Fluconazole (oral)
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Deepika Beereddy, MBBS [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
Title
See full prescribing information for complete Boxed Warning.
ConditionName:
|
Overview
Fluconazole (oral) is an antifungal agent that is FDA approved for the treatment of {{{indication}}}. There is a Black Box Warning for this drug as shown here. Common adverse reactions include .
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Condition1
- Dosing Information
- Dosage
Condition2
- Dosing Information
- Dosage
Condition3
- Dosing Information
- Dosage
Condition4
- Dosing Information
- Dosage
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
Condition1
- Developed by:
- Class of Recommendation:
- Strength of Evidence:
- Dosing Information
- Dosage
Condition2
There is limited information regarding Off-Label Guideline-Supported Use of Fluconazole (oral) in adult patients.
Non–Guideline-Supported Use
Condition1
- Dosing Information
- Dosage
Condition2
There is limited information regarding Off-Label Non–Guideline-Supported Use of Fluconazole (oral) in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Condition1
- Dosing Information
- Dosage
Condition2
There is limited information regarding FDA-Labeled Use of Fluconazole (oral) in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
Condition1
- Developed by:
- Class of Recommendation:
- Strength of Evidence:
- Dosing Information
- Dosage
Condition2
There is limited information regarding Off-Label Guideline-Supported Use of Fluconazole (oral) in pediatric patients.
Non–Guideline-Supported Use
Condition1
- Dosing Information
- Dosage
Condition2
There is limited information regarding Off-Label Non–Guideline-Supported Use of Fluconazole (oral) in pediatric patients.
Contraindications
DIFLUCAN (fluconazole) is contraindicated in patients who have shown hypersensitivity to fluconazole or to any of its excipients. There is no information regarding cross-hypersensitivity between fluconazole and other azole antifungal agents. Caution should be used in prescribing DIFLUCAN to patients with hypersensitivity to other azoles. Coadministration of terfenadine is contraindicated in patients receiving DIFLUCAN (fluconazole) at multiple doses of 400 mg or higher based upon results of a multiple dose interaction study. Coadministration of other drugs known to prolong the QT interval and which are metabolized via the enzyme CYP3A4 such as cisapride, astemizole, erythromycin, pimozide, and quinidine are contraindicated in patients receiving fluconazole. (See CLINICAL PHARMACOLOGY: Drug Interaction Studies and PRECAUTIONS.)
Warnings
|
Title
See full prescribing information for complete Boxed Warning.
ConditionName:
|
- Description
Precautions
- Description
Adverse Reactions
Clinical Trials Experience
There is limited information regarding Clinical Trial Experience of Fluconazole (oral) in the drug label.
Body as a Whole
Cardiovascular
Digestive
Endocrine
Hematologic and Lymphatic
Metabolic and Nutritional
Musculoskeletal
Neurologic
Respiratory
Skin and Hypersensitivy Reactions
Special Senses
Urogenital
Miscellaneous
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Fluconazole (oral) in the drug label.
Body as a Whole
Cardiovascular
Digestive
Endocrine
Hematologic and Lymphatic
Metabolic and Nutritional
Musculoskeletal
Neurologic
Respiratory
Skin and Hypersensitivy Reactions
Special Senses
Urogenital
Miscellaneous
Drug Interactions
- Drug
- Description
Use in Specific Populations
Pregnancy
- Pregnancy Category
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Fluconazole (oral) in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Fluconazole (oral) during labor and delivery.
Nursing Mothers
There is no FDA guidance on the use of Fluconazole (oral) with respect to nursing mothers.
Pediatric Use
There is no FDA guidance on the use of Fluconazole (oral) with respect to pediatric patients.
Geriatic Use
There is no FDA guidance on the use of Fluconazole (oral) with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Fluconazole (oral) with respect to specific gender populations.
Race
There is no FDA guidance on the use of Fluconazole (oral) with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Fluconazole (oral) in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Fluconazole (oral) in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Fluconazole (oral) in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Fluconazole (oral) in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
- Intravenous
Monitoring
There is limited information regarding Monitoring of Fluconazole (oral) in the drug label.
- Description
IV Compatibility
There is limited information regarding IV Compatibility of Fluconazole (oral) in the drug label.
Overdosage
Acute Overdose
Signs and Symptoms
- Description
Management
- Description
Chronic Overdose
There is limited information regarding Chronic Overdose of Fluconazole (oral) in the drug label.
