Fluticasone/salmeterol: Difference between revisions
No edit summary |
No edit summary |
||
| Line 118: | Line 118: | ||
(Description) | (Description) | ||
|clinicalTrials=======Central Nervous System====== | |clinicalTrials=======Central Nervous System====== | ||
| Line 208: | Line 207: | ||
|othersTitle=Others | |othersTitle=Others | ||
|useInOthers=(Description) | |useInOthers=(Description) | ||
|administration=(Oral/Intravenous/etc) | |administration=(Oral/Intravenous/etc) | ||
|monitoring======Condition 1===== | |monitoring======Condition 1===== | ||
| Line 370: | Line 368: | ||
(Description) | (Description) | ||
|drugBox={{Drugbox2 | |drugBox={{Drugbox2 | ||
| | | type = combo | ||
| | | image = Fluticasone propionate.svg | ||
| | | image2 = Salmeterol.png | ||
| | | component1 = Fluticasone | ||
| class1 = [[Glucocorticoids|Glucocorticoid]] | |||
| component2 = Salmeterol | |||
| | | class2 = [[Beta2-adrenergic receptor agonist|Long-Acting Beta2 Agonist]] | ||
| | | CAS_number = | ||
| | | ATC_prefix = | ||
| | | ATC_suffix = | ||
| | | PubChem = | ||
| | | DrugBank = | ||
| | | pregnancy_AU = <!-- A / B1 / B2 / B3 / C / D / X --> | ||
| pregnancy_US = <!-- A / B / C / D / X --> | |||
<!-- | | pregnancy_category= | ||
| | | legal_AU = <!-- Unscheduled / S2 / S3 / S4 / S5 / S6 / S7 / S8 / S9 --> | ||
| | | legal_CA = <!-- / Schedule I, II, III, IV, V, VI, VII, VIII --> | ||
| legal_UK = P | |||
| | | legal_US = <!-- OTC / Rx-only / Schedule I, II, III, IV, V --> | ||
| legal_status = | |||
<!-- | | routes_of_administration = Inhaled | ||
| | |||
| | |||
<!-- | |||
| | |||
| | |||
|mechAction=(Description) | |mechAction=(Description) | ||
|structure=(Description with picture) | |structure=(Description with picture) | ||
| Line 440: | Line 407: | ||
|howSupplied=(Description) | |howSupplied=(Description) | ||
|fdaPatientInfo=(Patient Counseling Information) | |fdaPatientInfo=(Patient Counseling Information) | ||
| | |alcohol=Alcohol-Fluticasone/salmeterol interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication. | ||
|lookAlike=* (Paired Confused Name 1a) — (Paired Confused Name 1b) | |lookAlike=* (Paired Confused Name 1a) — (Paired Confused Name 1b) | ||
* (Paired Confused Name 2a) — (Paired Confused Name 2b) | * (Paired Confused Name 2a) — (Paired Confused Name 2b) | ||
* (Paired Confused Name 3a) — (Paired Confused Name 3b) | * (Paired Confused Name 3a) — (Paired Confused Name 3b) | ||
|nlmPatientInfo=(Link to patient information page) | |||
|drugShortage=Drug Shortage | |drugShortage=Drug Shortage | ||
}} | }} | ||
{{Asthma_and_copd_rx}} | {{Asthma_and_copd_rx}} | ||
Revision as of 13:30, 4 August 2014
{{DrugProjectFormSinglePage |genericName=generic name |aOrAn=a |drugClass=Adrenergic receptor agonist |indication=a list of indications, separated by commas. |hasBlackBoxWarning=Yes |adverseReactions=a list of adverse reactions, separated by commas. |blackBoxWarningTitle=Warning Title |blackBoxWarningBody=Condition Name: (Content) |fdaLIADAdult======Condition 1=====
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
|offLabelAdultGuideSupport======Condition 1=====
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Condition 2
