Vinpocetine: Difference between revisions
m (Robot: Automated text replacement (-{{reflist}} +{{reflist|2}}, -<references /> +{{reflist|2}}, -{{WikiDoc Cardiology Network Infobox}} +)) |
Gerald Chi (talk | contribs) mNo edit summary |
||
| (2 intermediate revisions by one other user not shown) | |||
| Line 1: | Line 1: | ||
{{Drugbox| | {{Drugbox | ||
|IUPAC_name = | | Verifiedfields = changed | ||
| Watchedfields = changed | |||
| verifiedrevid = 470630524 | |||
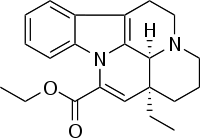
| IUPAC_name = (3α,16α)-Eburnamenine-14-carboxylic acid ethyl ester | |||
| image = Vinpocetine.png | | image = Vinpocetine.png | ||
| width = | | width = 200px | ||
| CAS_number=42971-09-5 | | image2 = Vinpocetine ball-and-stick.png | ||
| ATC_prefix=N06 | | width2 = 200px | ||
| ATC_suffix=BX18 | |||
| | <!--Clinical data--> | ||
| PubChem= | | tradename = | ||
| | | Drugs.com = {{drugs.com|international|vinpocetine}} | ||
| C = 22 | H = 26 | N = 2 | O = 2 | | pregnancy_category = not recommended | ||
| legal_status = OTC US, RX only Europe, not sold in Australia, Canada or New Zealand | |||
| routes_of_administration = Oral, intravenous | |||
<!--Pharmacokinetic data--> | |||
| bioavailability = 56.6 +/- 8.9% | |||
| metabolism = hepatic | |||
| elimination_half-life = 2.54 +/- 0.48 hours | |||
| excretion = renal | |||
<!--Identifiers--> | |||
| CAS_number_Ref = {{cascite|changed|??}} | |||
| CAS_number = 42971-09-5 | |||
| ATC_prefix = N06 | |||
| ATC_suffix = BX18 | |||
| ChEMBL_Ref = {{ebicite|correct|EBI}} | |||
| ChEMBL = 71752 | |||
| PubChem = 443955 | |||
| DrugBank_Ref = {{drugbankcite|correct|drugbank}} | |||
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} | |||
| ChemSpiderID = 392007 | |||
| UNII_Ref = {{fdacite|correct|FDA}} | |||
| UNII = 543512OBTC | |||
| KEGG_Ref = {{keggcite|correct|kegg}} | |||
| KEGG = D01371 | |||
<!--Chemical data--> | |||
| C=22 | H=26 | N=2 | O=2 | |||
| molecular_weight = 350.454 g/mol | | molecular_weight = 350.454 g/mol | ||
| | | smiles = O=C(OCC)C=4n1c3c(c2ccccc12)CCN5[C@H]3[C@](C=4)(CCC5)CC | ||
| | | InChI = 1/C22H26N2O2/c1-3-22-11-7-12-23-13-10-16-15-8-5-6-9-17(15)24(19(16)20(22)23)18(14-22)21(25)26-4-2/h5-6,8-9,14,20H,3-4,7,10-13H2,1-2H3/t20-,22+/m1/s1 | ||
| InChIKey = DDNCQMVWWZOMLN-IRLDBZIGBY | |||
| | | StdInChI_Ref = {{stdinchicite|correct|chemspider}} | ||
| | | StdInChI = 1S/C22H26N2O2/c1-3-22-11-7-12-23-13-10-16-15-8-5-6-9-17(15)24(19(16)20(22)23)18(14-22)21(25)26-4-2/h5-6,8-9,14,20H,3-4,7,10-13H2,1-2H3/t20-,22+/m1/s1 | ||
| | | StdInChIKey_Ref = {{stdinchicite|correct|chemspider}} | ||
| | | StdInChIKey = DDNCQMVWWZOMLN-IRLDBZIGSA-N | ||
}} | }} | ||
__NOTOC__ | |||
{{CMG}} | |||
==Overview== | |||
'''Vinpocetine''' (brand names: '''Cavinton''', '''Intelectol'''; chemical name: '''ethyl apovincaminate''') is a [[semisynthesis|semisynthetic]] [[derivative (chemistry)|derivative]] of the [[vinca alkaloid]] [[vincamine]] (sometimes described as "a [[Chemical synthesis|synthetic]] [[ethyl ester]] of [[apovincamine]]"),<ref>{{Cite journal|author=Lörincz C, Szász K, Kisfaludy L |title=The synthesis of ethyl apovincaminate |journal=Arzneimittel-Forschung |volume=26 |issue=10a |page=1907 |year=1976 |pmid=1037211}}</ref> an extract from the [[Vinca minor|lesser periwinkle]] plant. Vinpocetine was first isolated from the plant in 1975 by the Hungarian chemist Csaba Szántay. The mass production of the synthetic drug was started in 1978 by the Hungarian pharmaceutical company [[Gedeon Richter|Richter Gedeon]]. | |||
