Chlamydia infection
Template:DiseaseDisorder infobox
|
Chlamydia infection Microchapters |
|
Diagnosis |
|
Treatment |
|
Case Studies |
|
Chlamydia infection On the Web |
|
American Roentgen Ray Society Images of Chlamydia infection |
For patient information click here
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Detection
The diagnosis of genital chlamydial infections evolved rapidly from the 1990s through 2006. Nucleic acid amplification tests (NAAT), such as polymerase chain reaction (PCR), transcription mediated amplification (TMA), and the DNA strand displacement assay (SDA) now are the mainstays. As of January 2007, the most commonly used and widely studied chlamydia NAATs in the US and many other industrialized countries are Aptima (Gen-Probe), Probe-Tec (Becton-Dickinson), and Amplicor (Roche). The Aptima Combo II assay tests simltaneously for C. trachomatis and Neisseria gonorrhoeae, the cause of gonorrhea. NAAT for chlamydia may be performed on swab specimens collected from the cervix (women) or urethra (men), on self-collected vaginal swabs, or on voided urine. Urine and self-collected swab testing facilitates the performance of screening tests in settings where genital examination is impractical. At present, the NAATs have regulatory approval only for testing urogenital specimens, although rapidly evolving research indicates that the Aptima test may give reliable results on rectal specimens. Because of improved test accuracy, ease of specimen management, convenience in specimen management, and ease of screening sexually active men and women, the NAATs have largely replaced culture, the historic gold standard for chlamydia diagnosis, and the non-amplified probe tests, such as Pace II (Gen-Probe). The latter test is relatively insensitive, successfully detecting only 60-80% of infections in asymptomatic women, and often giving falsely positive results. Culture remains useful in selected circumstances and is currently the only assay approved for testing non-genital specimens.
Treatment
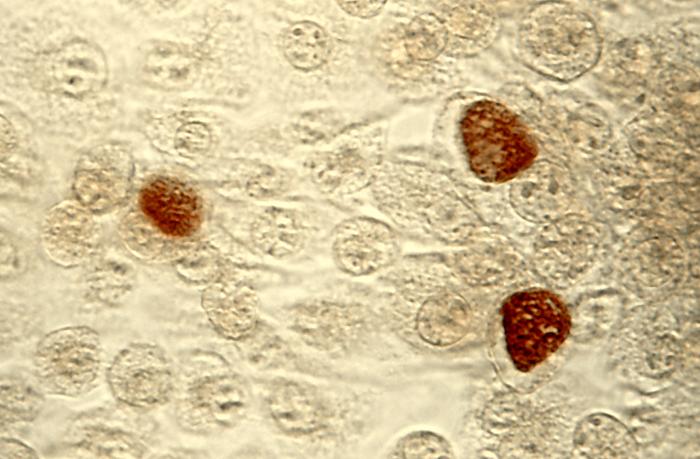
C. trachomatis infection can be effectively cured with antibiotics once it is detected. Current Centers for Disease Control guidelines provide for the following treatments:
- Azithromycin 1 gram oral as a single dose, or
- Doxycycline 100 milligrams twice daily for seven days.
- Tetracycline
- Erythromycin
Untested Treatments
- Ciprofloxacin 500 milligrams twice daily for 3 days. (Although this is not an approved method of treatment, as it is shown to be ineffective and may simply delay symptoms.)
β-lactams are not suitable drugs for the treatment of chlamydia. While they have the ability to halt growth of the organism (i.e. are microbistatic), these antibiotics do not eliminate the bacteria. Once treatment is stopped, the bacteria will begin to grow once more. (See below for Persistence.)
Pathophysiology
Chlamydial life cycle
Chlamydiae are obligate intracellular bacterial pathogens, which means they are unable to replicate outside of a host cell. However, to disseminate effectively, these pathogens have evolved a unique biphasic life cycle wherein they alternate between two functionally and morphologically distinct forms. [1]
- The elementary body (EB) is infectious, but metabolically inert (much like a spore), and can survive for limited amounts of time in the extracellular milieu. Once the EB attaches to a susceptible host cell, it mediates its own internalization through pathogen-specified mechanisms (via type III secretion system) that allows for the recruitment of actin with subsequent engulfment of the bacterium.
- The internalized EB, within a membrane-bound compartment, immediately begins differentiation into the reticulate body (RB). RBs are metabolically active but non-infectious, and in many regards, resemble normal replicating bacteria. The intracellular bacteria rapidly modifies its membrane-bound compartment into the so-called chlamydial inclusion so as to prevent phagosome-lysosome fusion. According to published data, the inclusion has no interactions with the endocytic pathway and apparently inserts itself into the exocytic pathway as it retains the ability to intercept sphingomyelin-containing vesicles.