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
|offLabelAdultNoGuideSupport======Condition 1=====
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Condition 3
- Dosing Information
- (Dosage)
|fdaLIADPed======Condition 1=====
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
|offLabelPedGuideSupport======Condition 1=====
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Condition 2
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
|offLabelPedNoGuideSupport======Condition 1=====
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Condition 3
- Dosing Information
- (Dosage)
|contraindications=* Condition 1
- Condition 2
- Condition 3
- Condition 4
- Condition 5
|warnings======Conidition 1=====
(Description) |clinicalTrials=======Central Nervous System======
- (list/description of adverse reactions)
Cardiovascular
- (list/description of adverse reactions)
Respiratory
- (list/description of adverse reactions)
Gastrointestinal
- (list/description of adverse reactions)
Hypersensitive Reactions
- (list/description of adverse reactions)
Miscellaneous
- (list/description of adverse reactions)
Condition 2
Central Nervous System
- (list/description of adverse reactions)
Cardiovascular
- (list/description of adverse reactions)
Respiratory
- (list/description of adverse reactions)
Gastrointestinal
- (list/description of adverse reactions)
Hypersensitive Reactions
- (list/description of adverse reactions)
Miscellaneous
- (list/description of adverse reactions)
|postmarketing=(Description) |drugInteractions=* Drug 1
- Drug 2
- Drug 3
- Drug 4
- Drug 5
Drug 1
(Description)
Drug 2
(Description)
Drug 3
(Description)
Drug 4
(Description)
Drug 5
(Description) |useInPregnancyFDA=(Description) |useInPregnancyAUS=(Description) |useInLaborDelivery=(Description) |useInNursing=(Description) |useInPed=(Description) |useInGeri=(Description) |useInGender=(Description) |useInRace=(Description) |useInRenalImpair=(Description) |useInHepaticImpair=(Description) |useInReproPotential=(Description) |useInImmunocomp=(Description) |othersTitle=Others |useInOthers=(Description) |administration=(Oral/Intravenous/etc) |monitoring======Condition 1=====
(Description regarding monitoring, from Warnings section)
Condition 2
(Description regarding monitoring, from Warnings section)
Condition 3
(Description regarding monitoring, from Warnings section) |IVCompat====Solution===
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Y-Site
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Admixture
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Syringe
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
TPN/TNA
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
|overdose====Acute Overdose===
Signs and Symptoms
(Description)
Management
(Description)
Chronic Overdose
Signs and Symptoms
(Description)
Management
(Description) |drugBox=
| Error creating thumbnail: File missing | |
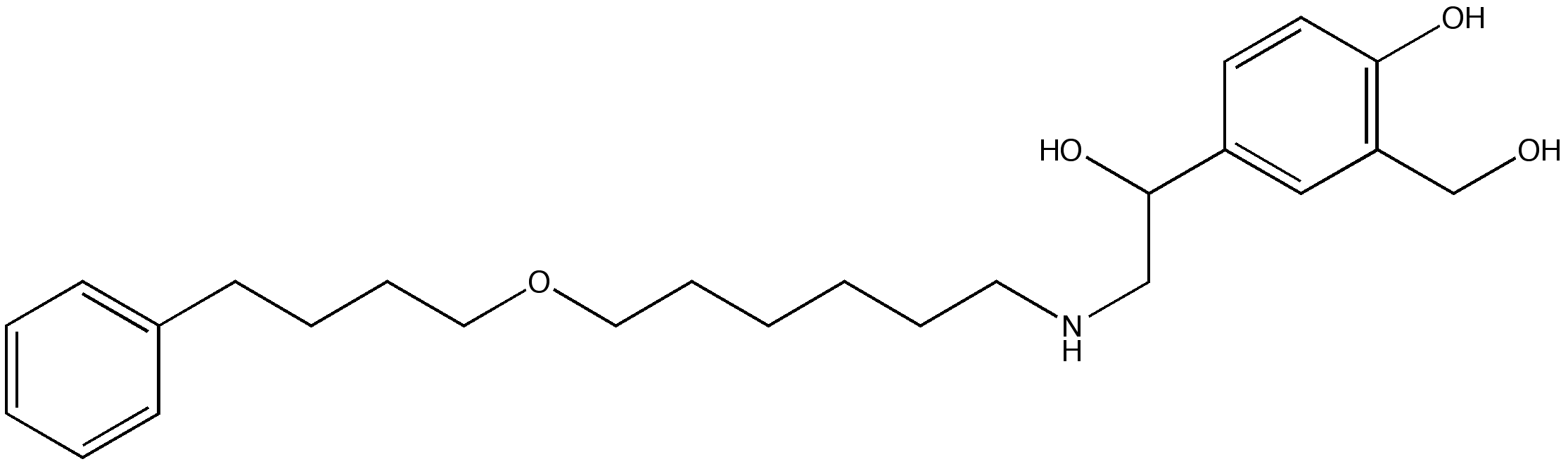
| |
Fluticasone/salmeterol
| |
| Combination of | |
| Fluticasone | Glucocorticoid |
| Salmeterol | Long-Acting Beta2 Agonist |
| Identifiers | |
| CAS number | ? |
| ATC code | ? |
| PubChem | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status |
P(UK) |
| Routes | Inhaled |