Pharmacology
There is limited information regarding Fluconazole (oral) Pharmacology in the drug label.
Mechanism of Action
Structure
DIFLUCAN® (fluconazole), the first of a new subclass of synthetic triazole antifungal agents, is available as tablets for oral administration, as a powder for oral suspension.
Fluconazole is designated chemically as 2,4-difluoro-α,α1-bis(1H-1,2,4-triazol-1-ylmethyl) benzyl alcohol with an empirical formula of C13H12F2N6O and molecular weight of 306.3. The structural formula is:

Fluconazole is a white crystalline solid which is slightly soluble in water and saline.
DIFLUCAN Tablets contain 50, 100, 150, or 200 mg of fluconazole and the following inactive ingredients: microcrystalline cellulose, dibasic calcium phosphate anhydrous, povidone, croscarmellose sodium, FD&C Red No. 40 aluminum lake dye, and magnesium stearate.
DIFLUCAN for Oral Suspension contains 350 mg or 1400 mg of fluconazole and the following inactive ingredients: sucrose, sodium citrate dihydrate, citric acid anhydrous, sodium benzoate, titanium dioxide, colloidal silicon dioxide, xanthan gum, and natural orange flavor. After reconstitution with 24 mL of distilled water or Purified Water (USP), each mL of reconstituted suspension contains 10 mg or 40 mg of fluconazole.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Fluconazole (oral) in the drug label.
Pharmacokinetics
The pharmacokinetic properties of fluconazole are similar following administration by the intravenous or oral routes. In normal volunteers, the bioavailability of orally administered fluconazole is over 90% compared with intravenous administration. Bioequivalence was established between the 100 mg tablet and both suspension strengths when administered as a single 200 mg dose.
Peak plasma concentrations (Cmax) in fasted normal volunteers occur between 1 and 2 hours with a terminal plasma elimination half-life of approximately 30 hours (range: 20–50 hours) after oral administration.
In fasted normal volunteers, administration of a single oral 400 mg dose of DIFLUCAN (fluconazole) leads to a mean Cmax of 6.72 µg/mL (range: 4.12 to 8.08 µg/mL) and after single oral doses of 50–400 mg, fluconazole plasma concentrations and AUC (area under the plasma concentration-time curve) are dose proportional.
The Cmax and AUC data from a food-effect study involving administration of DIFLUCAN (fluconazole) tablets to healthy volunteers under fasting conditions and with a high-fat meal indicated that exposure to the drug is not affected by food. Therefore, DIFLUCAN may be taken without regard to meals. (see DOSAGE AND ADMINISTRATION.)
Administration of a single oral 150 mg tablet of DIFLUCAN (fluconazole) to ten lactating women resulted in a mean Cmax of 2.61 µg/mL (range: 1.57 to 3.65 µg/mL).
Steady-state concentrations are reached within 5–10 days following oral doses of 50–400 mg given once daily. Administration of a loading dose (on day 1) of twice the usual daily dose results in plasma concentrations close to steady-state by the second day. The apparent volume of distribution of fluconazole approximates that of total body water. Plasma protein binding is low (11–12%). Following either single- or multiple oral doses for up to 14 days, fluconazole penetrates into all body fluids studied (see table below). In normal volunteers, saliva concentrations of fluconazole were equal to or slightly greater than plasma concentrations regardless of dose, route, or duration of dosing. In patients with bronchiectasis, sputum concentrations of fluconazole following a single 150 mg oral dose were equal to plasma concentrations at both 4 and 24 hours post dose. In patients with fungal meningitis, fluconazole concentrations in the CSF are approximately 80% of the corresponding plasma concentrations.
A single oral 150 mg dose of fluconazole administered to 27 patients penetrated into vaginal tissue, resulting in tissue:plasma ratios ranging from 0.94 to 1.14 over the first 48 hours following dosing.
A single oral 150 mg dose of fluconazole administered to 14 patients penetrated into vaginal fluid, resulting in fluid:plasma ratios ranging from 0.36 to 0.71 over the first 72 hours following dosing.

In normal volunteers, fluconazole is cleared primarily by renal excretion, with approximately 80% of the administered dose appearing in the urine as unchanged drug. About 11% of the dose is excreted in the urine as metabolites.
The pharmacokinetics of fluconazole are markedly affected by reduction in renal function. There is an inverse relationship between the elimination half-life and creatinine clearance. The dose of DIFLUCAN may need to be reduced in patients with impaired renal function. (See DOSAGE AND ADMINISTRATION.) A 3-hour hemodialysis session decreases plasma concentrations by approximately 50%.
