Ticlopidine clinical studies
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Ahmed Zaghw, M.D. [2]
Clinical Trials
Stroke Patients
The effect of ticlopidine on the risk of stroke and cardiovascular events was studied in two multicenter, randomized, double-blind trials.
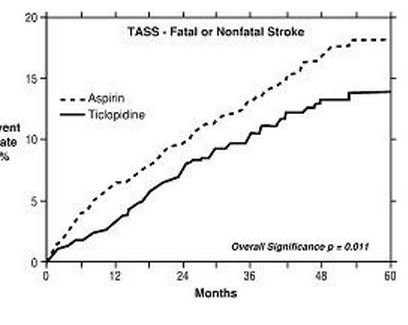
1. Study in Patients Experiencing Stroke Precursors: In a trial comparing ticlopidine and aspirin (The Ticlopidine Aspirin Stroke Study or TASS), 3069 patients (1987 men, 1082 women) who had experienced such stroke precursors as transient ischemic attack (TIA), transient monocular blindness (amaurosis fugax), reversible ischemic neurological deficit or minor stroke, were randomized to ticlopidine 250 mg bid or aspirin 650 mg bid. The study was designed to follow patients for at least 2 years and up to 5 years.
Over the duration of the study, ticlopidine hydrochloride significantly reduced the risk of fatal and nonfatal stroke by 24% (p = .011) from 18.1 to 13.8 per 100 patients followed for 5 years, compared to aspirin. During the first year, when the risk of stroke is greatest, the reduction in risk of stroke (fatal and nonfatal) compared to aspirin was 48%; the reduction was similar in men and women.
 |
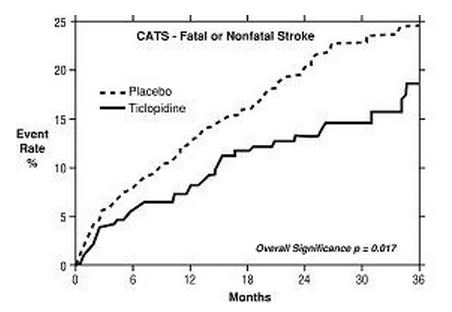
2. Study in Patients Who Had a Completed Atherothrombotic Stroke: In a trial comparing ticlopidine with placebo (The Canadian American Ticlopidine Study or CATS) 1073 patients who had experienced a previous atherothrombotic stroke were treated with ticlopidine hydrochloride 250 mg bid or placebo for up to 3 years.
Ticlopidine significantly reduced the overall risk of stroke by 24% (p = .017) from 24.6 to 18.6 per 100 patients followed for 3 years, compared to placebo. During the first year the reduction in risk of fatal and nonfatal stroke over placebo was 33%.
 |
Stent Patients
The ability of ticlopidine to reduce the rate of thrombotic events after the placement of coronary artery stents has been studied in five randomized trials, one of substantial size (Stent Anticoagulation Restenosis Study or STARS) described below, and four smaller studies. In these trials, ticlopidine 250 mg bid with ASA (dose range from 100 mg bid to 325 mg qd) was compared to aspirin alone or to anticoagulant therapy plus aspirin. The trials enrolled patients undergoing both planned (elective) and unplanned coronary stent placement. The types of stents used, the use of intravascular ultrasound, and the use of high-pressure stent deployment varied among the trials, although all patients in STARS received a Palmaz-Schatz stent. The primary efficacy endpoints of the trials were similar, and included death, myocardial infarction and the need for repeat coronary angioplasty or CABG. All trials followed patients for at least 30 days.
In STARS, patients were randomized to receive one of three regimens for 4 weeks: aspirin alone, aspirin plus coumadin, or aspirin plus ticlopidine. Therapy was initiated following successful coronary stent placement. The primary endpoint was the incidence of stent thrombosis, defined as death, Q-Wave MI, or angiographic thrombus within the stented vessel demonstrated at the time of documented ischemia requiring emergent revascularization. The incidence rates for the primary endpoint and its components at 30 days are shown in the table below.
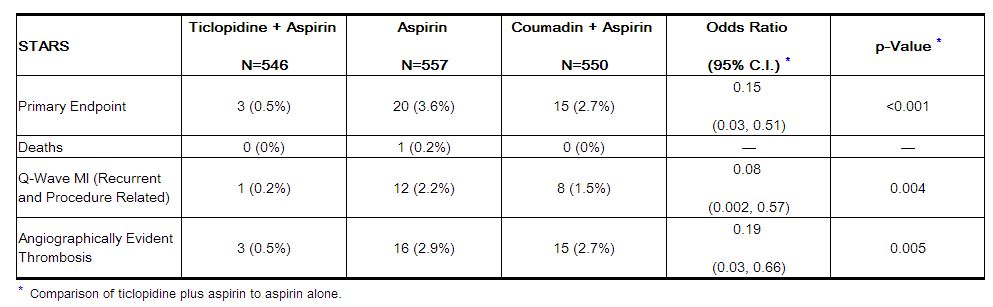 |
The use of ticlopidine plus aspirin did not affect the rate of non-Q-wave MIs when compared with aspirin alone or aspirin plus anticoagulants in STARS.
The use of ticlopidine plus aspirin was associated with a lower rate of recurrent cardiovascular events when compared with aspirin alone or aspirin plus anticoagulants in the other four randomized trials.
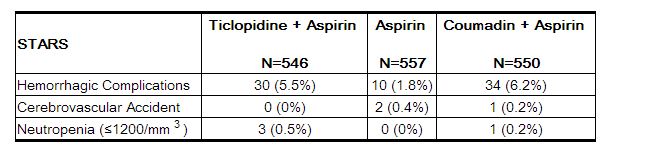
The rate of serious bleeding complications and neutropenia in STARS are shown in the table below. There were no cases of thrombotic thrombocytopenic purpura (TTP) or aplastic anemia reported in 1346 patients who received ticlopidine plus aspirin in the five randomized trials.[1]
 |
References
Adapted from the FDA Package Insert.