To date, no one has been able to detect a host cell protein that is trafficked to the inclusion through the exocytic pathway. As the RBs replicate, the inclusion grows as well to accommodate the increasing numbers of organisms. Through unknown mechanisms, RBs begin a differentiation program back to the infectious EBs, which are released from the host cell to initiate a new round of infection. Because of their obligate intracellular nature, Chlamydiae have no tractable genetic system, unlike E. coli, which makes Chlamydiae and related organisms difficult to investigate.
Persistence
Chlamydiae have the ability to establish long-term associations with host cells. When an infected host cell is starved for various nutrients such as amino acids (e.g. tryptophan),[2] iron, or vitamins, this has a negative consequence for Chlamydiae since the organism is dependent on the host cell for these nutrients. The starved chlamydiae enter a persistent growth state wherein they stop cell division and become morphologically aberrant by increasing in size.[3] Persistent organisms remain viable as they are capable of returning to a normal growth state once conditions in the host cell improve. There is much debate as to whether persistence has in vivo relevance. Many believe that persistent chlamydiae are the cause of chronic chlamydial diseases. Some antibiotics such as β-lactams can also induce a persistent-like growth state, which can contribute to the chronicity of chlamydial diseases.
Diseases caused by Chlamydia trachomatis
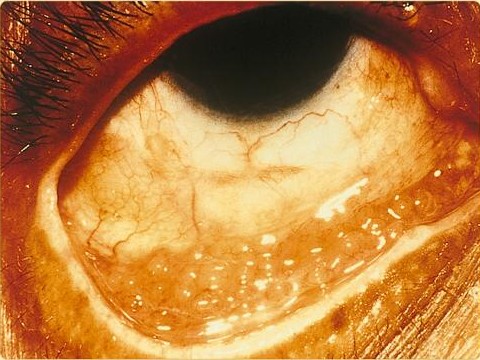
Chlamydia trachomatis can cause the following conditions:
Recent genetic discoveries
Recent phylogenetic studies have revealed that chlamydia shares a common ancestor with modern plants, and retains unusual plant-like traits (both genetically and physiologically). In particular, the enzyme L,L-diaminopimelate aminotransferase, which is related to lysine production in plants, is also linked with the construction of chlamydia's cell wall. The genetic encoding for the enzymes is remarkably similar in plants and chlamydia, demonstrating a close common ancestry.[4] This unexpected discovery may help scientists develop new treatment avenues: if scientists could find a safe and effective inhibitor of L,L-diaminopimelate aminotransferase, they might have a highly effective and extremely specific new antibiotic against chlamydia.
References
- ↑ "Structure". Retrieved 2007-10-26.
- ↑ Leonhardt RM, Lee SJ, Kavathas PB, Cresswell P (2007). "Severe Tryptophan Starvation Blocks Onset of Conventional Persistence and Reduces Reactivation of Chlamydia trachomatis". Infect. Immun. 75 (11): 5105–17. doi:10.1128/IAI.00668-07. PMID 17724071.
- ↑ Mpiga P, Ravaoarinoro M (2006). "Chlamydia trachomatis persistence: an update". Microbiol. Res. 161 (1): 9–19. doi:10.1016/j.micres.2005.04.004. PMID 16338585.
- ↑ McCoy AJ, Adams NE, Hudson AO, Gilvarg C, Leustek T, Maurelli AT (2006). "L,L-diaminopimelate aminotransferase, a trans-kingdom enzyme shared by Chlamydia and plants for synthesis of diaminopimelate/lysine". Proc. Natl. Acad. Sci. U.S.A. 103 (47): 17909–14. doi:10.1073/pnas.0608643103. PMID 17093042.
External links
| Wikimedia Commons has media related to Chlamydia. |
- "Chlamydia: Questions and Answers" from Planned Parenthood
- NetDoctor Chlamydia fact sheet
- Links to chlamydia pictures at University of Iowa
- Chlamydiae.com - Information for patients, as well as for doctors and researchers about Chlamydial infections. Note: Patient info is in multiple languages.
Template:STD/STI Template:Bacterial diseases
ar:كلاميديا da:Klamydia de:Chlamydien it:Infezioni da clamidia he:כלמידיה mk:Хламидија ms:Penyakit kelamin Chlamydia nl:Chlamydia no:Klamydia-infeksjon sl:Klamidioza fi:Klamydia sv:Klamydia ta:கிளமிடியா