Aripiprazole (intramuscular): Difference between revisions
Kiran Singh (talk | contribs) No edit summary |
Kiran Singh (talk | contribs) No edit summary |
||
| Line 271: | Line 271: | ||
(Description regarding monitoring, from ''Warnings'' section) | (Description regarding monitoring, from ''Warnings'' section) | ||
|IVCompat | |IVCompat= | ||
There is limited information regarding IV Compatibility of Aripiprazole in the drug label. | |||
|overdose===== OVERDOSAGE==== | |overdose===== OVERDOSAGE==== | ||
*MedDRA terminology has been used to classify the adverse reactions. | *MedDRA terminology has been used to classify the adverse reactions. | ||
Revision as of 13:35, 28 July 2014
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Kiran Singh, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
WARNINGS: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS and SUICIDALITY AND ANTIDEPRESSANT DRUGS
See full prescribing information for complete Boxed Warning.
* Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of seventeen placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (eg, heart failure, sudden death) or infectious (eg, pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. ABILIFY (aripiprazole) is not approved for the treatment of patients with dementia-related psychosis.
|
Overview
Aripiprazole (intramuscular) is an atypical antipsychotic that is FDA approved for the treatment of schizophrenia, bipolar I disorder, major depressive disorder, and irritability associated with autistic disorder. There is a Black Box Warning for this drug as shown here. Common adverse reactions include nausea, vomiting, constipation, headache, dizziness, akathisia, anxiety, insomnia, and restlessness.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Schizophrenia
- Dosing Information
- The recommended starting and target dose for ABILIFY is 10 mg/day or 15 mg/day administered on a once-a-day schedule without regard to meals.
- Maintenance Treatment: Maintenance of efficacy in schizophrenia was demonstrated in a trial involving patients with schizophrenia who had been symptomatically stable on other antipsychotic medications for periods of 3 months or longer. These patients were discontinued from those medications and randomized to either ABILIFY 15 mg/day or placebo, and observed for relapse. Patients should be periodically reassessed to determine the continued need for maintenance treatment.
Bipolar I Disorder
- Dosing Information
- (Dosage)
Adjunctive Treatment of Major Depressive Disorder
- Dosing Information
- (Dosage)
Irritability Associated with Autistic Disorder
- Dosing Information
- (Dosage)
Agitation Associated with Schizophrenia or Bipolar Mania (Intramuscular Injection)
- Dosing Information
- (Dosage)
Dosage Adjustment
Dosage adjustments in adults are not routinely indicated on the basis of age, gender, race, or renal or hepatic impairment status.
- Dosage adjustment for patients taking aripiprazole concomitantly with strong CYP3A4 inhibitors: When concomitant administration of aripiprazole with strong CYP3A4 inhibitors such as ketoconazole or clarithromycin is indicated, the aripiprazole dose should be reduced to one-half of the usual dose. When the CYP3A4 inhibitor is withdrawn from the combination therapy, the aripiprazole dose should then be increased.
- Dosage adjustment for patients taking aripiprazole concomitantly with potential CYP2D6 inhibitors: When concomitant administration of potential CYP2D6 inhibitors such as quinidine, fluoxetine, or paroxetine with aripiprazole occurs, aripiprazole dose should be reduced at least to one-half of its normal dose. When the CYP2D6 inhibitor is withdrawn from the combination therapy, the aripiprazole dose should then be increased. When adjunctive ABILIFY is administered to patients with major depressive disorder, ABILIFY should be administered without dosage adjustment.
- Dosing recommendation in patients taking aripiprazole concomitantly with strong CYP3A4 and CYP2D6 inhibitors: When concomitant administration of aripiprazole with strong inhibitors of CYP3A4 (such as ketoconazole or clarithromycin) and CYP2D6 (such as quinidine, fluoxetine, or paroxetine) is indicated, the aripiprazole dose should be reduced to one-quarter (25%) of the usual dose. When the CYP3A4 and/or CYP2D6 inhibitor is withdrawn from the combination therapy, the aripiprazole dose should be increased.