In normal volunteers, DIFLUCAN administration (doses ranging from 200 mg to 400 mg once daily for up to 14 days) was associated with small and inconsistent effects on testosterone concentrations, endogenous corticosteroid concentrations, and the ACTH-stimulated cortisol response.
Pharmacokinetics in Children
In children, the following pharmacokinetic data {Mean (%cv)} have been reported:
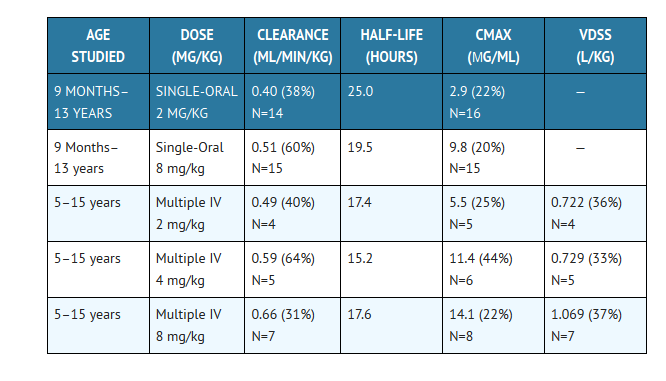
Clearance corrected for body weight was not affected by age in these studies. Mean body clearance in adults is reported to be 0.23 (17%) mL/min/kg.
In premature newborns (gestational age 26 to 29 weeks), the mean (%cv) clearance within 36 hours of birth was 0.180 (35%, N=7) mL/min/kg, which increased with time to a mean of 0.218 (31%, N=9) mL/min/kg six days later and 0.333 (56%, N=4) mL/min/kg 12 days later. Similarly, the half-life was 73.6 hours, which decreased with time to a mean of 53.2 hours six days later and 46.6 hours 12 days later.
Pharmacokinetics in Elderly
A pharmacokinetic study was conducted in 22 subjects, 65 years of age or older receiving a single 50 mg oral dose of fluconazole. Ten of these patients were concomitantly receiving diuretics. The Cmax was 1.54 mcg/mL and occurred at 1.3 hours post dose. The mean AUC was 76.4 ± 20.3 mcg∙h/mL, and the mean terminal half-life was 46.2 hours. These pharmacokinetic parameter values are higher than analogous values reported for normal young male volunteers. Coadministration of diuretics did not significantly alter AUC or Cmax. In addition, creatinine clearance (74 mL/min), the percent of drug recovered unchanged in urine (0–24 hr, 22%), and the fluconazole renal clearance estimates (0.124 mL/min/kg) for the elderly were generally lower than those of younger volunteers. Thus, the alteration of fluconazole disposition in the elderly appears to be related to reduced renal function characteristic of this group. A plot of each subject's terminal elimination half-life versus creatinine clearance compared with the predicted half-life – creatinine clearance curve derived from normal subjects and subjects with varying degrees of renal insufficiency indicated that 21 of 22 subjects fell within the 95% confidence limit of the predicted half-life – creatinine clearance curves. These results are consistent with the hypothesis that higher values for the pharmacokinetic parameters observed in the elderly subjects compared with normal young male volunteers are due to the decreased kidney function that is expected in the elderly.
Drug Interaction Studies
Oral contraceptives
Oral contraceptives were administered as a single dose both before and after the oral administration of DIFLUCAN 50 mg once daily for 10 days in 10 healthy women. There was no significant difference in ethinyl estradiol or levonorgestrel AUC after the administration of 50 mg of DIFLUCAN. The mean increase in ethinyl estradiol AUC was 6% (range: –47 to 108%) and levonorgestrel AUC increased 17% (range: –33 to 141%).
In a second study, twenty-five normal females received daily doses of both 200 mg DIFLUCAN tablets or placebo for two, ten-day periods. The treatment cycles were one month apart with all subjects receiving DIFLUCAN during one cycle and placebo during the other. The order of study treatment was random. Single doses of an oral contraceptive tablet containing levonorgestrel and ethinyl estradiol were administered on the final treatment day (day 10) of both cycles. Following administration of 200 mg of DIFLUCAN, the mean percentage increase of AUC for levonorgestrel compared to placebo was 25% (range: –12 to 82%) and the mean percentage increase for ethinyl estradiol compared to placebo was 38% (range: –11 to 101%). Both of these increases were statistically significantly different from placebo.
