Mesuximide
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Consumer Drug Information |
| MedlinePlus | a682028 |
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Hepatic (demethylation and glucuronidation) |
| Elimination half-life | 1.4–2.6 hours (mesuximide) 28–38 hours (active metabolite) |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
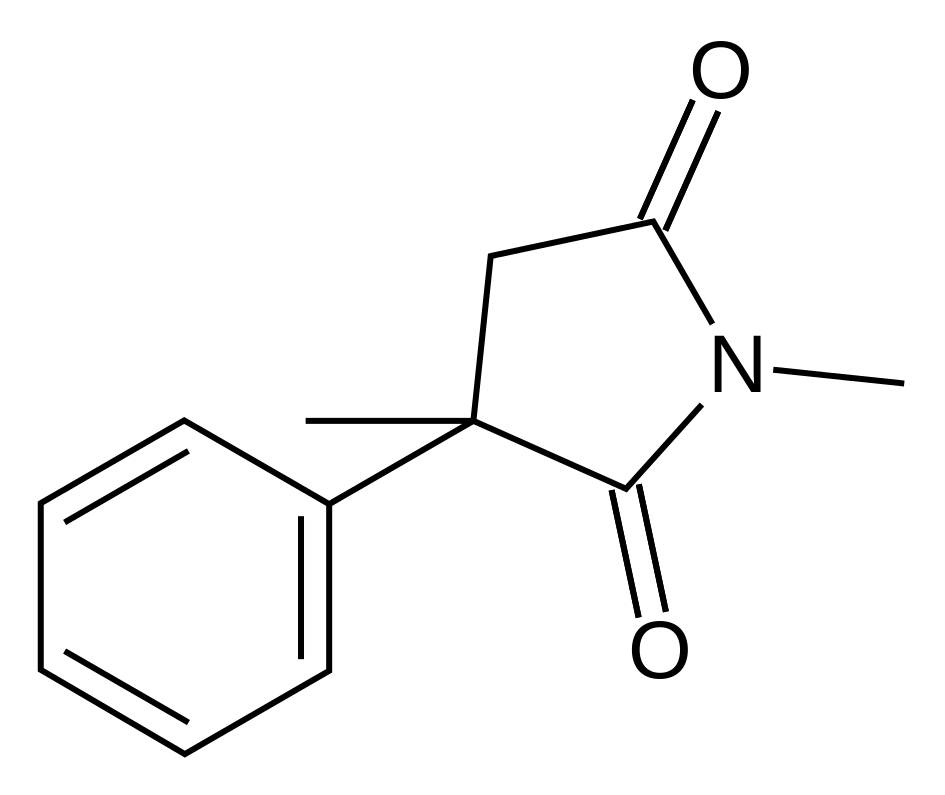
| Formula | C12H13NO2 |
| Molar mass | 203.237 g/mol |
| 3D model (JSmol) | |
| |
| |
| | |
|
WikiDoc Resources for Mesuximide |
|
Articles |
|---|
|
Most recent articles on Mesuximide |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Mesuximide at Clinical Trials.gov Clinical Trials on Mesuximide at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Mesuximide
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Mesuximide Discussion groups on Mesuximide Patient Handouts on Mesuximide Directions to Hospitals Treating Mesuximide Risk calculators and risk factors for Mesuximide
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Mesuximide |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Mesuximide (or methsuximide, methosuximide) is a succinimide anticonvulsant medication. It is sold as a racemate by Pfizer under the tradenames Petinutin (Switzerland)[1] and Celontin (United States).[2] The therapeutic efficacy of methosuximide is largely due to its pharmacologically active metabolite, N-desmethylmethosuximide, which has a longer half-life and attains much higher plasma levels than its parent.[3]
References
- ↑ Pfizer AG (2005). "Petinutin (Mésuximide)". Official Pfizer AG Website (in French). Retrieved August 21, 2006. [dead link]
- ↑ Pfizer Inc. (2008). "Celontin (methsuximide capsules, USP)". Official Pfizer Inc. Website. Retrieved November 21, 2014.
- ↑ Porter RJ, Penry JK, Lacy JR, Newmark ME, Kupferberg HJ. Plasma concentrations of phensuximide, methosuximide, and their metabolites in relation to clinical efficacy. Neurology 29: 1509-1513, 1979.
- Pages with script errors
- CS1 maint: Unrecognized language
- All articles with dead external links
- Articles with dead external links from November 2010
- Articles with invalid date parameter in template
- Template:drugs.com link with non-standard subpage
- Articles with changed DrugBank identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Infobox drug articles with non-default infobox title
- Drugboxes which contain changes to verified fields
- Anticonvulsants
- Drug
- Anticonvulsant stubs