Epiglottitis pathophysiology
|
Epiglottitis Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Epiglottitis pathophysiology On the Web |
|
American Roentgen Ray Society Images of Epiglottitis pathophysiology |
|
Risk calculators and risk factors for Epiglottitis pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1] Associate Editor(s)-in-Chief: Prince Tano Djan, BSc, MBChB [2]
Overview
Pathophysiology
Pathogenesis
Infectious epiglottitis
Infectious epiglottitis is a soft tissue swelling of epiglottis,[1] and the surrounding structures example; plica aryepiglottica , arytenoids, sinus piriformis and vestibular folds.[2] Understading the pathogenesis of epiglottitis involves a good knowlegde of the causative organisms. The only known reservoirs for H. influenzae in humans include:[3]
- Respiratory tract
- Conjunctival and
- Genital surfaces.
Healthy people are normal carriers of the organism. Pathogenicity of H. influenza is as a result of imbalance between the virulent factors of the organism and the host immune system.
This immunity is enhanced when children are vaccinated with the purified polyribosylribitol phosphate (PRP). H. influenza type b capsule is antiphagocytic. Serum anit-purified polyribosylribitol phosphate (anti-PRP) antibody is important in the complement dependent phagocytosis and lyses of the bacteria. [4] IgA antibody accords the mucosa surface protection again attachement of the organism.
The strategies deployed by a microbe to assist its survival and proliferation, may or may not lead to disease process. Epiglottitis caused by H. influenza may therefore be considered as an accidental consequence of the microbial factors that permit its survival.[5]
Acute epiglottis pathogenesis is well exemplified by H. influenzae, with the ability to colonize mucosal surfaces and to spread contiguously or invade epithelial cells, to disseminate within the bloodstream, and to localize to selected tissues among these is the epiglottis. The H. influenza like other infectious agents cause epiglottis mostly via invasion of the bloodstream by the help of its virulence factors. These include:[6]
- Capsular polysaccharide
- Lipopolysaccharide
- Outer-membrane proteins
- Pilus proteins
- IgA proteases
- Histamine
- Factors affecting cilial
The tissues surrounding the epiglottis have rich blood and lymphatic supply. Microbial invasion of the bloodstream around the epiglottis lead to inflammatory response and tissue edema most apparent at the lingual surface of the epiglottis compared to the laryngeal surface. The extravasation of fluid leads to remarkable tissue swelling that may lead to respiratory obstruction and the other symptoms of epiglottitis.[7]
Necrotizing epiglotitis
The pathogeneiis of necrotizing epiglottitis involves the infection with CMV or EBV usually in immunocompromised people. Affected patients are usually neutropenic and lymphopenic at presentation. CMV and EBV modulate the host's immune defense facilitating immune evasion and thereby predisposing the patient to a superimposed infections. The causative organism of necrotizing epiglottitis is unclear.[8]
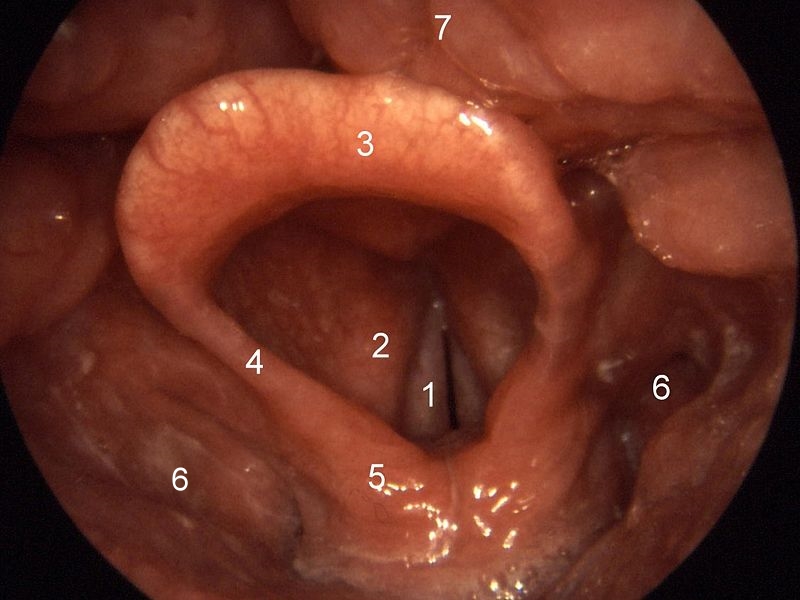
Gross pathology
On gross pathology the epiglottis and the surrounding structures appear erythematous and swollen as shown below.[9][10][2]
Microscopic pathology
The potential space between the mucosal epithelium of the epiglottis and the epiglottic cartilage consists of adipose tissue, fibrous tissue and glands. This space becomes occupied with extravasated fluid resulting from the inflammatory process. Nuetrophilic chemotaxis as well as other inflammatory molecules are responsible for the tissue response to the infectious agents.
References
- ↑ Shah RK, Stocks C (2010). "Epiglottitis in the United States: national trends, variances, prognosis, and management". Laryngoscope. 120 (6): 1256–62. doi:10.1002/lary.20921. PMID 20513048.
- ↑ 2.0 2.1 Ossoff RH, Wolff AP, Ballenger JJ (1980). "Acute epiglottitis in adults: experience with fifteen cases". Laryngoscope. 90 (7 Pt 1): 1155–61. PMID 6967138.
- ↑ Moxon ER, Wilson R (1991). "The role of Haemophilus influenzae in the pathogenesis of pneumonia". Rev Infect Dis. 13 Suppl 6: S518–27. PMID 1862281.
- ↑ Robbins JBH. influenza type b disease and immunity in humans, Ann intern medicine78:259, 1973
- ↑ Moxon ER, Wilson R (1991). "The role of Haemophilus influenzae in the pathogenesis of pneumonia". Rev Infect Dis. 13 Suppl 6: S518–27. PMID 1862281.
- ↑ Moxon ER, Wilson R (1991). "The role of Haemophilus influenzae in the pathogenesis of pneumonia". Rev Infect Dis. 13 Suppl 6: S518–27. PMID 1862281.
- ↑ Sato S, Kuratomi Y, Inokuchi A (2012). "Pathological characteristics of the epiglottis relevant to acute epiglottitis". Auris Nasus Larynx. 39 (5): 507–11. doi:10.1016/j.anl.2011.10.015. PMID 22113103.
- ↑ Tebruegge M, Connell T, Kong K, Marks M, Curtis N (2009). "Necrotizing epiglottitis in an infant: an unusual first presentation of human immunodeficiency virus infection". Pediatr Infect Dis J. 28 (2): 164–6. doi:10.1097/INF.0b013e318187a869. PMID 19106777.
- ↑ Charles R, Fadden M, Brook J (2013). "Acute epiglottitis". BMJ. 347: f5235. doi:10.1136/bmj.f5235. PMID 24052580.
- ↑ Mayo-Smith MF, Spinale JW, Donskey CJ, Yukawa M, Li RH, Schiffman FJ (1995). "Acute epiglottitis. An 18-year experience in Rhode Island". Chest. 108 (6): 1640–7. PMID 7497775.