Vinpocetine is reported to have cerebral blood-flow enhancing<ref>{{Cite pmid|15760651}}</ref> and neuroprotective effects,<ref>{{Cite pmid|12498034}}</ref> and is used as a drug in Eastern Europe for the treatment of [[cerebrovascular disorders]] and age-related memory impairment.<ref>{{Cite journal|title=Vinpocetine. Monograph |journal=Alternative Medicine Review |volume=7 |issue=3 |pages=240–3 |year=2002 |pmid=12126465 |url=http://addium.com/studies/240.pdf}}</ref> | |||
Vinpocetine is not approved in the United States for pharmaceutical use, but it can be sold as a dietary supplement. Vinpocetine is widely marketed as a supplement for [[vasodilation]] and as a [[nootropic]] for the improvement of memory and cerebral metabolism. Vinpocetine has been identified as a potent anti-inflammatory agent that might have a potential role in the treatment of [[Parkinson's disease]] and [[Alzheimer's disease]].<ref name="Jeon">{{Cite pmid|20448200}}</ref><ref name="Medina">{{Cite pmid|20495091}}</ref> | |||
== | ==Controlled clinical trials== | ||
As of 2003 only three controlled [[clinical trial]]s had tested "older adults with memory problems".<ref name=McDaniel2003>{{Cite journal|author=McDaniel MA, Maier SF, Einstein GO |title='Brain-specific' nutrients: a memory cure? |journal=Nutrition |volume=19 |issue=11–12 |pages=957–75 |year=2003 |pmid=14624946 |doi=10.1016/S0899-9007(03)00024-8}}</ref> However, a 2003 [[Cochrane review]] determined that the results were inconclusive.<ref name=Szatmari2003>{{Cite journal|author=Szatmari SZ, Whitehouse PJ|editor1-last=Szatmári|editor1-first=Szabolcs |title=Vinpocetine for cognitive impairment and dementia |journal=Cochrane Database of Systematic Reviews |issue=1 |pages=CD003119 |year=2003 |pmid=12535455 |doi=10.1002/14651858.CD003119}}</ref> | |||
Prior to 2003, a different study from 1985<ref>{{Cite journal|author=Subhan Z, Hindmarch I |title=Psychopharmacological effects of vinpocetine in normal healthy volunteers |journal=European Journal of Clinical Pharmacology |volume=28 |issue=5 |pages=567–71 |year=1985 |pmid=3899677 |doi=10.1007/BF00544068}}</ref> had tested young, healthy adults, but this study had 12 subjects and used a short treatment period.<ref name=McDaniel2003/> | |||
==Use as a vasodilator== | |||
Vinpocetine is widely used in the body building community as a [[vasodilator]]. Although no studies have been conducted on the effectiveness of vinpocetine on performance enhancement during exercise, both beneficial and adverse effects have been reported on body building forums.{{Citation needed|reason=Needs a reliable reference source |date=September 2013}} | |||
Vinpocetine | ==Anticonvulsant potential== | ||
[[Kindling model]]s in rats has shown Vinpocetine to exhibit anticonvulsant properties, the most pronounced anticonvulsant effects were observed in [[Pentylenetetrazol|Pentylenetetrazole]] (PTZ)-kindled rats although there was also an effect on amygdala-kindled and neocortically-kindled rats.<ref>{{cite journal | pmid = 2271012 | year = 1990 | last1 = Schmidt | first1 = J | title = Comparative studies on the anticonvulsant effectiveness of nootropic drugs in kindled rats | volume = 49 | issue = 5 | pages = 413–9 | journal = Biomedica biochimica acta}}</ref> Vinpocetine has also been shown to abolished [3H]Glu release after ''in vivo'' exposure to [[4-aminopyridine]] (4-AP) which suggests an important mechanism for vinpocetine anticonvulsant potential.<ref>{{cite journal | pmid = 21737246 | year = 2011 | last1 = Sitges | first1 = M | last2 = Sanchez-Tafolla | first2 = BM | last3 = Chiu | first3 = LM | last4 = Aldana | first4 = BI | last5 = Guarneros | first5 = A | title = Vinpocetine inhibits glutamate release induced by the convulsive agent 4-aminopyridine more potently than several antiepileptic drugs | volume = 96 | issue = 3 | pages = 257–66 | doi = 10.1016/j.eplepsyres.2011.06.006 | journal = Epilepsy research}}</ref> | |||