- Dosing recommendation in patients taking aripiprazole concomitantly with strong, moderate, or weak inhibitors of CYP3A4 and CYP2D6: Patients who may be receiving a combination of strong, moderate, and weak inhibitors of CYP3A4 and CYP2D6 (eg, a potent CYP3A4 inhibitor and a moderate CYP2D6 inhibitor or a moderate CYP3A4 inhibitor with a moderate CYP2D6 inhibitor), the dosing may be reduced to one-quarter (25%) of the usual dose initially and then adjusted to achieve a favorable clinical response.
- Dosing recommendation in patients who are classified as CYP2D6 poor metabolizers (PM): The aripiprazole dose in PM patients should initially be reduced to one-half (50%) of the usual dose and then adjusted to achieve a favorable clinical response. The dose of aripiprazole for PM patients who are administered a strong CYP3A4 inhibitor should be reduced to one-quarter (25%) of the usual dose.
- Dosage adjustment for patients taking potential CYP3A4 inducers: When a potential CYP3A4 inducer such as carbamazepine is added to aripiprazole therapy, the aripiprazole dose should be doubled. Additional dose increases should be based on clinical evaluation. When the CYP3A4 inducer is withdrawn from the combination therapy, the aripiprazole dose should be reduced to 10 mg to 15 mg.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Aripiprazole in adult patients.
Non–Guideline-Supported Use
Borderline personality disorder
- Dosing Information
- Aripiprazole 10-15 mg/day added to sertraline 100-200 mg/day[1]
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Condition 1
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
Condition 1
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Condition 2
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Non–Guideline-Supported Use
Condition 1
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Condition 3
- Dosing Information
- (Dosage)
Contraindications
- Known hypersensitivity reaction to ABILIFY. Reactions have ranged from pruritus/urticaria to anaphylaxis.
Warnings
|
WARNINGS: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS and SUICIDALITY AND ANTIDEPRESSANT DRUGS
See full prescribing information for complete Boxed Warning.
* Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of seventeen placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (eg, heart failure, sudden death) or infectious (eg, pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. ABILIFY (aripiprazole) is not approved for the treatment of patients with dementia-related psychosis.
|
Use in Elderly Patients with Dementia-Related Psychosis
Increased Mortality
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. ABILIFY (aripiprazole) is not approved for the treatment of patients with dementia-related psychosis
Cerebrovascular Adverse Events, Including Stroke
In placebo-controlled clinical studies (two flexible dose and one fixed dose study) of dementia-related psychosis, there was an increased incidence of cerebrovascular adverse events (eg, stroke, transient ischemic attack), including fatalities, in aripiprazole-treated patients (mean age: 84 years; range: 78-88 years). In the fixed-dose study, there was a statistically significant dose response relationship for cerebrovascular adverse events in patients treated with aripiprazole. Aripiprazole is not approved for the treatment of patients with dementia-related psychosis.
Safety Experience in Elderly Patients with Psychosis Associated with Alzheimer's Disease
- In three, 10-week, placebo-controlled studies of aripiprazole in elderly patients with psychosis associated with Alzheimer's disease (n=938; mean age: 82.4 years; range: 56-99 years), the treatment-emergent adverse events that were reported at an incidence of ≥3% and aripiprazole incidence at least twice that for placebo were lethargy [placebo 2%, aripiprazole 5%], somnolence (including sedation) [placebo 3%, aripiprazole 8%], and incontinence (primarily, urinary incontinence) [placebo 1%, aripiprazole 5%], excessive salivation [placebo 0%, aripiprazole 4%], and lightheadedness [placebo 1%, aripiprazole 4%].
- The safety and efficacy of ABILIFY in the treatment of patients with psychosis associated with dementia have not been established. If the prescriber elects to treat such patients with ABILIFY, vigilance should be exercised, particularly for the emergence of difficulty swallowing or excessive somnolence, which could predispose to accidental injury or aspiration.