A third study evaluated the potential interaction of once weekly dosing of fluconazole 300 mg to 21 normal females taking an oral contraceptive containing ethinyl estradiol and norethindrone. In this placebo-controlled, double-blind, randomized, two-way crossover study carried out over three cycles of oral contraceptive treatment, fluconazole dosing resulted in small increases in the mean AUCs of ethinyl estradiol and norethindrone compared to similar placebo dosing. The mean AUCs of ethinyl estradiol and norethindrone increased by 24% (95% C.I. range: 18–31%) and 13% (95% C.I. range: 8–18%), respectively, relative to placebo. Fluconazole treatment did not cause a decrease in the ethinyl estradiol AUC of any individual subject in this study compared to placebo dosing. The individual AUC values of norethindrone decreased very slightly (<5%) in 3 of the 21 subjects after fluconazole treatment.
Cimetidine
DIFLUCAN 100 mg was administered as a single oral dose alone and two hours after a single dose of cimetidine 400 mg to six healthy male volunteers. After the administration of cimetidine, there was a significant decrease in fluconazole AUC and Cmax. There was a mean ± SD decrease in fluconazole AUC of 13% ± 11% (range: –3.4 to –31%) and Cmax decreased 19% ± 14% (range: –5 to –40%). However, the administration of cimetidine 600 mg to 900 mg intravenously over a four-hour period (from one hour before to 3 hours after a single oral dose of DIFLUCAN 200 mg) did not affect the bioavailability or pharmacokinetics of fluconazole in 24 healthy male volunteers.
Antacid
Administration of Maalox® (20 mL) to 14 normal male volunteers immediately prior to a single dose of DIFLUCAN 100 mg had no effect on the absorption or elimination of fluconazole.
Hydrochlorothiazide
Concomitant oral administration of 100 mg DIFLUCAN and 50 mg hydrochlorothiazide for 10 days in 13 normal volunteers resulted in a significant increase in fluconazole AUC and Cmax compared to DIFLUCAN given alone. There was a mean ± SD increase in fluconazole AUC and Cmax of 45% ± 31% (range: 19 to 114%) and 43% ± 31% (range: 19 to 122%), respectively. These changes are attributed to a mean ± SD reduction in renal clearance of 30% ± 12% (range: –10 to –50%).
Rifampin
Administration of a single oral 200 mg dose of DIFLUCAN after 15 days of rifampin administered as 600 mg daily in eight healthy male volunteers resulted in a significant decrease in fluconazole AUC and a significant increase in apparent oral clearance of fluconazole. There was a mean ± SD reduction in fluconazole AUC of 23% ± 9% (range: –13 to –42%). Apparent oral clearance of fluconazole increased 32% ± 17% (range: 16 to 72%). Fluconazole half-life decreased from 33.4 ± 4.4 hours to 26.8 ± 3.9 hours. (See PRECAUTIONS.)
Warfarin
There was a significant increase in prothrombin time response (area under the prothrombin time-time curve) following a single dose of warfarin (15 mg) administered to 13 normal male volunteers following oral DIFLUCAN 200 mg administered daily for 14 days as compared to the administration of warfarin alone. There was a mean ± SD increase in the prothrombin time response (area under the prothrombin time-time curve) of 7% ± 4% (range: –2 to 13%). (See PRECAUTIONS.) Mean is based on data from 12 subjects as one of 13 subjects experienced a 2-fold increase in his prothrombin time response.
Phenytoin
Phenytoin AUC was determined after 4 days of phenytoin dosing (200 mg daily, orally for 3 days followed by 250 mg intravenously for one dose) both with and without the administration of fluconazole (oral DIFLUCAN 200 mg daily for 16 days) in 10 normal male volunteers. There was a significant increase in phenytoin AUC. The mean ± SD increase in phenytoin AUC was 88% ± 68% (range: 16 to 247%). The absolute magnitude of this interaction is unknown because of the intrinsically nonlinear disposition of phenytoin. (See PRECAUTIONS.)
Cyclosporine
Cyclosporine AUC and Cmax were determined before and after the administration of fluconazole 200 mg daily for 14 days in eight renal transplant patients who had been on cyclosporine therapy for at least 6 months and on a stable cyclosporine dose for at least 6 weeks. There was a significant increase in cyclosporine AUC, Cmax, Cmin (24-hour concentration), and a significant reduction in apparent oral clearance following the administration of fluconazole. The mean ± SD increase in AUC was 92% ± 43% (range: 18 to 147%). The Cmax increased 60% ± 48% (range: –5 to 133%). The Cmin increased 157% ± 96% (range: 33 to 360%). The apparent oral clearance decreased 45% ± 15% (range: –15 to –60%). (See PRECAUTIONS.)