==Anti-inflammatory action== | |||
Vinpocetine has been identified as a novel [[anti-inflammatory]] agent.<ref name="Jeon"/><ref name="Medina"/> Vinpocetine inhibits the up-regulation of [[NF-κB]] by [[TNFα]] in various cell tests. [[Reverse transcription polymerase chain reaction]] also shows that it reduced the TNFα-induced expression of the [[mRNA]] of proinflammatory molecules such as [[IL1B|interleukin-1 beta]], [[monocyte chemoattractant protein-1]] (MCP-1), and [[vascular cell adhesion molecule-1]] (VCAM-1). In mice, vinpocetine reduced [[lipopolysaccharide]] inoculation induced [[polymorphonuclear neutrophil]] infiltration into the lung.<ref name="Jeon"/><ref name="Medina"/> Neuroinflammatory processes can result in neuronal death in [[Parkinson's disease]] (PD) and [[Alzheimer's disease]] (AD). It has been suggested that "it would be interesting to test whether vinpocetine’s antiinflammatory properties would have a protective effect in models of neurodegenerative conditions such as [[Alzheimer’s disease|AD]] and [[Parkinson’s disease|PD]]."<ref name="Medina"/> | |||
== | ==Mechanism of action== | ||
Vinpocetine has been shown to selectively inhibit voltage-sensitive Na+ channels, resulting in a dose-dependent decrease in evoked extracellular Ca+ ions in striatal nerve endings.<ref>{{Cite journal|author=Sitges M, Galván E, Nekrassov V |title=Vinpocetine blockade of sodium channels inhibits the rise in sodium and calcium induced by 4-aminopyridine in synaptosomes |journal=Neurochemistry International |volume=46 |issue=7 |pages=533–40 |year=2005 |pmid=15843047 |doi=10.1016/j.neuint.2005.02.001}}</ref> The Na+ channel inhibiting properties of vinpocetine are thought to contribute to a general neuroprotective effect through blockade of excitotoxicity and attenuation of neuronal damage induced by cerebral ischemia/reperfusion.<ref>{{Cite journal|author=Adám-Vizi V |title=[Neuroprotective effect of sodium channel blockers in ischemia: the pathomechanism of early ischemic dysfunction] |language=Hungarian |journal=Orvosi Hetilap |volume=141 |issue=23 |pages=1279–86 |year=2000 |pmid=10905082}}</ref> | |||
Vinpocetine is also a [[phosphodiesterase]] (PDE) type-1 inhibitor,<ref>{{Cite journal|author=Hagiwara M, Endo T, Hidaka H |title=Effects of vinpocetine on cyclic nucleotide metabolism in vascular smooth muscle |journal=[[Biochemical Pharmacology]] |volume=33 |issue=3 |pages=453–7 |year=1984 |pmid=6322804 |doi=10.1016/0006-2952(84)90240-5}}</ref> (with an [[IC50|IC<sub>50</sub>]] of approximately 10<sup>−5</sup> M.) leading to increases in intracellular levels of cyclic guanosine 3'5'-monophosphate (cGMP), an action that causes the vasorelaxant effects of vinpocetine on cerebral smooth muscle tissue.<ref>{{Cite journal|author=Truss MC, Uckert S, Stief CG, Forssmann WG, Jonas U |title=Cyclic nucleotide phosphodiesterase (PDE) isoenzymes in the human detrusor smooth muscle. II. Effect of various PDE inhibitors on smooth muscle tone and cyclic nucleotide levels in vitro |journal=Urological Research |volume=24 |issue=3 |pages=129–34 |year=1996 |pmid=8839479|doi=10.1007/BF00304075}}</ref><ref>{{Cite journal|author=Gurkovskaia AV, Gokina NI, Buryĭ VA, Shuba MF |title=[Electrophysiological analysis of the action of kavinton on the smooth muscles] |language=Russian |journal=Biulleten' Eksperimental'noĭ Biologii I Meditsiny |volume=103 |issue=1 |pages=68–71 |year=1987 |pmid=3801654}}</ref> | |||