Clinical Worsening of Depression and Suicide Risk
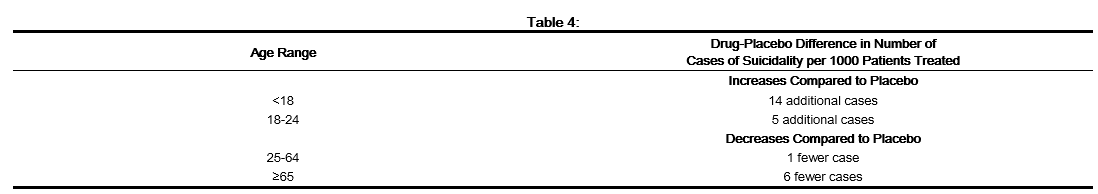
- Patients with major depressive disorder (MDD), both adult and pediatric, may experience worsening of their depression and/or the emergence of suicidal ideation and behavior (suicidality) or unusual changes in behavior, whether or not they are taking antidepressant medications, and this risk may persist until significant remission occurs. Suicide is a known risk of depression and certain other psychiatric disorders, and these disorders themselves are the strongest predictors of suicide. There has been a long-standing concern, however, that antidepressants may have a role in inducing worsening of depression and the emergence of suicidality in certain patients during the early phases of treatment. Pooled analyses of short-term, placebo-controlled trials of antidepressant drugs (SSRIs and others) showed that these drugs increase the risk of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults (ages 18-24) with MDD and other psychiatric disorders. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction with antidepressants compared to placebo in adults aged 65 and older.
- The pooled analyses of placebo-controlled trials in children and adolescents with MDD, Obsessive Compulsive Disorder (OCD), or other psychiatric disorders included a total of 24 short-term trials of 9 antidepressant drugs in over 4400 patients. The pooled analyses of placebo-controlled trials in adults with MDD or other psychiatric disorders included a total of 295 short-term trials (median duration of 2 months) of 11 antidepressant drugs in over 77,000 patients. There was considerable variation in risk of suicidality among drugs, but a tendency toward an increase in the younger patients for almost all drugs studied. There were differences in absolute risk of suicidality across the different indications, with the highest incidence in MDD. The risk differences (drug vs. placebo), however, were relatively stable within age strata and across indications. These risk differences (drug-placebo difference in the number of cases of suicidality per 1000 patients treated) are provided in Table 4.
Adverse Reactions
Clinical Trials Experience
Central Nervous System
- (list/description of adverse reactions)
Cardiovascular
- (list/description of adverse reactions)
Respiratory
- (list/description of adverse reactions)
Gastrointestinal
- (list/description of adverse reactions)
Hypersensitive Reactions
- (list/description of adverse reactions)
Miscellaneous
- (list/description of adverse reactions)
Condition 2
Central Nervous System
- (list/description of adverse reactions)
Cardiovascular
- (list/description of adverse reactions)
Respiratory
- (list/description of adverse reactions)
Gastrointestinal
- (list/description of adverse reactions)
Hypersensitive Reactions
- (list/description of adverse reactions)
Miscellaneous
- (list/description of adverse reactions)
Postmarketing Experience
(Description)
Drug Interactions
- Given the primary CNS effects of aripiprazole, caution should be used when ABILIFY is taken in combination with other centrally-acting drugs or alcohol.
- Due to its alpha adrenergic antagonism, aripiprazole has the potential to enhance the effect of certain antihypertensive agents.
Potential for Other Drugs to Affect ABILIFY
- Aripiprazole is not a substrate of CYP1A1, CYP1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C9, CYP2C19, or CYP2E1 enzymes. Aripiprazole also does not undergo direct glucuronidation. This suggests that an interaction of aripiprazole with inhibitors or inducers of these enzymes, or other factors, like smoking, is unlikely.
- Both CYP3A4 and CYP2D6 are responsible for aripiprazole metabolism. Agents that induce CYP3A4 (eg, carbamazepine) could cause an increase in aripiprazole clearance and lower blood levels. Inhibitors of CYP3A4 (eg, ketoconazole) or CYP2D6 (eg, quinidine, fluoxetine, or paroxetine) can inhibit aripiprazole elimination and cause increased blood level.
Ketoconazole and Other CYP3A4 Inhibitors
- Coadministration of ketoconazole (200 mg/day for 14 days) with a 15 mg single dose of aripiprazole increased the AUC of aripiprazole and its active metabolite by 63% and 77%, respectively. The effect of a higher ketoconazole dose (400 mg/day) has not been studied. When ketoconazole is given concomitantly with aripiprazole, the aripiprazole dose should be reduced to one-half of its normal dose.