Zidovudine
Plasma zidovudine concentrations were determined on two occasions (before and following fluconazole 200 mg daily for 15 days) in 13 volunteers with AIDS or ARC who were on a stable zidovudine dose for at least two weeks. There was a significant increase in zidovudine AUC following the administration of fluconazole. The mean ± SD increase in AUC was 20% ± 32% (range: –27 to 104%). The metabolite, GZDV, to parent drug ratio significantly decreased after the administration of fluconazole, from 7.6 ± 3.6 to 5.7 ± 2.2.
Theophylline
The pharmacokinetics of theophylline were determined from a single intravenous dose of aminophylline (6 mg/kg) before and after the oral administration of fluconazole 200 mg daily for 14 days in 16 normal male volunteers. There were significant increases in theophylline AUC, Cmax, and half-life with a corresponding decrease in clearance. The mean ± SD theophylline AUC increased 21% ± 16% (range: –5 to 48%). The Cmax increased 13% ± 17% (range: –13 to 40%). Theophylline clearance decreased 16% ± 11% (range: –32 to 5%). The half-life of theophylline increased from 6.6 ± 1.7 hours to 7.9 ± 1.5 hours. (See PRECAUTIONS.)
Terfenadine
Six healthy volunteers received terfenadine 60 mg BID for 15 days. Fluconazole 200 mg was administered daily from days 9 through 15. Fluconazole did not affect terfenadine plasma concentrations. Terfenadine acid metabolite AUC increased 36% ± 36% (range: 7 to 102%) from day 8 to day 15 with the concomitant administration of fluconazole. There was no change in cardiac repolarization as measured by Holter QTc intervals. Another study at a 400 mg and 800 mg daily dose of fluconazole demonstrated that DIFLUCAN taken in doses of 400 mg per day or greater significantly increases plasma levels of terfenadine when taken concomitantly. (See CONTRAINDICATIONS and PRECAUTIONS.)
Quinidine
Although not studied in vitro or in vivo, concomitant administration of fluconazole with quinidine may result in inhibition of quinidine metabolism. Use of quinidine has been associated with QT prolongation and rare occurrences of torsades de pointes. Coadministration of fluconazole and quinidine is contraindicated. (See CONTRAINDICATIONS and PRECAUTIONS.)
Oral hypoglycemics
The effects of fluconazole on the pharmacokinetics of the sulfonylurea oral hypoglycemic agents tolbutamide, glipizide, and glyburide were evaluated in three placebo-controlled studies in normal volunteers. All subjects received the sulfonylurea alone as a single dose and again as a single dose following the administration of DIFLUCAN 100 mg daily for 7 days. In these three studies, 22/46 (47.8%) of DIFLUCAN treated patients and 9/22 (40.1%) of placebo-treated patients experienced symptoms consistent with hypoglycemia. (See PRECAUTIONS.)
Tolbutamide
In 13 normal male volunteers, there was significant increase in tolbutamide (500 mg single dose) AUC and Cmax following the administration of fluconazole. There was a mean ± SD increase in tolbutamide AUC of 26% ± 9% (range: 12 to 39%). Tolbutamide Cmax increased 11% ± 9% (range: –6 to 27%). (See PRECAUTIONS.)
Glipizide
The AUC and Cmax of glipizide (2.5 mg single dose) were significantly increased following the administration of fluconazole in 13 normal male volunteers. There was a mean ± SD increase in AUC of 49% ± 13% (range: 27 to 73%) and an increase in Cmax of 19% ± 23% (range: –11 to 79%). (See PRECAUTIONS.)
Glyburide
The AUC and Cmax of glyburide (5 mg single dose) were significantly increased following the administration of fluconazole in 20 normal male volunteers. There was a mean ± SD increase in AUC of 44% ± 29% (range: –13 to 115%) and Cmax increased 19% ± 19% (range: –23 to 62%). Five subjects required oral glucose following the ingestion of glyburide after 7 days of fluconazole administration. (See PRECAUTIONS.)
Rifabutin
There have been published reports that an interaction exists when fluconazole is administered concomitantly with rifabutin, leading to increased serum levels of rifabutin. (See PRECAUTIONS.)