Independent of vinpocetine's action on PDE, vinpocetine inhibits [[IκB kinase|IKK]] preventing [[IκB]] degradation and the following translocation of [[NF-κB]] to the cell nucleus.<ref name="Jeon"/><ref name="Medina"/> | |||
Increases in neuronal levels of [[DOPAC]], a metabolic breakdown product of [[dopamine]], have been shown to occur in striatal isolated nerve endings as a result of exposure to vinpocetine.<ref name=pmid11478921>{{Cite journal|author=Trejo F, Nekrassov V, Sitges M |title=Characterization of vinpocetine effects on DA and DOPAC release in striatal isolated nerve endings |journal=Brain Research |volume=909 |issue=1–2 |pages=59–67 |year=2001 |pmid=11478921 |doi=10.1016/S0006-8993(01)02621-X}}</ref> Such an effect is consistent with the biogenic pharmacology of [[reserpine]], a structural relative of vinpocetine, which depletes catecholamine levels and causes [[Depression (mood)|depression]] as a side effect of the cardiovascular and anti-psychotic effects.<ref name=pmid11478921/> However, this effect tends to be reversible upon cessation of Vinpocetine administration, with full remission typically occurring within 3–4 weeks. | |||
==Side effects== | |||
Vinpocetine is generally well-tolerated in humans.<ref name="about.com">{{Cite web|url=http://thyroid.about.com/cs/alternativehelp/a/vinpocetine.htm |title=Is Vinpocetine the Answer to Brain Fog, Cognitive and Memory Problems? |publisher=about.com |accessdate=2011-06-30}}</ref> No serious side effects have thus far been noted in clinical trials,<ref>{{Cite web|url=http://www.foundhealth.com/vinpocetine/side-effects-and-warnings |title=Vinpocetine Side Effects and Warnings |publisher=foundhealth |accessdate=2011-07-02}}</ref> although none of these trials were long-term.<ref name=Szatmari2003/> Some users have reported headaches, especially at doses above 15 milligrams per day, as well as occasional upset stomach. Adverse drug-herb interactions have not been prevalent, and vinpocetine appears safe to take with other medications, including diabetes drugs, as well as blood thinners like [[Coumadin]].<ref name="about.com"/> However, it should be carefully noted that the safety of vinpocetine in pregnant women has not been evaluated. Vinpocetine has been implicated in one case to induce [[agranulocytosis]],<ref>Shimizu Y, Saitoh K, Nakayama M, et al. [http://www.priory.com/med/vinpocetine.htm Agranulocytosis induced by vinpocetine]. Medicine Online, Retrieved March 08, 2008.</ref> a condition in which [[granulocytes]] are markedly decreased. Some people have anecdotally noted that their continued use of vinpocetine reduces immune function. [[Commission E]] warned that vinpocetine reduced immune function and could cause [[apoptosis]] (cellular death) in the long term.<ref>''The Complete German Commission E Monographs, Therapeutic Guide to Herbal Medicines'', 1st ed. 1998, Integrative Medicine Communications, pub; Bk&CD-Rom edition, 1999.{{Page needed|date=September 2010}}</ref> | |||
==References== | ==References== | ||
{{Reflist|2}} | |||
==External links== | |||
* [http://www.cochrane.org/reviews/en/ab003119.html Vinpocetine for cognitive impairment and dementia] - [[Cochrane Collaboration|Cochrane]] review | |||
* [[Erowid]] [http://www.erowid.org/smarts/vinpocetine/ Vinpocetine Vault] | |||
{{Stimulants}} | {{Stimulants}} | ||
{{Nootropics}} | |||
{{Phosphodiesterase inhibitors}} | {{Phosphodiesterase inhibitors}} | ||
[[Category:Nootropics]] | [[Category:Nootropics]] | ||
[[Category: | [[Category:PDE1 inhibitors]] | ||
[[Category:Vasodilators]] | [[Category:Vasodilators]] | ||
[[Category:Ethyl esters]] | |||
[[Category:Vinca alkaloids]] | |||
[[ | |||
[[ | |||
Latest revision as of 20:44, 20 January 2015