- Other strong inhibitors of CYP3A4 (itraconazole) would be expected to have similar effects and need similar dose reductions; moderate inhibitors (erythromycin, grapefruit juice) have not been studied. When the CYP3A4 inhibitor is withdrawn from the combination therapy, the aripiprazole dose should be increased.
Quinidine and Other CYP2D6 Inhibitors
- Coadministration of a 10 mg single dose of aripiprazole with quinidine (166 mg/day for 13 days), a potent inhibitor of CYP2D6, increased the AUC of aripiprazole by 112% but decreased the AUC of its active metabolite, dehydro-aripiprazole, by 35%. Aripiprazole dose should be reduced to one-half of its normal dose when quinidine is given concomitantly with aripiprazole.
- Other significant inhibitors of CYP2D6, such as fluoxetine or paroxetine, would be expected to have similar effects and should lead to similar dose reductions. When the CYP2D6 inhibitor is withdrawn from the combination therapy, the aripiprazole dose should be increased. When adjunctive ABILIFY is administered to patients with major depressive disorder, ABILIFY should be administered without dosage adjustment.
Carbamazepine and Other CYP3A4 Inducers
- Coadministration of carbamazepine (200 mg twice daily), a potent CYP3A4 inducer, with aripiprazole (30 mg/day) resulted in an approximate 70% decrease in Cmax and AUC values of both aripiprazole and its active metabolite, dehydro-aripiprazole. When carbamazepine is added to aripiprazole therapy, aripiprazole dose should be doubled. Additional dose increases should be based on clinical evaluation. When carbamazepine is withdrawn from the combination therapy, the aripiprazole dose should be reduced
Potential for ABILIFY to Affect Other Drugs
- Aripiprazole is unlikely to cause clinically important pharmacokinetic interactions with drugs metabolized by cytochrome P450 enzymes. In in vivo studies, 10 mg/day to 30 mg/day doses of aripiprazole had no significant effect on metabolism by CYP2D6 (dextromethorphan), CYP2C9 (warfarin), CYP2C19 (omeprazole, warfarin), and CYP3A4 (dextromethorphan) substrates. Additionally, aripiprazole and dehydro-aripiprazole did not show potential for altering CYP1A2-mediated metabolism in vitro.
- No effect of aripiprazole was seen on the pharmacokinetics of lithium or valproate.
Drugs Having No Clinically Important Interactions with ABILIFY
Famotidine
- Coadministration of aripiprazole (given in a single dose of 15 mg) with a 40 mg single dose of the H2 antagonist famotidine, a potent gastric acid blocker, decreased the solubility of aripiprazole and, hence, its rate of absorption, reducing by 37% and 21% the Cmax of aripiprazole and dehydro-aripiprazole, respectively, and by 13% and 15%, respectively, the extent of absorption (AUC). No dosage adjustment of aripiprazole is required when administered concomitantly with famotidine.
Valproate
- When valproate (500 mg/day-1500 mg/day) and aripiprazole (30 mg/day) were coadministered, at steady-state the Cmax and AUC of aripiprazole were decreased by 25%. No dosage adjustment of aripiprazole is required when administered concomitantly with valproate.
- When aripiprazole (30 mg/day) and valproate (1000 mg/day) were coadministered, at steady-state there were no clinically significant changes in the Cmax or AUC of valproate. No dosage adjustment of valproate is required when administered concomitantly with aripiprazole.
Lithium
- A pharmacokinetic interaction of aripiprazole with lithium is unlikely because lithium is not bound to plasma proteins, is not metabolized, and is almost entirely excreted unchanged in urine. Coadministration of therapeutic doses of lithium (1200 mg/day-1800 mg/day) for 21 days with aripiprazole (30 mg/day) did not result in clinically significant changes in the pharmacokinetics of aripiprazole or its active metabolite, dehydro-aripiprazole (Cmax and AUC increased by less than 20%). No dosage adjustment of aripiprazole is required when administered concomitantly with lithium.
- Coadministration of aripiprazole (30 mg/day) with lithium (900 mg/day) did not result in clinically significant changes in the pharmacokinetics of lithium. No dosage adjustment of lithium is required when administered concomitantly with aripiprazole.