Tacrolimus
There have been published reports that an interaction exists when fluconazole is administered concomitantly with tacrolimus, leading to increased serum levels of tacrolimus. (See PRECAUTIONS.)
Cisapride
A placebo-controlled, randomized, multiple-dose study examined the potential interaction of fluconazole with cisapride. Two groups of 10 normal subjects were administered fluconazole 200 mg daily or placebo. Cisapride 20 mg four times daily was started after 7 days of fluconazole or placebo dosing. Following a single dose of fluconazole, there was a 101% increase in the cisapride AUC and a 91% increase in the cisapride Cmax. Following multiple doses of fluconazole, there was a 192% increase in the cisapride AUC and a 154% increase in the cisapride Cmax. Fluconazole significantly increased the QTc interval in subjects receiving cisapride 20 mg four times daily for 5 days. (See CONTRAINDICATIONS and PRECAUTIONS.)
Midazolam
The effect of fluconazole on the pharmacokinetics and pharmacodynamics of midazolam was examined in a randomized, cross-over study in 12 volunteers. In the study, subjects ingested placebo or 400 mg fluconazole on Day 1 followed by 200 mg daily from Day 2 to Day 6. In addition, a 7.5 mg dose of midazolam was orally ingested on the first day, 0.05 mg/kg was administered intravenously on the fourth day, and 7.5 mg orally on the sixth day. Fluconazole reduced the clearance of IV midazolam by 51%. On the first day of dosing, fluconazole increased the midazolam AUC and Cmax by 259% and 150%, respectively. On the sixth day of dosing, fluconazole increased the midazolam AUC and Cmax by 259% and 74%, respectively. The psychomotor effects of midazolam were significantly increased after oral administration of midazolam but not significantly affected following intravenous midazolam.
A second randomized, double-dummy, placebo-controlled, cross over study in three phases was performed to determine the effect of route of administration of fluconazole on the interaction between fluconazole and midazolam. In each phase the subjects were given oral fluconazole 400 mg and intravenous saline; oral placebo and intravenous fluconazole 400 mg; and oral placebo and IV saline. An oral dose of 7.5 mg of midazolam was ingested after fluconazole/placebo. The AUC and Cmax of midazolam were significantly higher after oral than IV administration of fluconazole. Oral fluconazole increased the midazolam AUC and Cmax by 272% and 129%, respectively. IV fluconazole increased the midazolam AUC and Cmax by 244% and 79%, respectively. Both oral and IV fluconazole increased the pharmacodynamic effects of midazolam. (See PRECAUTIONS.)
Azithromycin
An open-label, randomized, three-way crossover study in 18 healthy subjects assessed the effect of a single 800 mg oral dose of fluconazole on the pharmacokinetics of a single 1200 mg oral dose of azithromycin as well as the effects of azithromycin on the pharmacokinetics of fluconazole. There was no significant pharmacokinetic interaction between fluconazole and azithromycin.
Voriconazole
Voriconazole is a substrate for both CYP2C9 and CYP3A4 isoenzymes. Concurrent administration of oral Voriconazole (400 mg Q12h for 1 day, then 200 mg Q12h for 2.5 days) and oral fluconazole (400 mg on day 1, then 200 mg Q24h for 4 days) to 6 healthy male subjects resulted in an increase in Cmax and AUCτ of voriconazole by an average of 57% (90% CI: 20%, 107%) and 79% (90% CI: 40%, 128%), respectively. In a follow-on clinical study involving 8 healthy male subjects, reduced dosing and/or frequency of voriconazole and fluconazole did not eliminate or diminish this effect. Concomitant administration of voriconazole and fluconazole at any dose is not recommended. Close monitoring for adverse events related to voriconazole is recommended if voriconazole is used sequentially after fluconazole, especially within 24 h of the last dose of fluconazole. (See PRECAUTIONS)
Tofacitinib
Co-administration of fluconazole (400 mg on Day 1 and 200 mg once daily for 6 days [Days 2–7]) and tofacitinib (30 mg single dose on Day 5) in healthy subjects resulted in increased mean tofacitinib AUC and Cmax values of approximately 79% (90% CI: 64% – 96%) and 27% (90% CI: 12% – 44%), respectively, compared to administration of tofacitinib alone. (See PRECAUTIONS)
Microbiology
Mechanism of Action
Fluconazole is a highly selective inhibitor of fungal cytochrome P450 dependent enzyme lanosterol 14-α-demethylase. This enzyme functions to convert lanosterol to ergosterol. The subsequent loss of normal sterols correlates with the accumulation of 14-α-methyl sterols in fungi and may be responsible for the fungistatic activity of fluconazole. Mammalian cell demethylation is much less sensitive to fluconazole inhibition.