 | |
| File:Vinpocetine ball-and-stick.png | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| Routes of administration | Oral, intravenous |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 56.6 +/- 8.9% |
| Metabolism | hepatic |
| Elimination half-life | 2.54 +/- 0.48 hours |
| Excretion | renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C22H26N2O2 |
| Molar mass | 350.454 g/mol |
| 3D model (JSmol) | |
| |
| |
| | |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Vinpocetine (brand names: Cavinton, Intelectol; chemical name: ethyl apovincaminate) is a semisynthetic derivative of the vinca alkaloid vincamine (sometimes described as "a synthetic ethyl ester of apovincamine"),[1] an extract from the lesser periwinkle plant. Vinpocetine was first isolated from the plant in 1975 by the Hungarian chemist Csaba Szántay. The mass production of the synthetic drug was started in 1978 by the Hungarian pharmaceutical company Richter Gedeon.
Vinpocetine is reported to have cerebral blood-flow enhancing[2] and neuroprotective effects,[3] and is used as a drug in Eastern Europe for the treatment of cerebrovascular disorders and age-related memory impairment.[4]
Vinpocetine is not approved in the United States for pharmaceutical use, but it can be sold as a dietary supplement. Vinpocetine is widely marketed as a supplement for vasodilation and as a nootropic for the improvement of memory and cerebral metabolism. Vinpocetine has been identified as a potent anti-inflammatory agent that might have a potential role in the treatment of Parkinson's disease and Alzheimer's disease.[5][6]
Controlled clinical trials
As of 2003 only three controlled clinical trials had tested "older adults with memory problems".[7] However, a 2003 Cochrane review determined that the results were inconclusive.[8]
Prior to 2003, a different study from 1985[9] had tested young, healthy adults, but this study had 12 subjects and used a short treatment period.[7]
Use as a vasodilator
Vinpocetine is widely used in the body building community as a vasodilator. Although no studies have been conducted on the effectiveness of vinpocetine on performance enhancement during exercise, both beneficial and adverse effects have been reported on body building forums.[citation needed]
Anticonvulsant potential
Kindling models in rats has shown Vinpocetine to exhibit anticonvulsant properties, the most pronounced anticonvulsant effects were observed in Pentylenetetrazole (PTZ)-kindled rats although there was also an effect on amygdala-kindled and neocortically-kindled rats.[10] Vinpocetine has also been shown to abolished [3H]Glu release after in vivo exposure to 4-aminopyridine (4-AP) which suggests an important mechanism for vinpocetine anticonvulsant potential.[11]
Anti-inflammatory action
Vinpocetine has been identified as a novel anti-inflammatory agent.[5][6] Vinpocetine inhibits the up-regulation of NF-κB by TNFα in various cell tests. Reverse transcription polymerase chain reaction also shows that it reduced the TNFα-induced expression of the mRNA of proinflammatory molecules such as interleukin-1 beta, monocyte chemoattractant protein-1 (MCP-1), and vascular cell adhesion molecule-1 (VCAM-1). In mice, vinpocetine reduced lipopolysaccharide inoculation induced polymorphonuclear neutrophil infiltration into the lung.[5][6] Neuroinflammatory processes can result in neuronal death in Parkinson's disease (PD) and Alzheimer's disease (AD). It has been suggested that "it would be interesting to test whether vinpocetine’s antiinflammatory properties would have a protective effect in models of neurodegenerative conditions such as AD and PD."[6]