Lamotrigine
- Coadministration of 10 mg/day to 30 mg/day oral doses of aripiprazole for 14 days to patients with bipolar I disorder had no effect on the steady-state pharmacokinetics of 100 mg/day to 400 mg/day lamotrigine, a UDP-glucuronosyltransferase 1A4 substrate. No dosage adjustment of lamotrigine is required when aripiprazole is added to lamotrigine.
Dextromethorphan
- Aripiprazole at doses of 10 mg/day to 30 mg/day for 14 days had no effect on dextromethorphan's O-dealkylation to its major metabolite, dextrorphan, a pathway dependent on CYP2D6 activity. Aripiprazole also had no effect on dextromethorphan's N-demethylation to its metabolite 3-methoxymorphinan, a pathway dependent on CYP3A4 activity. No dosage adjustment of dextromethorphan is required when administered concomitantly with aripiprazole.
Warfarin
- Aripiprazole 10 mg/day for 14 days had no effect on the pharmacokinetics of R-warfarin and S-warfarin or on the pharmacodynamic end point of International Normalized Ratio, indicating the lack of a clinically relevant effect of aripiprazole on CYP2C9 and CYP2C19 metabolism or the binding of highly protein-bound warfarin. No dosage adjustment of warfarin is required when administered concomitantly with aripiprazole.
Omeprazole
- Aripiprazole 10 mg/day for 15 days had no effect on the pharmacokinetics of a single 20 mg dose of omeprazole, a CYP2C19 substrate, in healthy subjects. No dosage adjustment of omeprazole is required when administered concomitantly with aripiprazole.
Lorazepam
- Coadministration of lorazepam injection (2 mg) and aripiprazole injection (15 mg) to healthy subjects (n=40: 35 males and 5 females; ages 19-45 years old) did not result in clinically important changes in the pharmacokinetics of either drug. No dosage adjustment of aripiprazole is required when administered concomitantly with lorazepam. However, the intensity of sedation was greater with the combination as compared to that observed with aripiprazole alone and the orthostatic hypotension observed was greater with the combination as compared to that observed with lorazepam alone.
Escitalopram
- Coadministration of 10 mg/day oral doses of aripiprazole for 14 days to healthy subjects had no effect on the steady-state pharmacokinetics of 10 mg/day escitalopram, a substrate of CYP2C19 and CYP3A4. No dosage adjustment of escitalopram is required when aripiprazole is added to escitalopram.
Venlafaxine
- Coadministration of 10 mg/day to 20 mg/day oral doses of aripiprazole for 14 days to healthy subjects had no effect on the steady-state pharmacokinetics of venlafaxine and O-desmethylvenlafaxine following 75 mg/day venlafaxine XR, a CYP2D6 substrate. No dosage adjustment of venlafaxine is required when aripiprazole is added to venlafaxine.
Fluoxetine, Paroxetine, and Sertraline
- A population pharmacokinetic analysis in patients with major depressive disorder showed no substantial change in plasma concentrations of fluoxetine (20 mg/day or 40 mg/day), paroxetine CR (37.5 mg/day or 50 mg/day), or sertraline (100 mg/day or 150 mg/day) dosed to steady-state. The steady-state plasma concentrations of fluoxetine and norfluoxetine increased by about 18% and 36%, respectively, and concentrations of paroxetine decreased by about 27%. The steady-state plasma concentrations of sertraline and desmethylsertraline were not substantially changed when these antidepressant therapies were coadministered with aripiprazole. Aripiprazole dosing was 2 mg/day to 15 mg/day (when given with fluoxetine or paroxetine) or 2 mg/day to 20 mg/day (when given with sertraline).
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA):
There is no FDA guidance on usage of Aripiprazole (intramuscular) in women who are pregnant.
Pregnancy Category (AUS):
(Description)
Labor and Delivery
(Description)
Nursing Mothers
(Description)
Pediatric Use
(Description)
Geriatic Use
(Description)
Gender
(Description)
Race
(Description)
Renal Impairment
(Description)
Hepatic Impairment
(Description)
Females of Reproductive Potential and Males
(Description)
Immunocompromised Patients
(Description)
Others
(Description)
Administration and Monitoring
Administration
(Oral/Intravenous/etc)
Monitoring
Condition 1
(Description regarding monitoring, from Warnings section)
Condition 2
(Description regarding monitoring, from Warnings section)
Condition 3
(Description regarding monitoring, from Warnings section)
IV Compatibility
There is limited information regarding IV Compatibility of Aripiprazole in the drug label.