Drug Resistance
Fluconazole resistance may arise from a modification in the quality or quantity of the target enzyme (lanosterol 14-α-demethylase), reduced access to the drug target, or some combination of these mechanisms.
Point mutations in the gene (ERG11) encoding for the target enzyme lead to an altered target with decreased affinity for azoles. Overexpression of ERG11 results in the production of high concentrations of the target enzyme, creating the need for higher intracellular drug concentrations to inhibit all of the enzyme molecules in the cell.
The second major mechanism of drug resistance involves active efflux of fluconazole out of the cell through the activation of two types of multidrug efflux transporters; the major facilitators (encoded by MDR genes) and those of the ATP-binding cassette superfamily (encoded by CDR genes). Upregulation of the MDR gene leads to fluconazole resistance, whereas, upregulation of CDR genes may lead to resistance to multiple azoles.
Resistance in Candida glabrata usually includes upregulation of CDR genes resulting in resistance to multiple azoles. For an isolate where the MIC is categorized as Intermediate (16 to 32 µg/mL), the highest fluconazole dose is recommended.
Candida krusei should be considered to be resistant to fluconazole. Resistance in C. krusei appears to be mediated by reduced sensitivity of the target enzyme to inhibition by the agent.
There have been reports of cases of superinfection with Candida species other than C. albicans, which are often inherently not susceptible to DIFLUCAN (e.g., Candida krusei). Such cases may require alternative antifungal therapy.
Activity In Vitro and In Clinical Infections
Fluconazole has been shown to be active against most strains of the following microorganisms both in vitro and in clinical infections.
Candida albicans Candida glabrata (Many strains are intermediately susceptible)1 Candida parapsilosis Candida tropicalis Cryptococcus neoformans
The following in vitro data are available, but their clinical significance is unknown.
Fluconazole exhibits in vitro minimum inhibitory concentrations (MIC values) of 8 µg/mL or less against most (≥90%) strains of the following microorganisms, however, the safety and effectiveness of fluconazole in treating clinical infections due to these microorganisms have not been established in adequate and well-controlled trials.
Candida dubliniensis Candida guilliermondii Candida kefyr Candida lusitaniae
Candida krusei should be considered to be resistant to fluconazole. Resistance in C. krusei appears to be mediated by reduced sensitivity of the target enzyme to inhibition by the agent.
There have been reports of cases of superinfection with Candida species other than C. albicans, which are often inherently not susceptible to DIFLUCAN (e.g., Candida krusei). Such cases may require alternative antifungal therapy.
* In a majority of the studies, fluconazole MIC90 values against C. glabrata were above the susceptible breakpoint (≥16 µg/mL). Resistance in Candida glabrata usually includes upregulation of CDR genes resulting in resistance to multiple azoles. For an isolate where the MIC is categorized as intermediate (16 to 32 µg/mL, see Table 1), the highest dose is recommended (see DOSAGE AND ADMINISTRATION ). For resistant isolates, alternative therapy is recommended.
Susceptibility Testing Methods
Cryptococcus neoformans and filamentous fungi
No interpretive criteria have been established for Cryptococcus neoformans and filamentous fungi.
Candida species
Broth Dilution Techniques
Quantitative methods are used to determine antifungal minimum inhibitory concentrations (MICs). These MICs provide estimates of the susceptibility of Candida spp. to antifungal agents. MICs should be determined using a standardized procedure. Standardized procedures are based on a dilution method (broth)1 with standardized inoculum concentrations of fluconazole powder. The MIC values should be interpreted according to the criteria provided in Table 1.
Diffusion Techniques
Qualitative methods that require measurement of zone diameters also provide reproducible estimates of the susceptibility of Candida spp. to an antifungal agent. One such standardized procedure2 requires the use of standardized inoculum concentrations. This procedure uses paper disks impregnated with 25 µg of fluconazole to test the susceptibility of yeasts to fluconazole. Disk diffusion interpretive criteria are also provided in Table 1.