Mechanism of action
Vinpocetine has been shown to selectively inhibit voltage-sensitive Na+ channels, resulting in a dose-dependent decrease in evoked extracellular Ca+ ions in striatal nerve endings.[12] The Na+ channel inhibiting properties of vinpocetine are thought to contribute to a general neuroprotective effect through blockade of excitotoxicity and attenuation of neuronal damage induced by cerebral ischemia/reperfusion.[13]
Vinpocetine is also a phosphodiesterase (PDE) type-1 inhibitor,[14] (with an IC50 of approximately 10−5 M.) leading to increases in intracellular levels of cyclic guanosine 3'5'-monophosphate (cGMP), an action that causes the vasorelaxant effects of vinpocetine on cerebral smooth muscle tissue.[15][16]
Independent of vinpocetine's action on PDE, vinpocetine inhibits IKK preventing IκB degradation and the following translocation of NF-κB to the cell nucleus.[5][6]
Increases in neuronal levels of DOPAC, a metabolic breakdown product of dopamine, have been shown to occur in striatal isolated nerve endings as a result of exposure to vinpocetine.[17] Such an effect is consistent with the biogenic pharmacology of reserpine, a structural relative of vinpocetine, which depletes catecholamine levels and causes depression as a side effect of the cardiovascular and anti-psychotic effects.[17] However, this effect tends to be reversible upon cessation of Vinpocetine administration, with full remission typically occurring within 3–4 weeks.
Side effects
Vinpocetine is generally well-tolerated in humans.[18] No serious side effects have thus far been noted in clinical trials,[19] although none of these trials were long-term.[8] Some users have reported headaches, especially at doses above 15 milligrams per day, as well as occasional upset stomach. Adverse drug-herb interactions have not been prevalent, and vinpocetine appears safe to take with other medications, including diabetes drugs, as well as blood thinners like Coumadin.[18] However, it should be carefully noted that the safety of vinpocetine in pregnant women has not been evaluated. Vinpocetine has been implicated in one case to induce agranulocytosis,[20] a condition in which granulocytes are markedly decreased. Some people have anecdotally noted that their continued use of vinpocetine reduces immune function. Commission E warned that vinpocetine reduced immune function and could cause apoptosis (cellular death) in the long term.[21]
References
- ↑ Lörincz C, Szász K, Kisfaludy L (1976). "The synthesis of ethyl apovincaminate". Arzneimittel-Forschung. 26 (10a): 1907. PMID 1037211.
- ↑ PMID 15760651 (PMID 15760651)
Citation will be completed automatically in a few minutes. Jump the queue or expand by hand - ↑ PMID 12498034 (PMID 12498034)
Citation will be completed automatically in a few minutes. Jump the queue or expand by hand - ↑ "Vinpocetine. Monograph" (PDF). Alternative Medicine Review. 7 (3): 240–3. 2002. PMID 12126465.
- ↑ 5.0 5.1 5.2 5.3 PMID 20448200 (PMID 20448200)
Citation will be completed automatically in a few minutes. Jump the queue or expand by hand - ↑ 6.0 6.1 6.2 6.3 6.4 PMID 20495091 (PMID 20495091)
Citation will be completed automatically in a few minutes. Jump the queue or expand by hand - ↑ 7.0 7.1 McDaniel MA, Maier SF, Einstein GO (2003). "'Brain-specific' nutrients: a memory cure?". Nutrition. 19 (11–12): 957–75. doi:10.1016/S0899-9007(03)00024-8. PMID 14624946.
- ↑ 8.0 8.1 Szatmari SZ, Whitehouse PJ (2003). Szatmári, Szabolcs, ed. "Vinpocetine for cognitive impairment and dementia". Cochrane Database of Systematic Reviews (1): CD003119. doi:10.1002/14651858.CD003119. PMID 12535455.