Overdosage
OVERDOSAGE
- MedDRA terminology has been used to classify the adverse reactions.
Human Experience
- In clinical trials and in postmarketing experience, adverse reactions of deliberate or accidental overdosage with oral aripiprazole have been reported worldwide. These include overdoses with oral aripiprazole alone and in combination with other substances. No fatality was reported with aripiprazole alone. The largest known dose with a known outcome involved acute ingestion of 1260 mg of oral aripiprazole (42 times the maximum recommended daily dose) by a patient who fully recovered. Deliberate or accidental overdosage was also reported in children (age 12 and younger) involving oral aripiprazole ingestions up to 195 mg with no fatalities.
- Common adverse reactions (reported in at least 5% of all overdose cases) reported with oral aripiprazole overdosage (alone or in combination with other substances) include vomiting, somnolence, and tremor. Other clinically important signs and symptoms observed in one or more patients with aripiprazole overdoses (alone or with other substances) include acidosis, aggression, aspartate aminotransferase increased, atrial fibrillation, bradycardia, coma, confusional state, convulsion, blood creatine phosphokinase increased, depressed level of consciousness, hypertension, hypokalemia, hypotension, lethargy, loss of consciousness, QRS complex prolonged, QT prolonged, pneumonia aspiration, respiratory arrest, status epilepticus, and tachycardia.
Management of Overdosage
- No specific information is available on the treatment of overdose with aripiprazole. An electrocardiogram should be obtained in case of overdosage and if QT interval prolongation is present, cardiac monitoring should be instituted. Otherwise, management of overdose should concentrate on supportive therapy, maintaining an adequate airway, oxygenation and ventilation, and management of symptoms. Close medical supervision and monitoring should continue until the patient recovers.
- Charcoal: In the event of an overdose of ABILIFY, an early charcoal administration may be useful in partially preventing the absorption of aripiprazole. Administration of 50 g of activated charcoal, one hour after a single 15 mg oral dose of aripiprazole, decreased the mean AUC and Cmax of aripiprazole by 50%.
- Hemodialysis: Although there is no information on the effect of hemodialysis in treating an overdose with aripiprazole, hemodialysis is unlikely to be useful in overdose management since aripiprazole is highly bound to plasma proteins.
)
Pharmacology
Mechanism of Action
(Description)
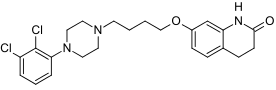
Structure
(Description with picture)
Pharmacodynamics
(Description)
Pharmacokinetics
(Description)
Nonclinical Toxicology
(Description)
Clinical Studies
Condition 1
(Description)
Condition 2
(Description)
Condition 3
(Description)
How Supplied
(Description)
Storage
There is limited information regarding Aripiprazole (intramuscular) Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Aripiprazole (intramuscular) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Aripiprazole (intramuscular) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
(Patient Counseling Information)
Precautions with Alcohol
Alcohol-Aripiprazole interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Aripiprazole (intramuscular) Brand Names in the drug label.
Look-Alike Drug Names
- (Paired Confused Name 1a) — (Paired Confused Name 1b)
- (Paired Confused Name 2a) — (Paired Confused Name 2b)
- (Paired Confused Name 3a) — (Paired Confused Name 3b)
Drug Shortage Status
Drug Shortage
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ Bellino S, Paradiso E, Bogetto F (2008). "Efficacy and tolerability of aripiprazole augmentation in sertraline-resistant patients with borderline personality disorder". Psychiatry Res. 161 (2): 206–12. doi:10.1016/j.psychres.2007.07.006. PMID 18848360.
- ↑ 2.0 2.1 2.2 2.3 2.4
- ↑ 3.0 3.1 3.2 3.3 3.4 "ABILIFY (aripiprazole) tablet ABILIFY (aripiprazole) solution ABILIFY DISCMELT (aripiprazole) tablet, orally disintegrating ABILIFY (aripiprazole) injection, solution [Otsuka America Pharmaceutical, Inc.]". DailyMed. Otsuka America Pharmaceutical, Inc. April 2013. Retrieved 22 October 2013.