The susceptible category implies that isolates are inhibited by the usually achievable concentrations of antifungal agent tested when the recommended dosage is used. The intermediate category implies that an infection due to the isolate may be appropriately treated in body sites where the drugs are physiologically concentrated or when a high dosage of drug is used. The resistant category implies that isolates are not inhibited by the usually achievable concentrations of the agent with normal dosage schedules and clinical efficacy of the agent against the isolate has not been reliably shown in treatment studies.
Quality Control
Standardized susceptibility test procedures require the use of quality control organisms to control the technical aspects of the test procedures. Standardized fluconazole powder and 25 µg disks should provide the following range of values noted in Table 2. NOTE: Quality control microorganisms are specific strains of organisms with intrinsic biological properties relating to resistance mechanisms and their genetic expression within fungi; the specific strains used for microbiological control are not clinically significant.

Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Fluconazole (oral) in the drug label.
Clinical Studies
Cryptococcal meningitis
In a multicenter study comparing DIFLUCAN (200 mg/day) to amphotericin B (0.3 mg/kg/day) for treatment of cryptococcal meningitis in patients with AIDS, a multivariate analysis revealed three pretreatment factors that predicted death during the course of therapy: abnormal mental status, cerebrospinal fluid cryptococcal antigen titer greater than 1:1024, and cerebrospinal fluid white blood cell count of less than 20 cells/mm3. Mortality among high risk patients was 33% and 40% for amphotericin B and DIFLUCAN patients, respectively (p=0.58), with overall deaths 14% (9 of 63 subjects) and 18% (24 of 131 subjects) for the 2 arms of the study (p=0.48). Optimal doses and regimens for patients with acute cryptococcal meningitis and at high risk for treatment failure remain to be determined. (Saag, et al. N Engl J Med 1992; 326:83–9.)
Vaginal candidiasis
Two adequate and well-controlled studies were conducted in the U.S. using the 150 mg tablet. In both, the results of the fluconazole regimen were comparable to the control regimen (clotrimazole or miconazole intravaginally for 7 days) both clinically and statistically at the one month post-treatment evaluation.
The therapeutic cure rate, defined as a complete resolution of signs and symptoms of vaginal candidiasis (clinical cure), along with a negative KOH examination and negative culture for Candida (microbiologic eradication), was 55% in both the fluconazole group and the vaginal products group.

Approximately three-fourths of the enrolled patients had acute vaginitis (<4 episodes/12 months) and achieved 80% clinical cure, 67% mycologic eradication, and 59% therapeutic cure when treated with a 150 mg DIFLUCAN tablet administered orally. These rates were comparable to control products. The remaining one-fourth of enrolled patients had recurrent vaginitis (≥4 episodes/12 months) and achieved 57% clinical cure, 47% mycologic eradication, and 40% therapeutic cure. The numbers are too small to make meaningful clinical or statistical comparisons with vaginal products in the treatment of patients with recurrent vaginitis.
Substantially more gastrointestinal events were reported in the fluconazole group compared to the vaginal product group. Most of the events were mild to moderate. Because fluconazole was given as a single dose, no discontinuations occurred.

Pediatric Studies
Oropharyngeal candidiasis
An open-label, comparative study of the efficacy and safety of DIFLUCAN (2–3 mg/kg/day) and oral nystatin (400,000 I.U. 4 times daily) in immunocompromised children with oropharyngeal candidiasis was conducted. Clinical and mycological response rates were higher in the children treated with fluconazole.
Clinical cure at the end of treatment was reported for 86% of fluconazole treated patients compared to 46% of nystatin treated patients. Mycologically, 76% of fluconazole treated patients had the infecting organism eradicated compared to 11% for nystatin treated patients.

The proportion of patients with clinical relapse 2 weeks after the end of treatment was 14% for subjects receiving DIFLUCAN and 16% for subjects receiving nystatin. At 4 weeks after the end of treatment, the percentages of patients with clinical relapse were 22% for DIFLUCAN and 23% for nystatin.
How Supplied
Storage
There is limited information regarding Fluconazole (oral) Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Fluconazole (oral) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Fluconazole (oral) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Fluconazole (oral) in the drug label.
Precautions with Alcohol
- Alcohol-Fluconazole (oral) interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- ®[1]
Look-Alike Drug Names
- A® — B®[2]
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ Empty citation (help)
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Page Name=Fluconazole (oral)
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Fluconazole (oral) |Label Name=Fluconazole (oral)11.png
}}
{{#subobject:
|Label Page=Fluconazole (oral) |Label Name=Fluconazole (oral)11.png
}}