- ↑ Subhan Z, Hindmarch I (1985). "Psychopharmacological effects of vinpocetine in normal healthy volunteers". European Journal of Clinical Pharmacology. 28 (5): 567–71. doi:10.1007/BF00544068. PMID 3899677.
- ↑ Schmidt, J (1990). "Comparative studies on the anticonvulsant effectiveness of nootropic drugs in kindled rats". Biomedica biochimica acta. 49 (5): 413–9. PMID 2271012.
- ↑ Sitges, M; Sanchez-Tafolla, BM; Chiu, LM; Aldana, BI; Guarneros, A (2011). "Vinpocetine inhibits glutamate release induced by the convulsive agent 4-aminopyridine more potently than several antiepileptic drugs". Epilepsy research. 96 (3): 257–66. doi:10.1016/j.eplepsyres.2011.06.006. PMID 21737246.
- ↑ Sitges M, Galván E, Nekrassov V (2005). "Vinpocetine blockade of sodium channels inhibits the rise in sodium and calcium induced by 4-aminopyridine in synaptosomes". Neurochemistry International. 46 (7): 533–40. doi:10.1016/j.neuint.2005.02.001. PMID 15843047.
- ↑ Adám-Vizi V (2000). "[Neuroprotective effect of sodium channel blockers in ischemia: the pathomechanism of early ischemic dysfunction]". Orvosi Hetilap (in Hungarian). 141 (23): 1279–86. PMID 10905082.
- ↑ Hagiwara M, Endo T, Hidaka H (1984). "Effects of vinpocetine on cyclic nucleotide metabolism in vascular smooth muscle". Biochemical Pharmacology. 33 (3): 453–7. doi:10.1016/0006-2952(84)90240-5. PMID 6322804.
- ↑ Truss MC, Uckert S, Stief CG, Forssmann WG, Jonas U (1996). "Cyclic nucleotide phosphodiesterase (PDE) isoenzymes in the human detrusor smooth muscle. II. Effect of various PDE inhibitors on smooth muscle tone and cyclic nucleotide levels in vitro". Urological Research. 24 (3): 129–34. doi:10.1007/BF00304075. PMID 8839479.
- ↑ Gurkovskaia AV, Gokina NI, Buryĭ VA, Shuba MF (1987). "[Electrophysiological analysis of the action of kavinton on the smooth muscles]". Biulleten' Eksperimental'noĭ Biologii I Meditsiny (in Russian). 103 (1): 68–71. PMID 3801654.
- ↑ 17.0 17.1 Trejo F, Nekrassov V, Sitges M (2001). "Characterization of vinpocetine effects on DA and DOPAC release in striatal isolated nerve endings". Brain Research. 909 (1–2): 59–67. doi:10.1016/S0006-8993(01)02621-X. PMID 11478921.
- ↑ 18.0 18.1 "Is Vinpocetine the Answer to Brain Fog, Cognitive and Memory Problems?". about.com. Retrieved 2011-06-30.
- ↑ "Vinpocetine Side Effects and Warnings". foundhealth. Retrieved 2011-07-02.
- ↑ Shimizu Y, Saitoh K, Nakayama M, et al. Agranulocytosis induced by vinpocetine. Medicine Online, Retrieved March 08, 2008.
- ↑ The Complete German Commission E Monographs, Therapeutic Guide to Herbal Medicines, 1st ed. 1998, Integrative Medicine Communications, pub; Bk&CD-Rom edition, 1999.[page needed]
External links
Template:Stimulants
Template:Nootropics
Template:Phosphodiesterase inhibitors
- Pages with script errors
- CS1 maint: Multiple names: authors list
- Pages with incomplete PMID references
- CS1 maint: Unrecognized language
- Wikipedia articles needing page number citations from September 2010
- Articles with invalid date parameter in template
- Pages with broken file links
- Template:drugs.com link with non-standard subpage
- Drugs with non-standard legal status
- Articles with changed CASNo identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Chemical pages without DrugBank identifier
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- All articles with unsourced statements
- Articles with unsourced statements from September 2013
- Nootropics
- PDE1 inhibitors
- Vasodilators
- Ethyl esters
- Vinca alkaloids