Esomeprazole (oral)
| |
| Systematic (IUPAC) name | |
| ? | |
| Identifiers | |
| CAS number | ? |
| ATC code | ? |
| PubChem | ? |
| Chemical data | |
| Formula | ? |
| Mol. mass | ? |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | ? |
Mechanism of Action
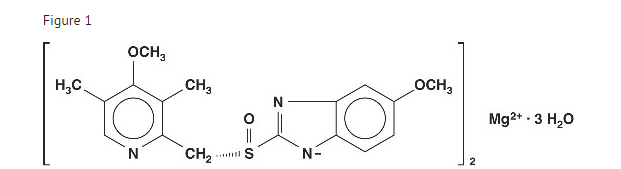
Structure
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Esomeprazole (oral) in the drug label.
Pharmacokinetics
There is limited information regarding Pharmacokinetics of Esomeprazole (oral) in the drug label.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Esomeprazole (oral) in the drug label.
Clinical Studies
How Supplied
- NEXIUM Delayed-Release Capsules, 20 mg, are opaque, hard gelatin, amethyst colored capsules with two radial bars in yellow on the cap and NEXIUM 20 mg in yellow on the body. They are supplied as follows:
- NDC 0186-5020-31 unit of use bottles of 30
- NDC 0186-5022-28 unit dose packages of 100
- NDC 0186-5020-54 bottles of 90
- NDC 0186-5020-82 bottles of 1000
- NEXIUM Delayed-Release Capsules, 40 mg, are opaque, hard gelatin, amethyst colored capsules with three radial bars in yellow on the cap and NEXIUM 40 mg in yellow on the body. They are supplied as follows:
- NDC 0186-5040-31 unit of use bottles of 30
- NDC 0186-5042-28 unit dose packages of 100
- NDC 0186-5040-54 bottles of 90
- NDC 0186-5040-82 bottles of 1000
- NDC 0186-5040-35 unit of use bottles of 30
- NDC 0186-5042-25 unit dose packages of 100
- NDC 0186-5040-55 bottles of 90
- NDC 0186-5040-85 bottles of 1000
- NEXIUM For Delayed-Release Oral Suspension is supplied as a unit dose packet containing a fine yellow powder, consisting of white to pale brownish esomeprazole granules and pale yellow inactive granules. NEXIUM unit dose packets are supplied as follows:
- NDC 0186-4025-01 unit dose packages of 30: 2.5 mg packets
- NDC 0186-4050-01 unit dose packages of 30: 5 mg packets
- NDC 0186-4010–01 unit dose packages of 30: 10 mg packets
- NDC 0186-4020–01 unit dose packages of 30: 20 mg packets
- NDC 0186-4040–01 unit dose packages of 30: 40 mg packets.
Storage
- Store at 25°C (77°F); excursions permitted to 15 to 30°C (59 to 86°F). Keep NEXIUM Delayed-Release Capsules container tightly closed. Dispense in a tight container if the NEXIUM Delayed-Release Capsules product package is subdivided.
Images
Drug Images
{{#ask: Page Name::Esomeprazole (oral) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel










{{#ask: Label Page::Esomeprazole (oral) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Advise patients to let you know if they are taking, or begin taking, other medications, because NEXIUM can interfere with antiretroviral drugs and drugs that are affected by gastric pH changes.
- Let patients know that antacids may be used while taking NEXIUM.
- Advise patients to take NEXIUM at least one hour before a meal.
- For patients who are prescribed NEXIUM Delayed-Release Capsules, advise them not to chew or crush the capsules.
- Advise patients that, if they open NEXIUM Delayed-Release Capsules to mix the granules with food, the granules should only be mixed with applesauce. Use with other foods has not been evaluated and is not recommended.
- For patients who are advised to open the NEXIUM Delayed-Release Capsules before taking them or who are prescribed NEXIUM For Delayed-Release Oral Suspension, instruct them in the proper technique for administration and tell them to follow the dosing instructions in the PATIENT INFORMATION insert included in the package. Instruct patients to rinse the syringe with water after each use.
- For patients who are prescribed NEXIUM for Delayed-Release Oral Suspension and need to use more than one packet for their dose, instruct them regarding the correct amount of water to use when mixing their dose.
Precautions with Alcohol
- Alcohol-Esomeprazole (oral) interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- NEXIUM®[1]
Look-Alike Drug Names
There is limited information regarding Esomeprazole (oral) Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Page Name=Esomeprazole (oral)
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Esomeprazole (oral) |Label Name=Esomeprazole (oral)11.png
}}
{{#subobject:
|Label Page=Esomeprazole (oral) |Label Name=Esomeprazole (oral)11.png
}}