- ↑ 4.0 4.1 4.2 4.3 4.4 "Abilify Tablets, Orodispersible Tablets, Oral Solution - Summary of Product Characteristics (SPC)". electronic Medicines Compendium. Otsuka Pharmaceuticals (UK) Ltd. 20 September 2013. Retrieved 22 October 2013.
- ↑ 5.0 5.1 5.2 5.3 5.4 "ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS" (PDF). European Medicines Agency. Otsuka Pharmaceutical Europe Ltd. Retrieved 22 October 2013.
{{#subobject:
|Page Name=Aripiprazole (intramuscular) |Pill Name=ABILIFY_NDC_591480008.jpg |Drug Name=ABILIFY |Pill Ingred=ARIPIPRAZOLE[ARIPIPRAZOLE]|+sep=; |Pill Imprint=A;008;10 |Pill Dosage=10 mg |Pill Color=Pink|+sep=; |Pill Shape= |Pill Size (mm)=8 |Pill Scoring=1 |Pill Image= |Drug Author=Otsuka America Pharmaceutical, Inc. |NDC=591480008
}}
{{#subobject:
|Page Name=Aripiprazole (intramuscular) |Pill Name=ABILIFY_NDC_591480006.jpg |Drug Name=ABILIFY |Pill Ingred=ARIPIPRAZOLE[ARIPIPRAZOLE]|+sep=; |Pill Imprint=A;006;2 |Pill Dosage=2 mg |Pill Color=Green|+sep=; |Pill Shape= |Pill Size (mm)=8 |Pill Scoring=1 |Pill Image= |Drug Author=Otsuka America Pharmaceutical, Inc. |NDC=591480006
}}
{{#subobject:
|Page Name=Aripiprazole (intramuscular) |Pill Name=ABILIFY_NDC_591480007.jpg |Drug Name=ABILIFY |Pill Ingred=ARIPIPRAZOLE[ARIPIPRAZOLE]|+sep=; |Pill Imprint=A;007;5 |Pill Dosage=5 mg |Pill Color=Blue|+sep=; |Pill Shape= |Pill Size (mm)=8 |Pill Scoring=1 |Pill Image= |Drug Author=Otsuka America Pharmaceutical, Inc. |NDC=591480007
}}
{{#subobject:
|Page Name=Aripiprazole (intramuscular) |Pill Name=ABILIFY_NDC_591480009.jpg |Drug Name=ABILIFY |Pill Ingred=ARIPIPRAZOLE[ARIPIPRAZOLE]|+sep=; |Pill Imprint=A;009;15 |Pill Dosage=15 mg |Pill Color=Yellow|+sep=; |Pill Shape=Round |Pill Size (mm)=6 |Pill Scoring=1 |Pill Image= |Drug Author=Otsuka America Pharmaceutical, Inc. |NDC=591480009
}}
{{#subobject:
|Page Name=Aripiprazole (intramuscular) |Pill Name=ABILIFY_NDC_591480010.jpg |Drug Name=ABILIFY |Pill Ingred=ARIPIPRAZOLE[ARIPIPRAZOLE]|+sep=; |Pill Imprint=A;010;20 |Pill Dosage=20 mg |Pill Color=White|+sep=; |Pill Shape=Round |Pill Size (mm)=8 |Pill Scoring=1 |Pill Image= |Drug Author=Otsuka America Pharmaceutical, Inc. |NDC=591480010
}}
{{#subobject:
|Page Name=Aripiprazole (intramuscular) |Pill Name=ABILIFY_NDC_591480011.jpg |Drug Name=ABILIFY |Pill Ingred=ARIPIPRAZOLE[ARIPIPRAZOLE]|+sep=; |Pill Imprint=A;011;30 |Pill Dosage=30 mg |Pill Color=Pink|+sep=; |Pill Shape=Round |Pill Size (mm)=9 |Pill Scoring=1 |Pill Image= |Drug Author=Otsuka America Pharmaceutical, Inc. |NDC=591480011
}}